Subfamily: Alpharhabdovirinae
Genus: Ephemerovirus
Distinguishing features
Viruses assigned to the genus Ephemerovirus form a distinct monophyletic group based on well-supported Maximum Likelihood or Maximum Clade Credibility trees inferred from complete L sequences. The ephemerovirus clade is part of a larger phylogenetic group of arthropod-borne rhabdoviruses with large and complex genomes that also includes hapaviruses, tibroviruses and curioviruses. All ephemeroviruses have multiple accessory genes between the G gene and L gene, including: i) a gene encoding a large non-structural glycoprotein (GNS); and ii) a gene encoding a class 1a viroporin (α1 or U1). Viruses in the genus cross-react strongly in complement-fixation (CF) and indirect immunofluorescence tests.
Virion
Morphology
Bullet-shaped or cone-shaped bovine ephemeral fever virus (BEFV; species Ephemerovirus febris) virions (140–200 nm × 60–80 nm) have been observed in negatively-stained preparations of infected culture supernates and in ultrathin sections of infected cells. Virions display a prominent axial channel intruding from the base and a precisely coiled, helical nucleocapsid with 35 cross-striations at intervals of 4.8 nm (Lecatsas et al., 1969, Holmes and Doherty 1970, Murphy et al., 1972b).
Physicochemical and physical properties
BEFV virions have a buoyant density in CsCl of 1.19 g cm−3 and sedimentation coefficient of 625 S. BEFV is sensitive to acid and alkali; the virus is most stable at pH 7.0–8.0 (Tanaka et al., 1969, Heuschele 1970, Della-Porta and Brown 1979).
Nucleic acid
Ephemerovirus genomes consist of a single molecule of negative-sense, single-stranded RNA ranging from 14.5 kb to 16.1 kb (Walker et al., 1992, Blasdell et al., 2012a, Blasdell et al., 2012b, Walker et al., 2015).
Proteins
Ephemerovirus N, P, M, G and L share sequence homology and/or structural characteristics with the cognate proteins of other rhabdoviruses (Walker et al., 1991, Walker et al., 2011). Other ephemerovirus proteins encoded in the genome have not been identified definitively in virions. The GNS glycoproteins are non-structural and range from 534–609 amino acids (unprocessed range 62.0–71.0 kDa); they share significant amino acid sequence identity with G and appear to have arisen by gene duplication (Walker et al., 1992, Wang and Walker 1993). The α1 proteins (also designated U1) are class 1a viroporins (Joubert et al., 2014); they range from 88 to 108 amino acids (10.6–12.5 kDa) and feature an N-terminal domain containing large hydrophobic residues, a central transmembrane domain and a highly basic C-terminal domain. The α2 proteins (also designated U1x) range from 92 to 116 amino acids (10.7–14.1 kDa); the β proteins (also designated U2) range from 146 to 157 amino acids (16.5–18.5 kDa); the γ proteins range from 100 to 115 amino acids (11.7–13.8 kDa); and the kotonkan virus (KOTV; species Ephemerovirus kotonkan) and Koolpinyah virus (KOOLV; species Ephemerovirus koolpinyah) δ proteins are each 109 amino acids (12.2 and 12.4 kDa, respectively). Yata virus (YATV; species Ephemerovirus yata) proteins encoded in the unique alternative ORFs U2 and U2x are, respectively, 123 amino acids (14.4 kDa) and 53 amino acids (6.1 kDa). Other than the α1 proteins, the sequences of small ephemerovirus accessory proteins reveal no remarkable structural characteristics and are only highly conserved between closely related viruses; their functions are not known (Walker et al., 2015).
Carbohydrates
The envelope glycoprotein (G) and the large non-structural glycoprotein (GNS) are each glycosylated and contain multiple predicted sites for N-linked glycosylation (Walker et al., 1991, Walker et al., 1992, Wang and Walker 1993, Blasdell et al., 2012a, Blasdell et al., 2012b, Walker et al., 2015).
Genome organisation and replication
Ephemerovirus genomes include five genes (N, P, M, G and L) encoding the structural proteins and multiple additional long ORFs between the G gene and L gene (Walker et al., 2015) (Figure 1 Ephemerovirus). In all ephemeroviruses, an ORF encoding a non-structural glycoprotein (GNS) lies in an independent transcriptional unit immediately following the G gene. The GNS gene is then followed by an ORF (α1 or U1) encoding a class 1a viroporin. In most ephemeroviruses, the α1 ORF is followed by a second consecutive ORF (α2 or U1x) within the same transcriptional unit. Overlapping termination and initiation codons and an upstream ‘termination upstream ribosome-binding site’ (TURBS) indicate that α2 is likely to be expressed by a stop-start mechanism of translation. The α1/α2 gene is followed by up to three additional ORFs (β or U2; γ or U3; and δ or U4), each in an independent transcriptional unit. However, some ephemeroviruses lack the δ gene and others lack both the γ gene and δ gene. YATV lacks the α2 (U1x) ORF but has an additional independent transcriptional unit containing two alternative ORFs between the α1 and β genes; the proteins encoded in these ORFs appear unrelated to other ephemerovirus proteins. In all ephemeroviruses, ORFs in independent transcriptional units are flanked by conserved transcription initiation (AACAG) and transcription termination/polyadenylation (TGAAAAAAA) sequences.
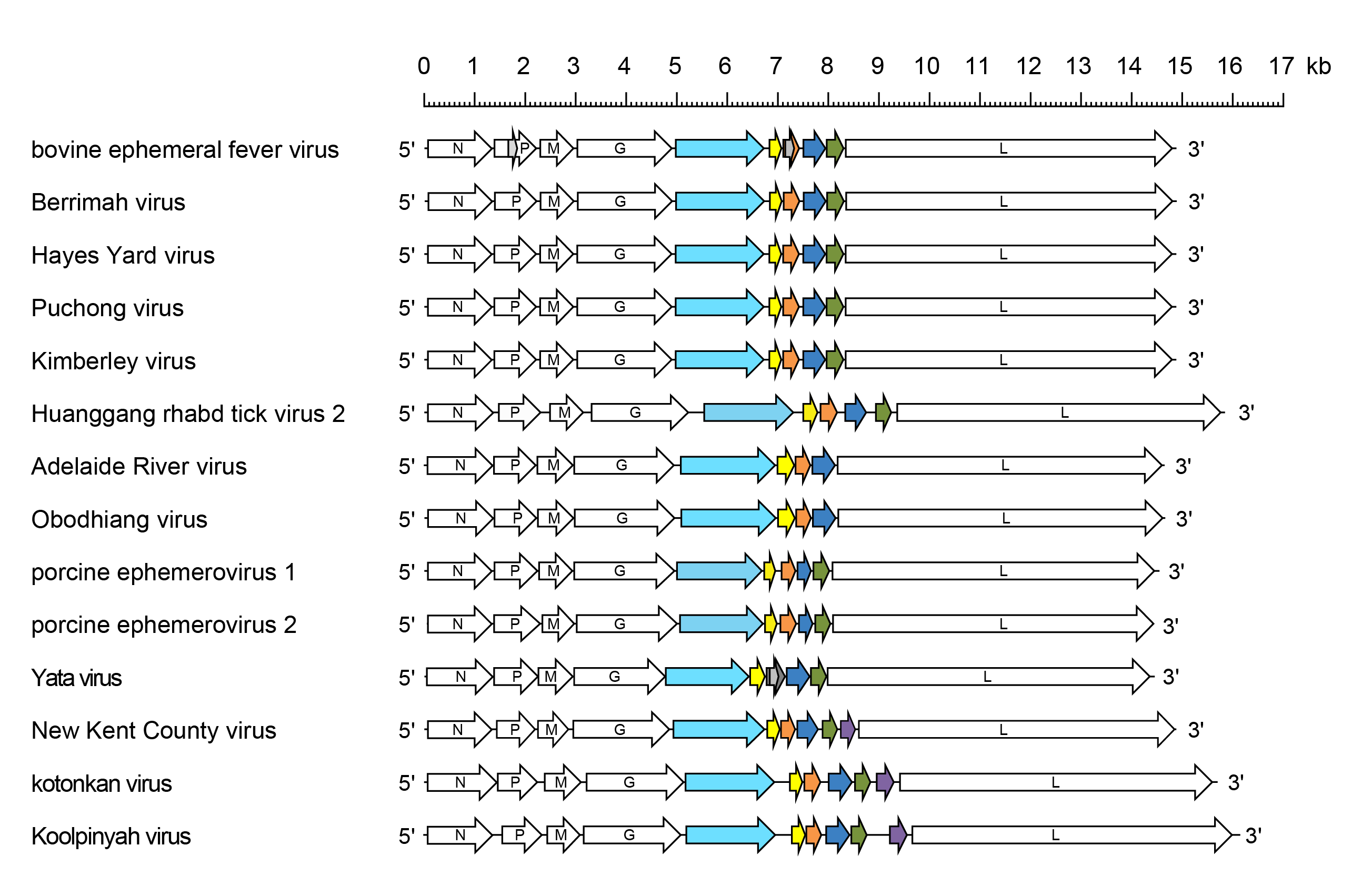 |
| Figure 1 Ephemerovirus. Schematic representation of ephemerovirus genomes shown in reverse (positive-sense) polarity. N, P, M, G and L represent ORFs encoding the structural proteins. The GNS (light blue), α1 (yellow), α2 (orange), β (dark blue), γ (green) and δ (purple) ORFs are highlighted. The GNS ORF encodes a non-structural class I transmembrane glycoprotein; the α1 ORF encodes a class 1a viroporin; other ORFs in the region between the G gene and L gene (grey and black) encode proteins of unknown function. |
Biology
Ephemeroviruses have been isolated exclusively from cattle, pigs and haematophagous insects (mosquitoes and biting midges) (Walker and Klement 2015, Wu et al., 2022). Two ephemeroviruses (New Kent County virus, species Ephemerovirus kent and Huanggang rhabd tick virus 2, species Ephemerovirus huanggang) have been detected by metagenomic sequencing of ticks from the USA and China, but it is not known if these were due to active infections of the ticks (Tokarz et al., 2018). Bovine ephemeral fever is an economically important disease of cattle and water buffalo in most tropical and sub-tropical regions of Africa, Australia, and Asia. BEFV infection causes a sudden onset of fever and other clinical signs including lameness, anorexia and ruminal stasis, followed by a sustained drop in milk production. Although the mortality rate is usually low (1–2%), it is highest in well-conditioned beef cattle and high-producing dairy cattle (Walker 2005, Walker and Klement 2015). KOTV has also been associated with an outbreak of ephemeral fever in Africa (Kemp et al., 1973, Tomori et al., 1974) and Hayes Yard virus (HYV; species Ephemerovirus hayes) was isolated from a bull (Bos indicus) with a severe ephemeral fever-like illness (Blasdell et al., 2020). Porcine ephemerovirus 1 (PoEV1; species Ephemerovirus henan) and porcine ephemerovirus 2 (PoEV2; species Ephemerovirus guangdong) were detected by high throughput sequencing of archived tissue samples (kidney, spleen and lymph node) collected in China from pigs suffering classical swine fever (Wu et al., 2022). Other members of the genus are not recognised as animal pathogens; however, most have been isolated from healthy sentinel cattle or are known to infect cattle.
Antigenicity
Ephemeroviruses cross-react strongly in CF or indirect immunofluorescence tests; they may also show low level cross-reactions by indirect immunofluorescence with viruses of the genus Lyssavirus (Calisher et al., 1989). However, sequence comparisons with other rhabdoviruses indicate that, in evolutionary terms, the ephemeroviruses are closer to tibroviruses, hapaviruses, curioviruses and vesiculoviruses than to lyssaviruses (Walker et al., 2015). There is only one known BEFV serotype worldwide, with virus isolates from different regions (Australia, Asia, Africa) displaying significant cross-neutralisation (Walker and Klement 2015). There is a low level of cross-neutralisation between BEFV and Berrimah virus (BRMV; species Ephemerovirus berrimah), between Adelaide River virus (ARV; species Ephemerovirus adelaide) and Obodhiang virus (OBOV; species Ephemerovirus obodhiang) (Gard et al., 1983, Blasdell et al., 2012a) and KOOLV and KOTV (Blasdell et al., 2014); no significant cross-neutralisation between other ephemeroviruses has been reported. BEFV G contains four distinct neutralisation sites (Cybinski et al., 1990, Kongsuwan et al., 1998). BEFV G purified from virions or expressed from recombinant vaccinia virus protects cattle from experimental infection (Uren et al., 1994, Hertig et al., 1996). The GNS glycoprotein does not induce neutralising antibodies and is not protective (Hertig et al., 1996, Johal et al., 2008).
Species demarcation criteria
Viruses assigned to different species within the genus Ephemerovirus have several of the following characteristics: A) minimum amino acid sequence divergence of 15% in the L proteins; B) minimum amino acid sequence divergence of 8% in the N proteins; C) can be distinguished in serological tests; and D) significant differences in genome organisation as evidenced by numbers and locations of ORFs.
Related, unclassified viruses
| Virus name | Accession number
| Virus abbreviation
|
| Hefer Valley virus | OQ679991 | HVV |
| Kokolu virus | OU592964 | KOKOV |
| Mavingoni virus | MN148799* | MVGV |
Virus names and virus abbreviations are not official ICTV designations.
* Coding region sequence incomplete

