Family: Flaviviridae
Authors:
Rebecca Rico-Hesse, Tatjana Avsic-Zupanc, Bradley Blitvich, Jens Bukh, Van-Mai Cao-Lormeau, Allison Imrie, Amit Kapoor, Laura D Kramer, Brett D Lindenbach, Peter Simmonds, Donald B. Smith and Pedro Fernando da Costa Vasconcelos
The citation for this ICTV Report chapter is the summary published as Simmonds et al., (2017):
ICTV Virus Taxonomy Profile: Flaviviridae, Journal of General Virology, 98:2–3.
Corresponding author: Rebecca Rico-Hesse ([email protected])
Edited by: Nick J. Knowles and Stuart G. Siddell
Posted: January 2017, updated February 2019, May 2023
PDF: ICTV_Flaviviridae.pdf (2019 version)
Summary
Flaviviridae is a family of small enveloped viruses with positive-sense RNA genomes of approximately 9.0–13 kb (Table 1.Flaviviridae). Most infect mammals and birds, and many are host-specific and pathogenic, such as hepatitis C virus (HCV) in the genus Hepacivirus. Most members of the genus Orthoflavivirus are arthropod-borne, and many are important human and veterinary pathogens (e.g., yellow fever virus, dengue virus, West Nile virus).
Table 1.Flaviviridae. Characteristics of members of the family Flaviviridae.
| Characteristic | Description |
|---|---|
| Typical member | yellow fever virus-17D (X03700), species Orthoflavivirus flavi, genus Orthoflavivirus |
| Virion | Enveloped, 40–60 nm virions with a single core protein (except for genus Pegivirus) and 2 or 3 envelope glycoproteins |
| Genome | 9.0–13 kb of positive-sense, non-segmented RNA |
| Replication | Cytoplasmic, in membrane vesicles derived from the endoplasmic reticulum (ER); assembled virions bud into the lumen of the ER and are secreted through the vesicle transport pathway |
| Translation | Directly from genomic RNA containing a type I cap (genus Orthoflavivirus) or an internal ribosome entry site (other genera) |
| Host range | Mammals (all genera); most members of genus Orthoflavivirus are arthropod-borne |
| Taxonomy | Realm Riboviria, kingdom Orthornavirae, phylum Kitrinoviricota, class Flasuviricetes, order Amarillovirales; four genera and 97 species |
Orthoflavivirus. Most members of this genus, which includes 53 species, are arthropod-borne viruses, with distinct groups infecting mosquitoes or ticks. Mammals and birds are the usual primary hosts, in which infections range from asymptomatic to severe or fatal haemorrhagic fever or neurological disease. Important human pathogens include yellow fever virus, dengue virus, Zika virus, Japanese encephalitis virus, West Nile virus and tick-borne encephalitis virus. Other members cause economically important diseases in domestic or wild animals. Additional viruses infect only arthropods or only mammals (e.g., Tamana bat virus).
Pestivirus. Pestiviruses infect pigs and ruminants, including cattle, sheep, goats and wild ruminants, and are transmitted through contact with infected secretions (respiratory droplets, urine or faeces). Infections may be subclinical or cause enteric, haemorrhagic or wasting diseases, including the economically important bovine viral diarrhoea virus and classical swine fever virus. Additional pestiviruses of unknown pathogenicity infect bats and rats.
Hepacivirus. This genus includes hepatitis C virus (HCV), a major human pathogen causing chronic liver disease, including cirrhosis and cancer. Other viruses in the genus are of unknown pathogenicity and infect horses, rodents, bats, cows and primates. Infections are typically persistent and target the liver.
Pegivirus. Members of the genus Pegivirus are associated with persistent infections of a wide range of mammalian species. They have not been clearly associated with disease.
A Table with current (MSL38) and previous (MSL37) species names is available at Species names: Flaviviridae.
Virion
Morphology
Virions are 40–60 nm in diameter, spherical in shape with a lipid envelope. The capsid is comprised of a single protein and the envelope contains two or three virus-encoded membrane proteins. Specific descriptions of members of the four individual genera are given in the corresponding genus pages.
Physicochemical and physical properties
The virion Mr, buoyant density, sedimentation coefficient and other physicochemical properties differ among the members of the genera and are described separately in the corresponding genus pages.
Nucleic acid
Genomes are positive-sense ssRNA of approximately 9.2–11.0, 11.3–13.0, 8.9–10.5 and 8.9–11.3 kb for members of the genera Orthoflavivirus, Pestivirus, Hepacivirus and Pegivirus, respectively. All members of the family lack a 3′-terminal poly(A) tract. Only the genomes of members of the genus Orthoflavivirus contain a 5′-terminal type I cap structure, the others possess an internal ribosomal entry site (IRES).
Proteins
Virions of members of the family have a single, small basic capsid (C) and two (Orthoflavivirus, Hepacivirus and Pegivirus) or three (Pestivirus) membrane-associated envelope proteins. Pegiviruses appear to lack a complete nucleocapsid protein gene. The nonstructural proteins contain sequence motifs characteristic of a serine protease, RNA helicase and RNA-dependent RNA polymerase (RdRP) that are encoded at similar locations along the genome in all genera. Further details of specific functional properties are given in the corresponding sections of the individual genera pages.
Lipids
Lipids present in virions are derived from host cell membranes and make up 17% of the total virion weight in the case of members of the genus Orthoflavivirus. The lipid content of pestiviruses, hepaciviruses and pegiviruses is unknown.
Carbohydrates
Virions contain carbohydrates in the form of glycolipids and glycoproteins.
Genome organization and replication
The genomic RNA of all members of the family has a similar organization and is the viral mRNA found in infected cells. It contains a single long open reading frame (ORF) flanked by 5′- and 3′-terminal non-coding regions (NCRs) that form specific secondary structures required for genome replication and translation. Members of the genus Orthoflavivirus, but not pestiviruses, hepaciviruses or pegiviruses produce a unique, subgenomic, small (300–500 nt) non-coding RNA that is derived from the 3′-NCR of genomic RNA (Lin et al., 2004) that is essential for virus replication in cells and modulates pathogenicity in animals. Translation-initiation of genomic RNA is cap-dependent for members of the genus Orthoflavivirus, whereas IRES elements are present in viruses of the other genera. Viral proteins are synthesized as part of a polyprotein that is co- and post-translationally cleaved by viral and cellular proteases. The structural proteins are contained in the N-proximal portion of this polyprotein and the nonstructural proteins in the remainder. The latter include a serine protease, an RNA helicase and the RdRP. Genome replication occurs in the cytoplasm in association with modified cellular membranes via the synthesis of genome-length negative-strand intermediates. Virion assembly, including acquisition of a glycoprotein-containing lipid envelope, occurs by budding through intracellular membranes. Viral particles are transported in cytoplasmic vesicles through the secretory pathway before they are released by exocytosis, as shown for members of the genus Orthoflavivirus and assumed for members of the other genera. In addition, release of infectious RNA via exosomes has recently been demonstrated (Ramakrishnaiah et al., 2013).
Biology
The biological properties of viruses in the four genera exhibit different characteristics and are described in the corresponding sections of the genus pages.
Antigenicity
The viruses of different genera are antigenically unrelated, but serological cross-reactivity exists among members within each genus.
Derivation of names
Flaviridae, Orthoflavivirus: from Latin flavus, “yellow”.
Pestivirus: from Latin pestis, “plague”.
Hepacivirus: from Greek hepar, hepatos, “liver” and identifying letter from hepatitis C virus
Pegivirus: from persistent, and the original names of the GB viruses and hepatitis G, deriving from the initials of the original source, the surgeon “GB”
Relationships within the family
Phylogenetic relationships of amino acid sequences in a conserved domain of the RdRP show clustering of members of the Flaviviridae into the four currently assigned genera, although there is a closer phylogenetic relationship between members of the Hepacivirus and Pegivirus genera than between others (Figure 1.Flaviviridae). Another exception is the outlier position of Tamana bat virus, currently listed as a potential member of the Orthoflavivirus genus, but sufficiently distinct to potentially merit assignment into a new genus, should further related viruses be found in the future.
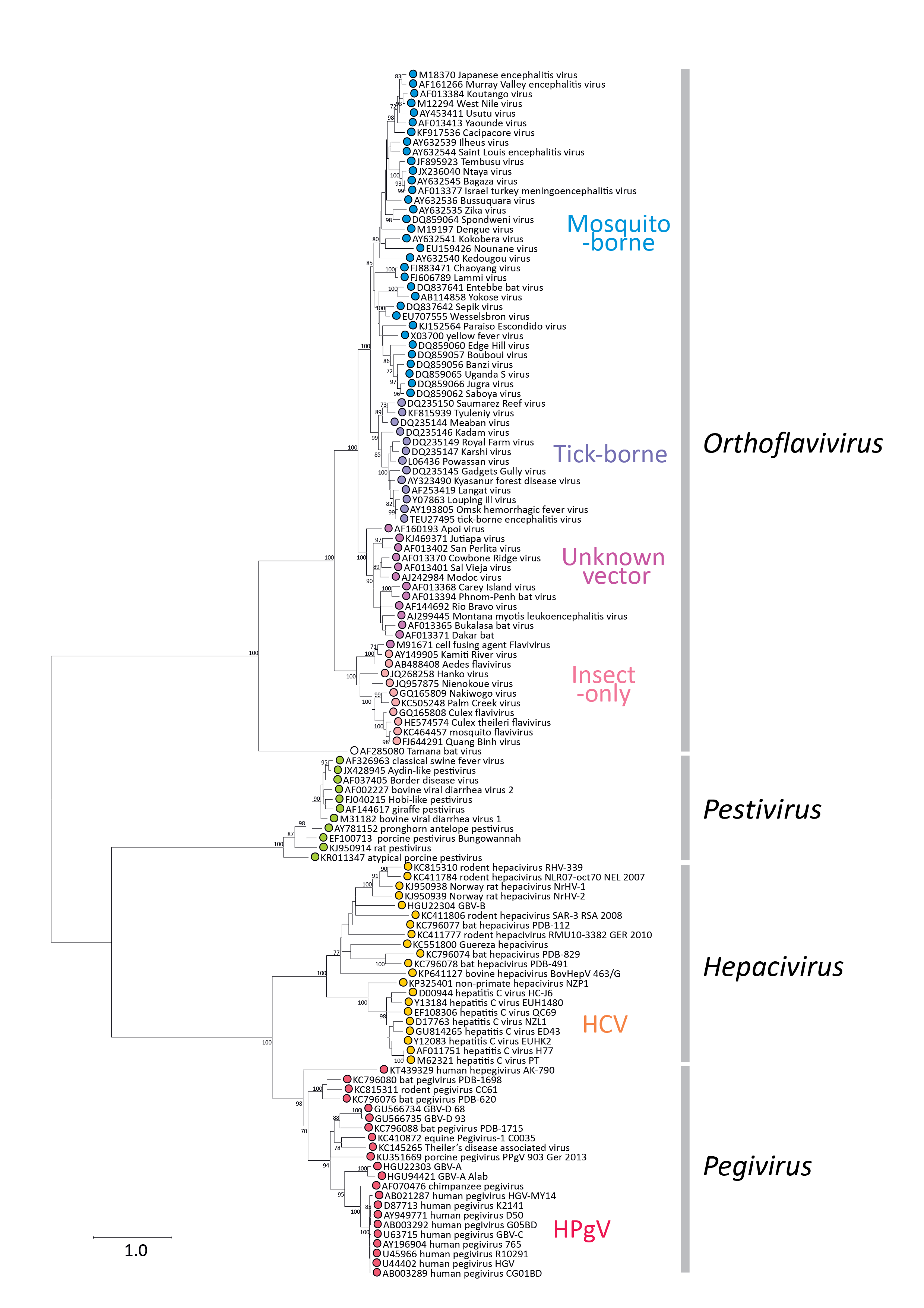 |
| Figure 1.Flaviviridae. Phylogeny of conserved amino acid sequences in the RdRP (NS5 or NS5B) of members of the family Flaviviridae. Partial gene sequences between positions 8,040–8,897 (numbered using positions in the HCV sequence, AF011751) from representative isolates of each species and from several related unclassified viruses were aligned as inferred amino acid sequences using MUSCLE (Edgar 2004) and verified by the presence of aligned motifs. An unrooted phylogenetic tree was constructed from the sequence alignment by maximum likelihood using an empirically determined optimal substitution model – Le Gascuel 2008 with a gamma distribution (5 categories) and invariant sites (LG + G+I) computed with the MEGA version 6.1 package (Tamura et al., 2013). Data was bootstrap re-sampled 100 times; values of >=70% are shown next to the branches. This phylogenetic tree and corresponding sequence alignment are available to download from the Resources page. |
Relationships with other taxa
Members of the Flaviviridae have been placed into RNA virus supergroup II, a group that also includes members of the Tombusviridae (plant), members of the Luteovirus genus in the Luteoviridae (plant), Leviviridae (bacterial virus) and a series of recently described insect-derived flavi-like viruses, many with segmented genomes (Shi et al., 2015). However, the virion structure and other viral structural and nonstructural genes in these other virus groups are distinct and likely non-homologous.

