Order: Reovirales
Jelle Matthijnssens, Houssam Attoui, Krisztián Bányai, Corina P. D. Brussaard, Pranav Danthi, Mariana del Vas, Terence S. Dermody, Roy Duncan, Qín Fāng (方勤), Reimar Johne, Peter P. C. Mertens, Fauziah Mohd Jaafar, John Patton, Takahide Sasaya (笹谷孝英), Nobuhiro Suzuki (鈴木信弘) and Taiyun Wei (魏太云)
The citations for this ICTV Report chapter are the summaries published as Matthijnssens et al., (2022) ICTV Virus Taxonomy Profile: Sedoreoviridae, Journal of General Virology (2022) 103:001782 and Matthijnssens et al., (2022) ICTV Virus Taxonomy Profile: Spinareoviridae, Journal of General Virology (2022) 103:001781.
Corresponding author: Jelle Matthijnssens ([email protected])
Edited by: Jens H. Kuhn and Stuart G. Siddell
Posted: October 2021, updated June 2022, May 2025
Summary
Members of the order Reovirales have linear dsRNA genomes and the family is divided into the two families Spinareoviridae and Sedoreoviridae, including, respectively, nine and seven genera (Table 1 Reovirales). These 16 genera include 103 species. The number of genome segments (9–12) is characteristic of viruses belonging to a single genus, although the genus Mycoreovirus includes viruses with either 11 or 12 segments. Other factors distinguishing different genera are host (and vector) range, disease signs, and capsid structure. Although sequences of homologous genome segments have, in many cases, diverged significantly between members of distinct genera, the amino acid sequence of the more conserved RNA-directed RNA polymerase (RdRP) can be used for comparison across taxonomic boundaries. Viruses belonging to individual species have higher levels of identity of conserved proteins and RNAs, including specific RNA packaging signals (Chapell et al., 1994). This high degree of functional and structural compatibility allows viable progeny virus strains to be generated by reassortment between viruses of the same species (Nibert et al., 1996).
Viruses classified in the order Reovirales have a broad host range, infecting mammals, fish, birds, reptiles, arthropods, algae, fungi, and plants. Some of these viruses have important pathogenic potential for humans (e.g., rotavirus A), livestock (e.g., bluetongue virus), and plants (e.g., rice dwarf virus). Virions are icosahedral (Dryden et al., 1993). The protein capsid is organized as 1–3 concentric layers of capsid proteins, with an overall diameter of 60–95 nm. The positive-sense strands of each viral RNA duplex are modified with a 5′-terminal type 1 cap structure but do not have a 3′-poly(A) tail (Furuichi et al., 1975). The viral RNAs are mostly monogenic with relatively short 5′- and 3′-non-coding regions (Chapell et al., 1994), although some segments have second or third functional protein-coding ORFs. Virion cell entry mechanisms vary between genera, but usually result in loss of outer-capsid components (Ebert et al., 2002). The resulting transcriptionally active particles are released into the cytoplasm. The 5′-capped, non-polyadenylated mRNAs are synthesized by structural enzymatic components of the particle and released through pores at the icosahedral apices of the virion into the cytoplasm. Viroplasms or viral inclusion bodies are distributed throughout the cytoplasm (Silverstein and Schur 1970). These neoorganelles are sites of viral mRNA synthesis, genome replication, and particle assembly. Sets of a single copy of each capped mRNA are incorporated into progeny virions. These mRNAs serve as templates for negative-sense strand synthesis, thereby reconstituting genomic encapsidated dsRNAs. The steps involved in virion morphogenesis and virion egress from cells differ depending on the genus. Progeny virions are released from some cell types without compromising cell viability (e.g., by budding) or from other cell types following lysis (Fernández de Castro et al., 2020).
Table 1 Reovirales Characteristics of members of the order Reovirales
| Characteristic | Description |
| Example | mammalian orthoreovirus 3, Dearing T3D (L1: HM159613; L2: HM159614; L3: HM159615; M1: HM159616; M2: HM159617; M3: HM159618; S1: HM159619; S2: HM159620; S3: HM159621; S4: HM159622), species Orthoreovirus mammalis |
| Virion | Nonenveloped, icosahedral, 60–95 nm virions composed of 1–3 concentric capsid proteins layers |
| Genome | Approximately 18–29 kbp of segmented (9–12) linear dsRNA, with. segments ranging from 0.7–5.8 kbp |
| Replication | Replication occurs in the cytoplasm in electron-dense structures called viroplasms or virus inclusion bodies |
| Translation | Translation occurs from full-length transcribed mRNAs, which possess a 5′-terminal cap but no poly(A)-tail. |
| Host Range | Mammals, aquatic animals, birds, reptiles, arthropods, algae, fungi, and plants |
| Taxonomy | Realm Riboviria, kingdom Orthornavirae, phylum Duplornaviricota, class Resentoviricetes, order Reovirales. The two families Spinareoviridae and Sedoreoviridae, include, respectively, nine (Aquareovirus, Coltivirus, Cypovirus, Dinovernavirus, Fijivirus, Idnoreovirus, Mycoreovirus, Orthoreovirus and Oryzavirus) and seven (Cardoreovirus, Crabreovirus, Mimoreovirus, Orbivirus, Phytoreovirus, Rotavirus and Seadornavirus) genera, collectively including 103 species |
Virion
Morphology
Members of the order Reovirales have particles with icosahedral symmetry but may appear spherical in shape. The protein capsid is organized as one, two, or three concentric layers of capsid proteins, which surround the linear dsRNA segments of the viral genome, with an overall diameter of 60–95 nm (Figure 1 Reovirales).
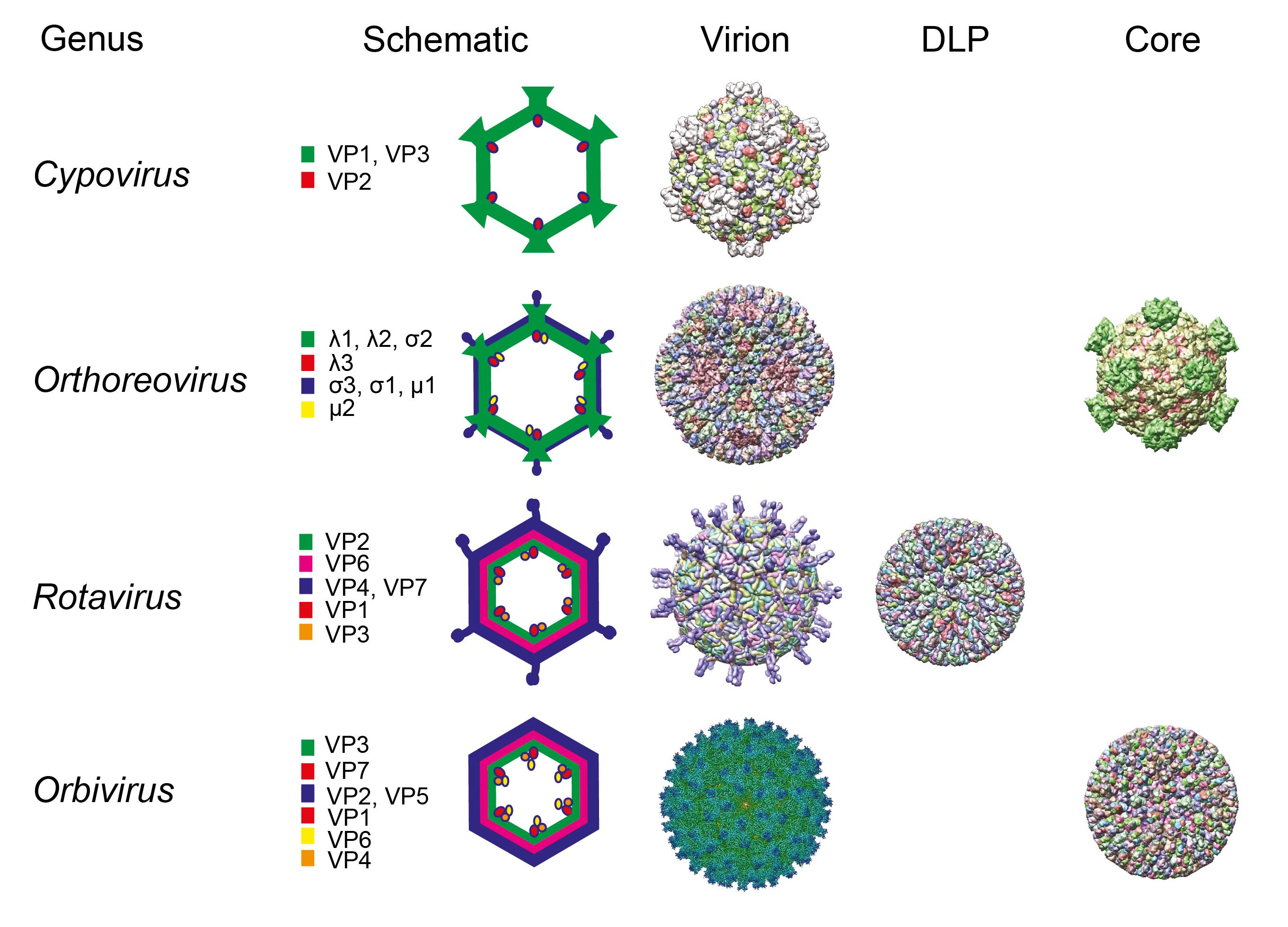 |
| Figure 1 Reovirales. Particle morphologies of members of different genera of the order Reovirales. Schematic representations, cryoEM reconstructions, or X-ray crystal structures of virions and subvirion particles (DLP: Double layered particle) of representative members of the order Reovirales are shown. For each schematic representation, core proteins are shown in green, middle layer (for rotaviruses and orbiviruses) is shown in magenta, and the outer capsid is shown in blue. The RdRP is shown in red, capping enzyme when not part of the turret structure (such as in rotaviruses and orbiviruses) is shown in orange, and helicase/NTPase is shown in yellow. Proteins that make up each protein shell are indicated. Cypoviruses (single layer, spiked core), orthoreoviruses (two layers, spiked core), and rotaviruses and orbiviruses (three layers, unspiked core) structures were obtained from the protein databank or EM data bank: (Bombyx mori cypovirus 1(3IZX), mammalian orthoreovirus 3 (2CSE, 1EJ6), rotavirus A (4V7Q, 3KZ4), and bluetongue virus (EMD-6444, 2BTV). The schematics and structures are not shown to scale. |
The 16 genera within the order Reovirales are divided between two families. Spinareoviridae includes viruses that have relatively large spikes or turrets situated at the 12 icosahedral vertices of either the virion or core particle. Sedoreoviridae includes viruses that do not have large surface projections on their virions or core particles, giving them an almost spherical or “smooth” appearance.
The terminology that has been used to describe particles of members of the order Reovirales with different numbers of capsid layers varies between genera. The current nomenclature will therefore be explained in each case. The transcriptionally active core particle of the spiked viruses (spinareovirids) appears to contain only a single complete capsid layer (which has been interpreted as having T=1 or T=2 symmetry) to which the projecting spikes or turrets are attached. In most cases, the core is surrounded (in the complete virion) by an incomplete protein layer (with T=13 symmetry) that forms the outer capsid, which is penetrated by the projections from the core surface (Dryden et al., 1993). These virions are therefore usually regarded as double-layered.
Exceptions are the cypoviruses, which have transcriptionally active but fully intact virions composed of only a single capsid shell that is equivalent to the core particles of viruses from other genera. However, virions of most cypoviruses are characteristically occluded (either singly or multiply) within the matrix of proteinaceous crystals called polyhedra. These are composed primarily (>90%) of the viral polyhedrin protein.
In contrast, virions of the non-spiked viruses (sedoreovirids) have an inner ‘subcore’ protein layer, which may be relatively fragile, having structural similarities to the innermost shell of the spiked viruses (interpreted as having T=2 symmetry). However, in transcriptionally active core particles, the subcore is surrounded and reinforced by a complete core-surface layer, which has T=13 symmetry. These double-layered cores have no surface spikes and (in intact virions) are surrounded by an outer-capsid shell, giving rise to three-layered virions that are equivalent to the two-layered particles of spinareovirids.
The innermost protein layer of particles of members of the order Reovirales has an internal diameter of approximately 50–60 nm and surrounds the 9, 10, 11, or 12 linear dsRNA genome segments. For sedereovirids, the enzymatically-active minor proteins of the virion are anchored to the inner surface of the central space at the five-fold axes of symmetry. These include the RdRP (which functions as a transcriptase and replicase), NTPase and helicase, and capping and transmethylase enzymes. However, for spinareovirids, some of these enzymatic proteins form turrets on the surface of the core. These hollow projections appear to act as conduits for the exit of nascent mRNA synthesized by the core-associated enzymes.
Particles of some members of the order Reovirales can leave infected cells by budding (e.g., orbiviruses and seadornaviruses) or can bud into the endoplasmic reticulum during morphogenesis (rotaviruses), acquiring an envelope derived from cellular membranes. However, in most cases, the envelope appears to be transient and is not usually considered to be part of the intact virion. The significance and role, if any, of these membrane envelopes has not been fully defined. In viruses of some genera, the protein components of the outer-capsid shell can be modified by proteases (e.g., trypsin or chymotrypsin) to form infectious or intermediate subvirion particles (ISVPs). ISVP formation may occur intracellularly (within endocytic vesicles, which represent an entry route for virions taken in from the cell surface) (Ebert et al., 2002), extracellularly (e.g., in the intestinal lumen following ingestion or peroral inoculation or in the host blood stream) (Bass et al., 1990) or in vitro following treatment with proteases (including those present in the saliva of insect vector species) (Nibert et al., 1995). The virion-to-ISVP transition can significantly increase, and may even be essential for, infectivity of these viruses.
Physicochemical and physical properties
The virion Mr is about 12×107. The buoyant density in CsCl is 1.36–1.39 g cm−3. Virion infectivity is moderately resistant to heat, organic solvents (e.g., ether), and non-ionic detergents (depending on both virus strain and detergent). The pH stability of virions varies among the genera.
Nucleic acid
Particles of members of the order Reovirales can contain 9, 10, 11, or 12 segments of linear dsRNA, depending on the genus. The individual Mr of these RNA molecules ranges from 0.45 to 3.77×106. The total Mr of the genome is 12–19×106. The RNA constitutes about 15–20% of the virion dry weight. The positive-sense strands of each duplex have a 5′-terminal type 1 cap structure (7mGpppN2’OmpNp…), which was first discovered in cypoviruses (Furuichi and Miura 1975). The negative-sense strands may have phosphorylated 5′-termini. However, in some cases (e.g., bluetongue virus, genus Orbivirus), the negative-sense strand is poorly labeled (with the same efficiency as the positive-sense strand) by treatment with polynucleotide kinase and [γ-32P] ATP, suggesting that it may also have a blocked 5′-structure. Both RNA strands have a 3′-OH group, and viral mRNAs lack 3′-poly(A) tails. The viral dsRNA segments are present within virions in equimolar proportions, representing one copy of each genome segment per virion. Intact virions of some genera also contain significant amounts of short ssRNA oligonucleotides.
Transfection of susceptible cells with plasmids encoding full-length viral RNAs or with viral mRNAs generated in vitro can lead to recovery of fully viable virions for several genera (Kobayashi et al., 2007), thus providing research opportunities using ‘reverse genetics’ technologies.
Proteins
At least three internal virion structural proteins have enzymatic activities involved in RNA synthesis and capping, including a fully conservative RdRP (which may function as a transcriptase, i.e., involved in positive-sense strand synthesis on a dsRNA template, or a replicase, i.e., involved in negative-sense strand synthesis on a positive-sense strand ssRNA template), nucleotide phosphohydrolase, guanylyltransferase, two distinct transmethylases, dsRNA unwinding (i.e., helicase) activity, and pyrophosphatase. Several of these functions (e.g., nucleotide phosphohydrolase, guanylyltransferase, and two distinct transmethylases) may be mediated by a single protein. Some of the minor proteins also may play a structurally significant role as components of the virion together with at least three major capsid proteins. The virion structural proteins range in mass from 15 to 155 kDa and constitute about 80–85% of the dry weight of virions.
Lipids
Mature virions are usually regarded as lacking a lipid envelope. Depending on the genus, a myristoyl residue may be covalently attached to one of the virion proteins (Nibert et al., 1991). Coltiviruses, rotaviruses, and orbiviruses have an intermediate in virion morphogenesis or release, which may have a lipid envelope that subsequently can be lost or removed. This may help to explain why virions are in some cases strongly associated with membrane fractions in infected cell lysates.
Carbohydrates
In members of some genera, one of the outer capsid proteins can be glycosylated with high mannose glycans or O-linked N-acetylglucosamine. A small non-structural (NS) viral protein may also be glycosylated.
Genome organization and replication
Most viral RNA segments are monogenic, although some segments have a second functional, in-frame initiation codon or additional, protein-encoding ORFs in an alternative-reading frame. Proteins are encoded by one strand only of each duplex (the positive-sense or mRNA strand). The mode of particle entry into cells varies between genera but usually results in loss of outer-capsid components. Transcriptionally active particles derived from the parental virions are released into the cytoplasm (Figure 2 Reovirales). Repetitive asymmetric transcription of each dsRNA segment occurs within these particles throughout the course of infection or until the infecting core particles reassociate with newly synthesized outer-capsid components. The mRNAs, which are produced in larger copy numbers from the shorter segments, are extruded from the icosahedral apices of these particles. This unequal level of mRNA synthesis suggests that each dsRNA seqment may be associated with only a single RdRP molecule that repeatedly re-initiates transcription of the same mRNA. Structures that have been called viroplasms or viral inclusion bodies (VIBs) occur in localized areas of the infected cell. They appear to be sites of viral mRNA synthesis, genome replication, and particle assembly (Tenorio et al., 2019). VIBs have a granular and moderately electron-dense appearance when viewed by electron microscopy and usually contain nascent subviral particles. Outer-capsid components appear to be added to progeny virions at the periphery of the VIB and are thought to stop further mRNA synthesis.
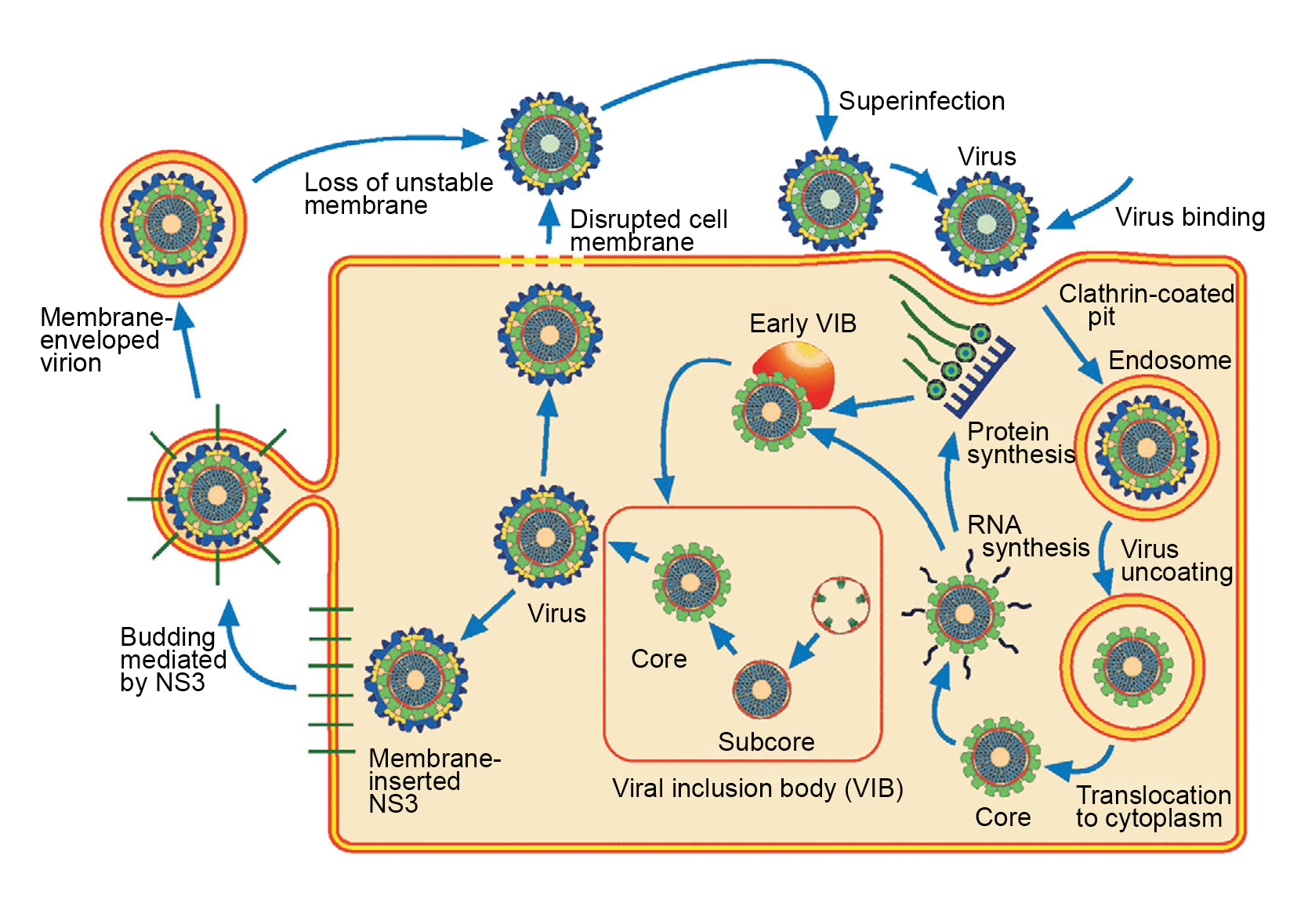 |
| Figure 2 Reovirales Replication cycle of a typical reovirus (presented for an orbivirus). |
The virus particle attaches to cell surface via the cell attachment outer capsid protein VP2 and is internalized by endocytosis. In the endosome, the virus particle is uncoated, releasisng the transcriptionally active core particle. The 10 segments of dsRNA remain contained with the cytoplasm. Within the core, all 10 dsRNA genome segments are transcribed [the transcription complex comprises three proteins VP1 (RdRP), VP4 (capping enzyme) and VP6 (ATPase and helicase)] into capped (but no polyadenylated) positive-sense mRNA. The 10 mRNAs are extruded into the cytoplasm where they are translated by ribosomes. Viral includion bodies (VIBs, largely made of the NS2 protein) are formed and are closely associated with the assembly of core paticles. This process involves the initial sorting all 10 mRNAs into neonascent core particles and transcription of the positive-sense strands into negative-sense strands, therby generating a dsRNA genome. Assembly of the outer capsid proteins VP2 and VP5 occurs at the periphery of the VIB and full virus particles leave the cell: i) either by budding, thus acquiring a transient evelope which is lost therfater, or ii) by cell lysis.
The mechanism of genome assembly and synthesis remains largely uncharacterized. For orthoreoviruses and rotaviruses, sets of capped mRNAs and certain NS proteins are incorporated into assortment complexes that are the precursors of progeny virions. These mRNAs are then used as templates for a single round of negative-sense strand synthesis, thereby reforming the dsRNA genome segments of a progeny virion. The various types of mRNAs are synthesized and present in the cytoplasm in non-equimolar ratios. However, the dsRNA genome segments are usually packaged in equimolar ratios (i.e., one copy of each genome segment per particle). The selection of viral mRNAs for packaging is, therefore, thought to be highly specific, involving recognition signals on each mRNA. There is inter-segment complementarity between short oligonucleotide regions near the termini of positive-sense strands corresponding to orbivirus genome segments 5 and 10 as well as between segments 1 and 7. This complementarity is essential for the successful recovery of viable virions using synthestic mRNAs and reverse genetics techniques. The sequence of these complementary regions also varies between different viruses and may mediate specific RNA-RNA interactions that identify mRNAs to be packaged. The shortest orbivirus genome segment is required to initiate viral RNA-RNA interactions with other short segments prior to recruitment of medium to long ssRNAs until a complete set of the viral RNA is packaged (Borodavka et al., 2018). Such strategies may be used by viruses of other genera but have not been confirmed.
Genome segment reassortment, involving the selection and packaging of mRNAs from different parental strains, occurs readily in cells that are co-infected with different viruses of the same species, which presumably share the same packaging signals.
RNA segments have conserved terminal sequences at both ends, which may serve as recognition signals for the viral transcriptase and replicase functions. These sequences may be essential for selection and incorporation of RNAs into nascent progeny particles and also function in efficient initiation of translation of the viral mRNAs. In many cases, sequences near the 5′- and 3′-termini of the positive-sense viral RNAs share extensive complementarity, interrupted by short sequences predicted to form stem loops and other secondary structures (Chapell et al., 1994, Borodavka et al., 2018). These findings and mounting experimental evidence suggest that the viral mRNAs contain both primary sequences and higher-order structures that regulate RNA function (i.e., translation, replication, or packaging). A consistent feature of the secondary structures predicted for viral positive-sense RNAs is that the conserved 5′- and 3′-terminal sequences remain non-hybridized. Indeed, non-hybridized 3′-terminal sequences are required for efficient negative-sense strand synthesis. At least in viruses of some genera, the dsRNA within assembled particles is packaged as a series of concentric and highly organized shells, which also have elements of icosahedral symmetry.
In addition to the parental cores, progeny cores (single- or double-layered particles from spinareovirids and sedoreovirids, respectively) also synthesize mRNAs, providing an amplification step for replication. Depending on the genus, some NS proteins are involved in translocation of virions within cells or virion egress by budding. Many cypoviruses also form polyhedra, which are large crystalline protein matrices that occlude virions (either singly or multiply) and which appear to be involved in transmission between individual insect hosts. The steps involved in virion morphogenesis and virion egress from cells vary according to genus. Members of the order Reovirales are the only known examples where non-enveloped virions induce cell-cell fusion and syncytium formation in virus-infected cells. In the case of fusogenic orthoreoviruses, syncytium formation promotes lysis and release of progeny virions.
Biology
The biological properties of the viruses vary according to genus. Some viruses replicate only in certain vertebrate hosts (e.g., orthoreoviruses, rotaviruses) and are transmitted between hosts by respiratory or fecal-oral routes. Other vertebrate viruses (e.g., coltiviruses, orbiviruses, seadornaviruses) replicate in both arthropod vectors (e.g., biting midges, mosquitoes, ticks) and vertebrate hosts. The recently described cardoreoviruses replicate in crustaceans (Chinese mitten crab) (Zhang et al., 2004). Plant viruses (e.g., fijiviruses, oryzaviruses, phytoreoviruses) replicate in both plants and arthropod vectors (e.g., leafhoppers, planthoppers) (Wei and Li 2016). Algal viruses (mimoreoviruses) replicate in specific marine photoautotrophic protists (Brussaard et al., 2004, Attoui et al., 2006). Viruses that infect insects (cypoviruses) are transmitted by contact or fecal-oral routes.
Antigenicity
Those viruses that infect vertebrate hosts generally possess both species-specific (sometimes also referred to as serogroup-specific) antigens (i.e., specific to a virus species), and more variable serotype-specific antigens (i.e., specific to groups of viruses within a species). Those viruses that infect plants and insects show greater uniformity and less antigenic variation in their proteins, possibly due to the lack of neutralizing antibodies in the host and, therefore, the absence of antibody selective pressure on neutralization-specific antigens. No antigenic relationship has been found between the viruses in different genera. Some viruses bind erythrocytes (i.e., hemagglutinate).
Derivation of names
Reovirales: from respiratory enteric orphan, due to the early recognition that the viruses caused respiratory and enteric infections and the (incorrect) conclusion that they were not associated with disease and so were considered orphan viruses.
Sedoreoviridae: from sedo, Latin for “smooth”, denoting a smooth appearance and the absence of spikes or turrets on the surface of the core particles.
Spinareoviridae: from spina, Latin for “spike”, denoting the presence of spikes or turrets on the surface of the core particles. The term 'spiked' is an alternative to 'turreted', that was used in early research to describe the structure of the particle, particularly with the cypoviruses (Hill et al., 1999).
Family demarcation criteria
Viruses in genera belonging to the family Spinareoviridae contain spikes or turrets at the 12 icosahedral vertices (e.g., orthoreoviruses) of the core, whereas viruses in the family Sedoreoviridae lack large surface projections (e.g., rotaviruses) on their core, giving them an almost spherical or “smooth” appearance (Hill et al., 1999). However, the outer capsid of some sedoreovirids may possess surface projections (e.g., rotavirus, Figure 1 Reovirales).
Relationships within the order
Comparison of RdRP sequences indicates that members of different genera have amino acid sequence identities of less than 30%. There are two exceptions: (i) members of the species Rotavirus betagastroenteritidis are only 22% identitcal (amino acid level) to other rotaviruses and (ii) aquareoviruses and orthoreoviruses have amino acid sequence identities of up to 42%. In most cases, phylogenetic analysis of RdRP sequences (Figure 3 Reovirales) identifies the turreted spinareovirids and non-turreted sedoreovirids as members of separate clades. However, it is noteworthy that the functional RdRP core contains similar motifs across the entire order (Table 2 Reovirales). Of note, sequence comparisons suggest that there has been an evolutionary jump, involving a change in the number of genome segments, between the aquareoviruses (11 segments) and coltiviruses (12 segments). Seg7 of members of the species Aquareovirus ctenopharyngodontis and Aquareovirus graminis encodes two proteins, from different ORFs, which are homologues of two coltivirus proteins encoded separately on Seg9 and Seg12.
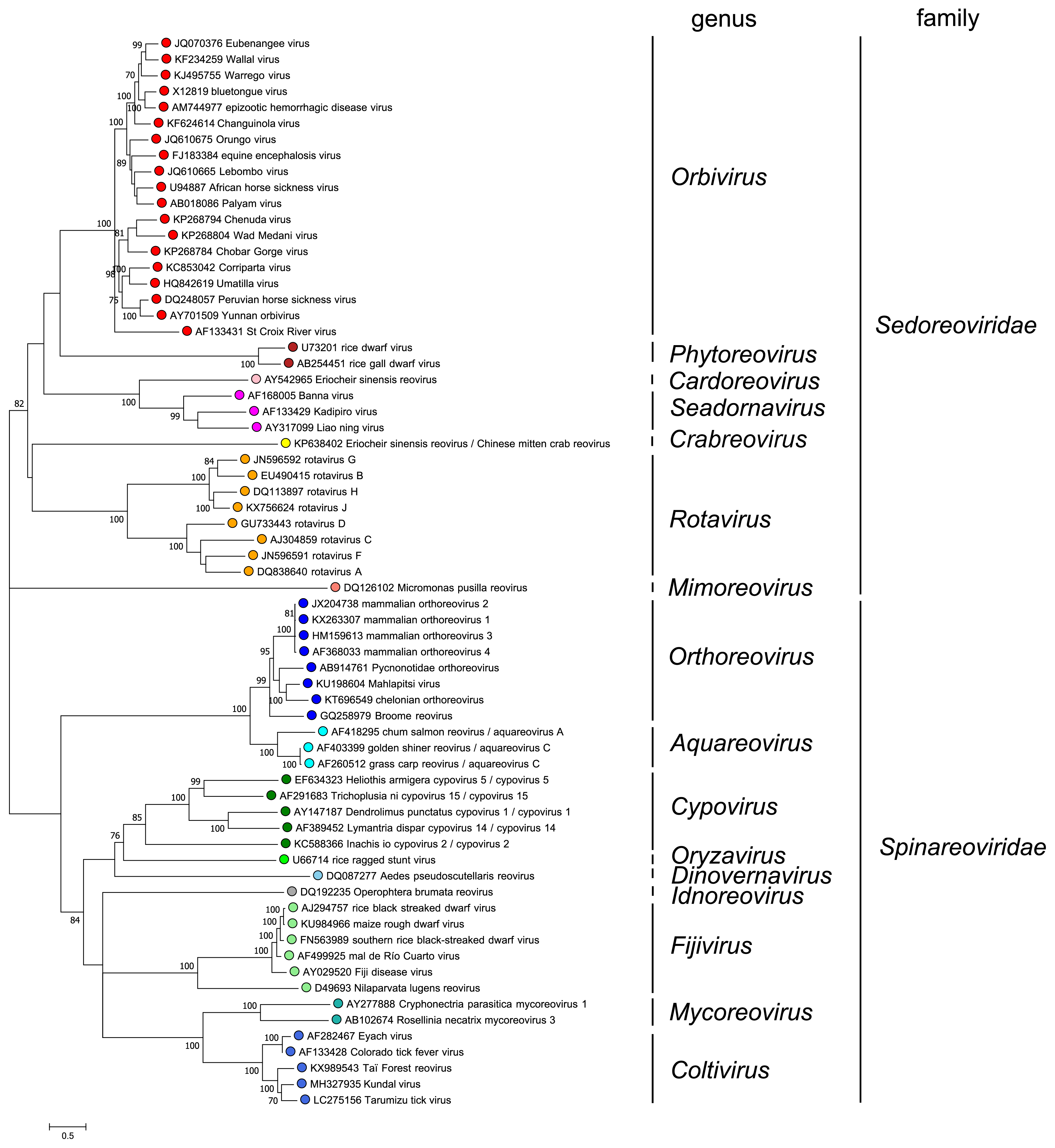 |
| Figure 3.Reovirales. Maximum likelihood tree constructed using the amino acid sequence of the putative RdRP of representative viruses in the order Reovirales. The analysis used the Whelan and Goldman + Freq. model with a discrete gamma distribution of rates between sites, including some invariant sites amd was produced MEGA7 (Kumar et al., 2016). Values at the nodes represent bootstrap confidence levels (500 replications). This phylogenetic tree and corresponding sequence alignment are available to download from the Resources page. Diadromus pulchellus idnoreovirus 1 was not used as an isolate of the genus Idnoreovirus because its genome displays evidence of recombination. |
Table 2.Reovirales Comparison of sequences surrounding conserved RdRP motifs of reoviruses*
| Motifs** | I | IV, 1, A | V, 2, B | VI, 3, C | D |
|---|---|---|---|---|---|
| Consensus | grrtRiI | D.s.wd. | SGe.aTs.a..nla | qvqGDDtlm.ikdg | he.n.sK.s |
bluetongue virus (BTV) (515) | PIKATRTI (72) | DYSEYDTH (119) | SGENSTLIANSMHNMA (21) | EQYVGDDTLFYTKLD (22) | HEASPSKTM (804) |
| rotavirus A (455) | PGRRTRII (57) | DVSQWDSS (63) | SGEKQTKAANSIANLA (19) | IRVDGDDNYAVLQFN (20) | RMNAKVKAL (669) |
rice dwarf virus (RDV) (643) | AWRPVRPI (73) | DCTSWDQT (76) | SGRLDTFFMNSVQNLI (20) | FQVAGDDAIM.VYDG (24) | HIINPQKTV (890) |
| mammalian orthoreovirus S3 (521) | VQRRPRSI (56) | DISACDAS (89) | SGSTATSTEHTANNST (31) | YVCQGDDGLM.IIDG (21) | GEEFGWKYD (772) |
| Nilaparvata lugens reovirus (NLRV) (646) | IDRRGRII (60) | DMSGMDAH (90) | SGLFATSGQHT.MFLV (20) | NYVMGDDIFQNIKNG (24) | IDGNYSKYS (894) |
| rice ragged stunt virus (RRSV) (500) | IGRRQRAI (62) | DASVQASV (83) | SGQPFTTVHHTFTLSN (1) | LTVQGDDTRT.INYG (15) | VSDWGFKVS (735) |
| Banna virus (BAV) (557) | LVRGTRAK (74) | DTSQWGQI (62) | SGELTTQTRNTTTNIS (25) | DNKVGDDSVEVLRVV (24) | HLEISAKRTI (415) |
*The regions covering the putative polymerase module in RRSV P4 (amino acid positions 500 to 735) and other reoviruses were analyzed using the GCG program PILEUP and further aligned manually, taking into account the polymerase motifs presented and aligned by (Poch et al., 1989) (A–D), (Bruenn 1991) (1–3) and (Koonin 1992) (I, IV–VI).
** The numbers in brackets in the first column indicate the amino acid position of the start of the motif, in subsequent columns the number of amino acids between the motif in that column and that in the following column, and in the last column the amino acid position of the end of the motif.
Overall phylogenetic analyses support the ‘co-speciation hypothesis’ that members of the order Reovirales have co-evolved with their respective hosts or arthropod vectors. Although members of the order Reovirales form a single monophyletic group, they have been evolving for at least 550 million years (Attoui et al., 2002), resulting in extensive sequence divergence and clear structural differences between the more distantly related genera. However, the replication mechanisms used, and certain structural parameters of the internal proteins (e.g., the subcore shell and polymerase complexes) remain fundamentally and remarkably similar. Evolutionary analyses suggest that the non-turreted viruses may represent an ancestral lineage from which the turreted viruses have evolved (Attoui et al., 2006).
Closer relations between certain genera have been identified by structural analyses of the viral proteins and may be heralded by signature sequences. For example, comparisons of the RdRP, capping enzyme, and capsid protein sequences, as well as structural analyses of outer-capsid proteins, suggest that an evolutionary jump has occurred between the seadornaviruses and rotaviruses within the family Sedoreoviridae. This process is thought to have involved gene duplication and rearrangement, changing the number of genome segments (from 11 for the rotaviruses to 12 for the seadornaviruses).
Closer relationships have also been identified between the aquareoviruses and orthoreoviruses within the family Spinareoviridae, with homologous proteins having up to 42% amino acid identity and a particle morphology, as revealed by cryoEM, that is super-imposable. Gene duplication followed by rearrangement also appears to have occurred between the aquareoviruses and coltiviruses, increasing the number of segments from 11 (aquareoviruses) to 12 (coltiviruses). Co-evolution of orbiviruses and their vectors has been suggested, wherby an ancestral orbivirus gave rise to both mosquito-borne and Culicoides-borne orbiviruses. During this process, duplication of the full-length sequence of the receptor-binding outer-capsid protein of the ancestral orbivirus generated outer-capsid proteins that are twice as long in mosquito-borne and culicoides-borne orbiviruses.
Relationships with other taxa
Although there is little evidence for nucleotide or amino acid sequence similarities with other dsRNA viruses, it may be significant that some viruses (e.g., those in the families Cystoviridae and Totiviridae) also produce particles in which the inner shell is characteristically composed of 120 copies of a triangular protein, which are arranged in a manner like that of members of the order Reovirales (Mata et al., 2020). This protein provides an elegant mechanism of assembling the inner icosahedral capsid shell, which has been described alternatively as having T=1 or T=2 symmetry, although it is important to note these are different academic interpretations of a similar particle architecture. These similarities also may indicate common, if distant, ancestry and again suggest that these viruses may even have diversified and evolved along with their host species. A recent study placed the RdRP of viruses from different dsRNA virus families into at least two distinct evolutionary braches, these being related to positive-sense (+) RNA, and negative-sense (-) RNA viruses, respectively (Wolf et al., 2018).
Related, unclassified viruses
| Virus name | Accession number | Abbreviation |
| Cimodo virus | Seg1: KF880748; Seg2: KF880749; Seg3: KF880750; Seg4: KF880751; Seg5: KF880752; Seg6: KF880753; Seg7: KF880754; Seg8: KF880755; Seg9: KF880756; Seg10: KF880757; Seg11: KF880758; Seg12: KF880759 |
Virus names and virus abbreviations are not official ICTV designations.

