Family: Rhabdoviridae
Genus: Betaricinrhavirus
Distinguishing features
Viruses assigned to the genus Betaricinrhavirus form a distinct monophyletic group based on well-supported Maximum Likelihood or Maximum Clade Credibility trees inferred from complete L sequences. Members of the genus have been detected in hard ticks (Acari: Ixodidae). They are distinct phylogenetically from viruses assigned to the genus Alpharicinrhavirus.
Virion
Morphology
Viruses assigned to the genus have not yet been isolated or visualized by electron microscopy.
Nucleic acid
Betaricinrhavirus genomes consist of a single molecule of negative-sense, single-stranded RNA and range from approximately 13.7–13.8 kb (Tokarz et al., 2018).
Proteins
Betaricinrhavirus N, P, M, G and L proteins share sequence homology and/or structural characteristics with the cognate proteins of other rhabdoviruses. Betaricinrhavirus G proteins are class I transmembrane glycoproteins. Alignment with the G protein of vesicular stomatitis Indiana virus (species Vesiculovirus indiana) indicates likely conservation of 10-12 of the 12 conserved cysteine residues that are typical of animal rhabdovirus G proteins (Walker and Kongsuwan 1999, Roche et al., 2006).
Genome organisation and replication
Betaricinrhavirus genomes include only the five genes (N, P, M, G and L) encoding the structural proteins (Figure 1.Betaricinrhavirus). In both Chimay rhabdovirus (CRV; species Betaricinrhavirus chimay) and blacklegged tick rhabdovirus 1 (BLTRV-1; species Betaricinrhavirus scapularis), there is an alternative overlapping ORF (Nx) in the N gene and an alternative ORF (Px) in the P gene. Although there is sequence identity between proteins encoded in the corresponding alternative ORFs, it is not known if they are expressed in infected cells.
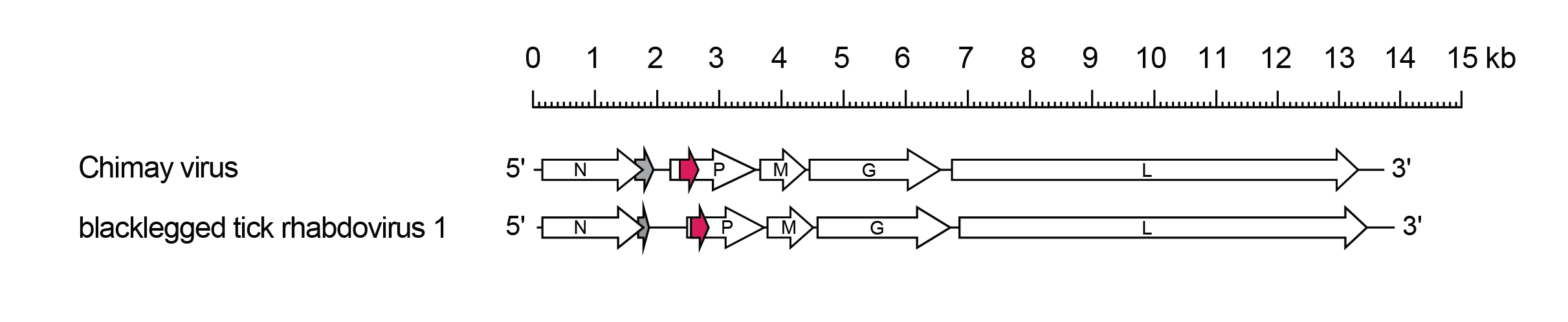 |
| Figure 1.Betaricinrhavirus. Schematic representation of betaricinrhaviruses genomes in reverse (positive-sense) polarity. The five long open reading frames (ORFs) in the N, P, M, G and L genes are shown (open arrows). Alternative overlapping ORFs (Nx) at the end of each N gene (grey) and alternative ORFs (Px) within each P gene (red) are shown. |
Biology
CRV was detected in a pool of hard ticks (Ixodes ricinus) sampled in Belgium, in 2009. BLTRV-1 was detected in pools of hard ticks (Ixodes scapularis) sampled in Connecticut and New York, USA, in 2016 (Tokarz et al., 2018). No isolates are currently available for either of these viruses.
Species demarcation criteria
Viruses assigned to different species within the genus have several of the following characteristics: A) minimum amino acid sequence divergence of 10% in N proteins; B) minimum amino acid sequence divergence of 10% in the L proteins; C) minimum amino acid sequence divergence of 15% in G proteins; D) significant differences in genome organisation as evidenced by numbers and locations of ORFs; E) can be distinguished in virus neutralisation tests; and F) occupy different ecological niches as evidenced by differences in arthropod and/ or vertebrate hosts.
Related, unclassified viruses
| Virus name | Accession number | Virus abbreviation |
| Fairlight virus | MK026565* | FLTV |
| Mudanjiang rhabd tick virus 1 | ON746525 | MjRTV1 |
| Quarantine Head virus | MK026568* | QHV |
| Tongren rhabd tick virus 1 | ON746523 | TrRTV1 |
| Yanbian rhabd tick virus 2 | ON746528* | YbRTV2 |
| Yanbian rhabd tick virus 3 | ON746524 | YbRTV3 |
Virus names and virus abbreviations are not official ICTV designations.
* Coding region sequence incomplete

