Family: Birnaviridae
Bernard Delmas, Houssam Attoui, Souvik Ghosh, Yashpal S. Malik, Egbert Mundt and Vikram N. Vakharia
The citation for this ICTV Report chapter is the summary published as Delmas et al., (2018):
ICTV Virus Taxonomy Profile: Birnaviridae, Journal of General Virology, 100, 5–6.
Corresponding author: Bernard Delmas ([email protected])
Edited by: Jens H. Kuhn and Stuart G. Siddell
Posted: October 2018, updated May 2023
PDF: ICTV_Birnaviridae.pdf
Summary
Birnaviridae is a family of viruses with bisegmented dsRNA genomes with a total of about 6 kbp forming icosahedral, non-enveloped virions (Table 1. Birnaviridae). The family includes seven genera, members of three of which (Aquabirnavirus, Avibirnavirus and Blosnavirus) infect vertebrates (excluding mammals), members of two genera (Entomobirnavirus and Dronavirus) infect insects, with other hosts being rotifers (Ronavirus) and molluscs (Telnavirus). Each genus includes 1–3 species. Infectious pancreatic necrosis virus of salmonids and infectious bursal disease virus are two economically important birnaviruses.
Table 1. Birnaviridae. Characteristics of members of the family Birnaviridae.
|
Characteristic |
Description |
|
Example |
infectious bursal disease virus P2 (A: X84034; B:X84035), species Avibirnavirus gumboroense, genus Avibirnavirus |
|
Virion |
Non-enveloped, icosahedral virion with internal ribonucleoprotein complexes |
|
Genome |
Two double-stranded RNA segments (2.9 to 3.6 kbp) with the RNA-dependent RNA-polymerase covalently linked to the 5′-end of the genomic positive-strand |
|
Replication |
Cytoplasmic |
|
Translation |
Capped mRNAs, which lack poly(A) tracts, are translated by the cellular translation machinery |
|
Host range |
Vertebrates (excluding mammals); invertebrates |
|
Taxonomy |
Realm Riboviria, kingdom Orthornavirae; seven genera: Aquabirnavirus, Avibirnavirus, Blosnavirus, Dronavirus, Entomobirnavirus, Ronavirus and Telnavirus, each including one to three species |
Virion
Morphology
Virus particles are non-enveloped, single-shelled particles with a diameter of about 65 nm (Figure 1. Birnaviridae). The capsid follows a T=13 laevo icosahedral geometry and is made up of a single capsid protein, VP2, clustered in trimers and forming 260 projections of about 4 nm at the surface of the particle (Coulibaly et al., 2005). Two ribonucleoprotein complexes made by the genome segments associated to multiple copies of a ribonucleoprotein (VP3) and several molecules of the RNA-dependent RNA polymerase (RdRP, VP1) are packaged inside each particle (Hjalmarsson et al., 1999).
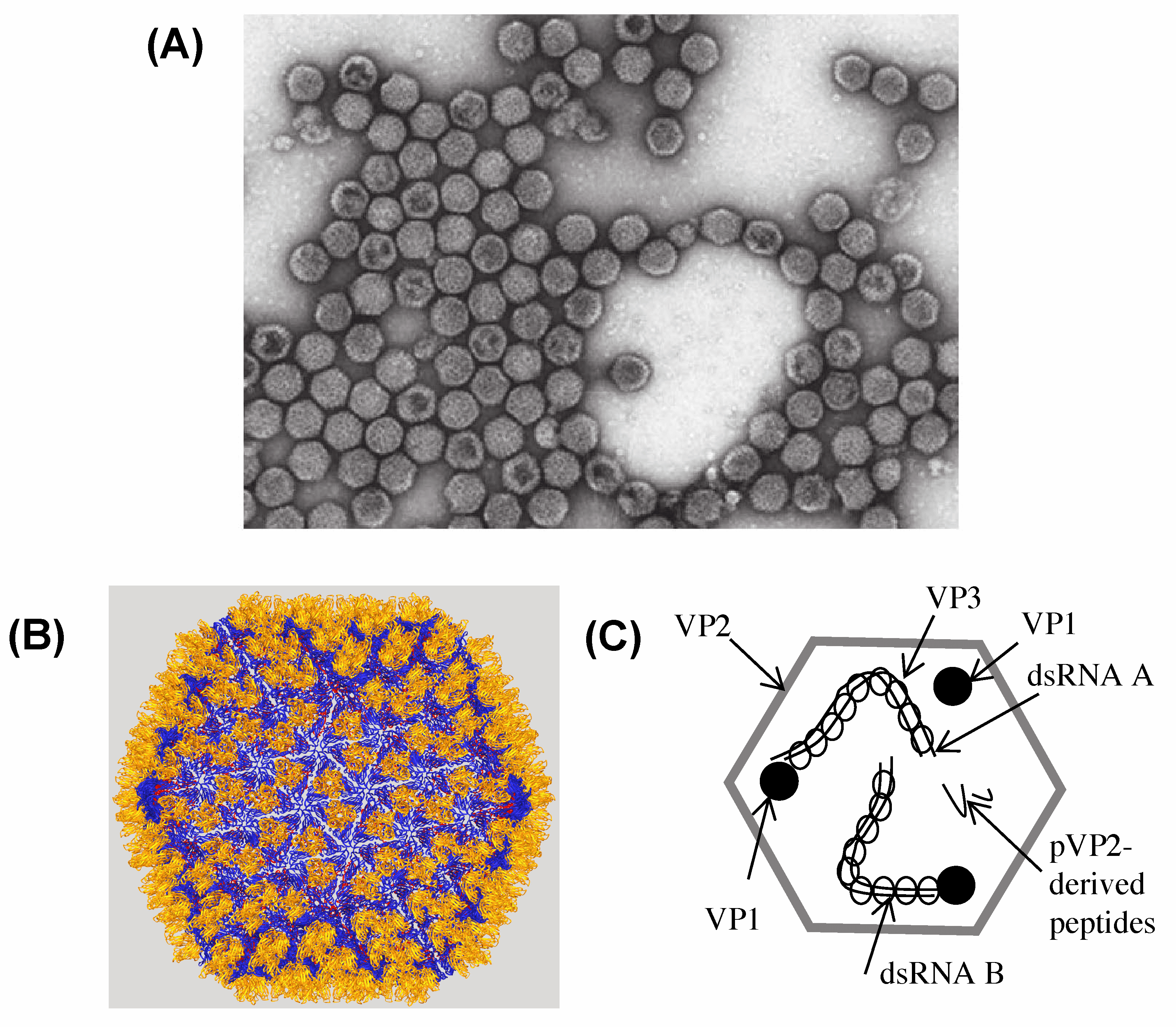 |
| Figure 1. . Birnaviridae. Birnavirus particle structure (A) Negative-contrast electron micrograph of infectious bursal disease virus particles (courtesy of J. Lepault). Bar represents 100 nm. (B) A three-dimensional model of the infectious bursal disease virus virion derived from X-ray crystallography (Courtesy of F. Rey) with its T=13 laevo icosahedral shell. (C) Diagrammatic representation of an infectious bursal disease virus particle showing the distribution of the polypeptides and the genome in the virus particle. |
Physicochemical and physical properties
Virion buoyant density in CsCl is 1.33 g cm-3. Viruses are stable at pH 3–9, resistant to heat (60°C, 1 h), ether and 1% SDS at 20°C, pH 7.5 for 30 min.
Nucleic acid
The genome of birnaviruses comprises two linear double-stranded RNA genomic segments (A and B) that together constitute 8–10% of virus particles by weight. Virions can package more than one complete copy of each segment (Luque et al., 2009). The larger segment (A) is 3.1–3.6 kbp, and the smaller segment (B) is 2.8 to 3.3 kbp, depending on the virus. The genome RNA sequences have a nucleotide composition of 53–58% G+C, with the exception of those of rotifer birnavirus (RBV, species Ronavirus rotiferae) and Drosophila X virus (DXV, species Entomobirnavirus drosophilae), which are 44–47% G+C. The dsRNA segments are completely base-paired, and the positive-sense strand of each segment is covalently linked to a viral protein at its 5′-terminus, but has neither a polyadenylation signal nor a terminal poly-A tail at its 3′-end (Calvert et al., 1991). Segment B encodes VP1 and segment A encodes the polyprotein preVP2-VP4-VP3. An alternative small open reading frame is often present in segment A, and in some cases overlaps the initiation codon of the polyprotein ORF.
Proteins
The polyprotein encoded by the large ORF of segment A is first processed during translation to generate preVP2, VP4 (the viral protease), and VP3 (Birghan et al., 2000). Further processing of preVP2 occurs at its C-terminal domain to generate the mature capsid protein (VP2) and three to four peptides (ranging from 7 to about 50 aa) that remain in the particles (Da Costa et al., 2002). In the case of infectious bursal disease virus (IBDV), these peptides comprise 46, 7, 7, and 11 aa. The capsid is formed by trimers of VP2 (417–442 aa). VP2 possesses a unique structural fold that is composed of two beta-barrels with a jelly-roll topology, with the beta-strands oriented tangentially and radially to the virus particle (Coulibaly et al., 2005). Inside the capsid, VP3 (238–309 aa) and the genomic RNA form thread-like ribonucleoprotein complexes that do not follow the icosahedral symmetry of the capsid particle (Hjalmarsson et al., 1999). Different ratios of VP3 over VP2 have been reported in virions from members of different species.
The RdRP VP1 (844–1045 aa) is found free in the viral particle and also covalently associated to the 5′-end of the positive-sense strand of the genomic dsRNA through its N-terminal serine (as shown for infectious pancreatic necrosis virus (IPNV); (Graham et al., 2011). The 2.5 Å resolution structure of IBDV and IPNV VP1 further reveals a characteristic rearrangement of motifs, from A–B–C to C–A–B, in the RNA polymerase catalytic palm domain, which is not found in viral RdRPs from other dsRNA viruses (Graham et al., 2011, Gorbalenya et al., 2002, Pan et al., 2007). VP1 can guanylylate itself to produce VP1-pG and VP1-pGpG independently from its RNA polymerase activity. VP1 possesses viral mRNA 5′-guanylyl transferase and capping activities (Dobos 1993).
The viral protease VP4 (also called NS in IPNV, 212–244 aa) cleaves its own N- and C-termini in the polyprotein and further processes preVP2 to generate VP2 and peptides arising from the C-terminal domain of preVP2 (Birghan et al., 2000). Its catalytic site comprises a serine-lysine dyad. The VP4 catalytic domain is structurally similar to the protease domain of bacterial ATP-dependent Lon proteases.
A nonstructural, positively-charged polypeptide encoded by the small ORF of segment A has been designated VP5 (17 kDa in IBDV and 15 kDa in IPNV). This protein is nonessential for replication of IBDV and IPNV. A second ORF, encoding an arginine-rich protein, has also been identified in genome segment A of DXV, blotched snakehead virus (BSNV), and Tellina virus 1 (TV-1, species Telnavirus tellinae) (Nobiron et al., 2008).
Lipids
None reported.
Carbohydrates
No carbohydrate has been observed in the structures of any of the birnavirus proteins that have been determined by X-ray crystallography.
Genome organization and replication
Segment A generally contains two ORFs: ORF2 encoding a large polyprotein of about 105–120 kDa, and an overlapping (IPNV, IBDV, and Drosophila B birnavirus (DBV - species Dronavirus drosophilae, (Wu et al., 2010)) or internal (DXV, BSNV, and TV-1) ORF1, which encodes a protein of 15–27 kDa (Figure 2. Birnaviridae). Positions of the polyprotein cleavage sites to generate preVP2, VP4, and VP3 during translation have been determined experimentally for IBDV, IPNV, BSNV, DXV, and TV-1 (Figure 2. Birnaviridae). For BSNV and TV-1, an additional polypeptide, named X, is encoded between the preVP2 and the VP4 domains. The proteolytic processing of preVP2 to generate VP2 and the structural peptides, which occurs during particle assembly, has been characterized for IBDV, IPNV, BSNV, and TV-1.
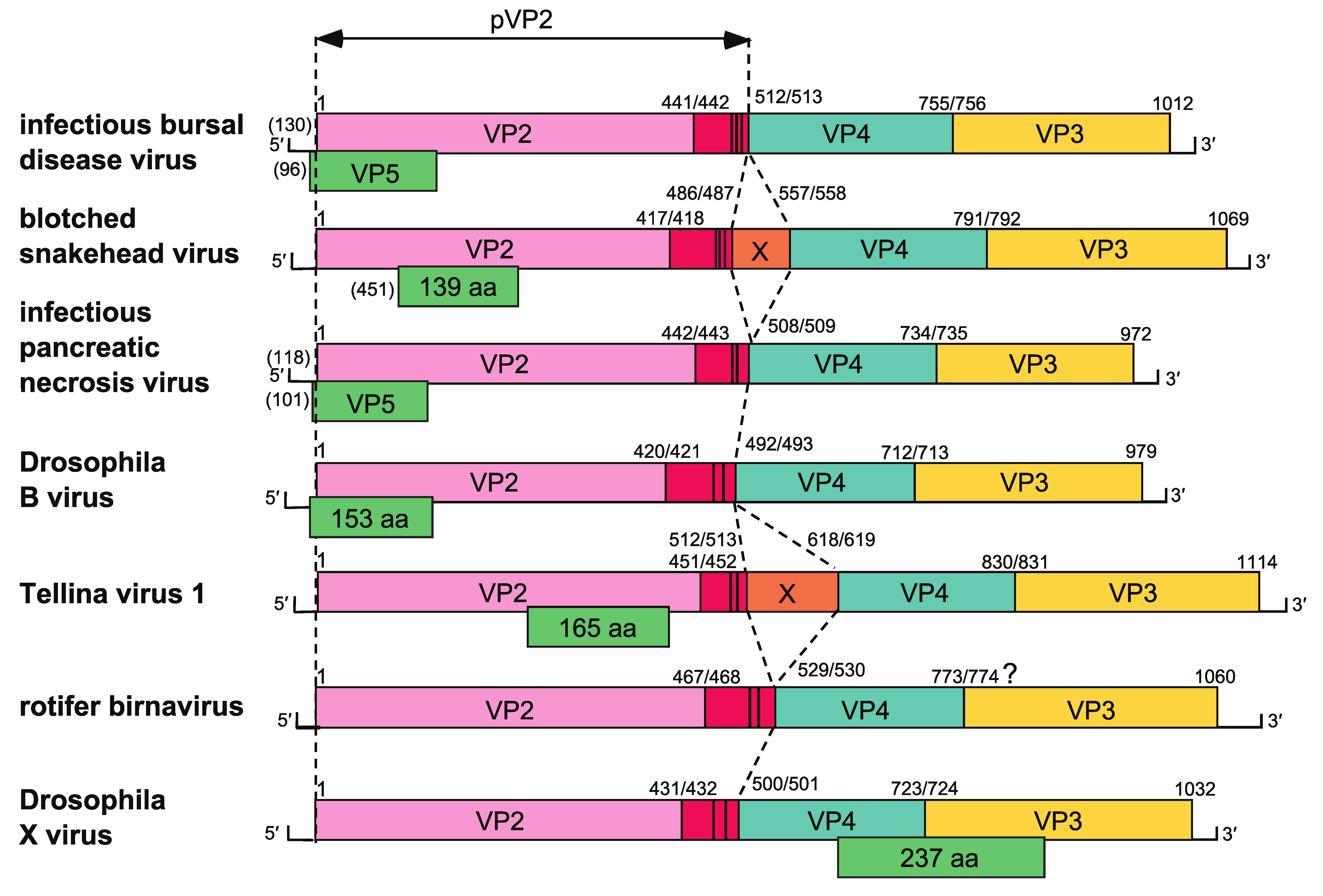 |
| Figure 2. . Birnaviridae. Schematic representation of the gene arrangement in the coding strand of genome segment A of representative birnaviruses. Polyprotein cleavage sites are indicated by a vertical bar and identified by amino acid position. Related ORFs are color-coded with red indicating the peptides derived from processing of preVP2; green indicates the small additional ORFs. Parentheses at the 5′-ends indicate the length of the 5′-non-coding region where known. |
A single cycle of replication takes about 18–22 h for IPNV and 4–8 h for IBDV. The mode of entry of viruses into cells is not well understood. For IBDV binding at the cell surface, proteins such as heat shock protein 90 and α4β2 integrin have been proposed to serve as functional receptors in various types of chicken cells. One of the small structural IBDV peptides, pep46 (a 46 aa amphiphilic peptide), and its homologs in other birnaviruses, are able to induce pores in target membranes, suggesting a role in virus entry. Endosomal acidification is not a prerequisite for virus internalization in IPNV-infected cells. After delivery into the cytoplasm, the viral RdRP becomes activated and produces two genome-length (24S) mRNA molecules from each of the 14S dsRNA genome segments. These mRNAs are capped, but they lack 3′-polyA sequences. Replicative intermediates have been identified in infected cells. Virus RNA is transcribed by a semi-conservative strand displacement mechanism in vitro. There is no information on negative-strand RNA synthesis. The two mRNAs can be detected in infected cells by 3–4 h post-infection (p.i.) and are synthesized in the same relative proportions throughout the replicative cycle (i.e., about twice as many A as B mRNA molecules). Virus-specific polypeptides can be detected at 4–5 h p.i. and are present in the same relative proportions until the end of the replication cycle. There are no specific early or late proteins.
Segment A mRNA is translated to yield a 105 kDa polyprotein precursor that comprises the preVP2, VP4 (NS), and VP3 polypeptides (Figure 2. Birnaviridae). The VP4 protease co-translationally cleaves the polyprotein to generate the three (or four in BSNV and TV-1) polypeptides (Figure 3. Birnaviridae). PreVP2 is later processed during virus assembly by a slow maturation cleavage to produce the mature VP2 and small peptides that remain associated to the virus particles. This cleavage can be incomplete since traces of preVP2 are found in purified virus particles, although the mature VP2 predominates. Virus assembly and maturation of the capsid protein preVP2/VP2 are concomitant and interdependent. In addition to VP4, the preVP2 processing requires the presence of VP3 and VP1. This requirement acts in favor of the existence of a large quaternary maturation complex formed by preVP2, VP4, VP3, and VP1. During infection, rigid tubes 55 nm in diameter are formed by preVP2. In the case of IBDV, additional tubules 25 nm in diameter, made of VP4, appear in late steps of the virus replication cycle. The translation product of the 17 or 15 kDa ORF has been detected in IBDV- or IPNV-infected cells, respectively.
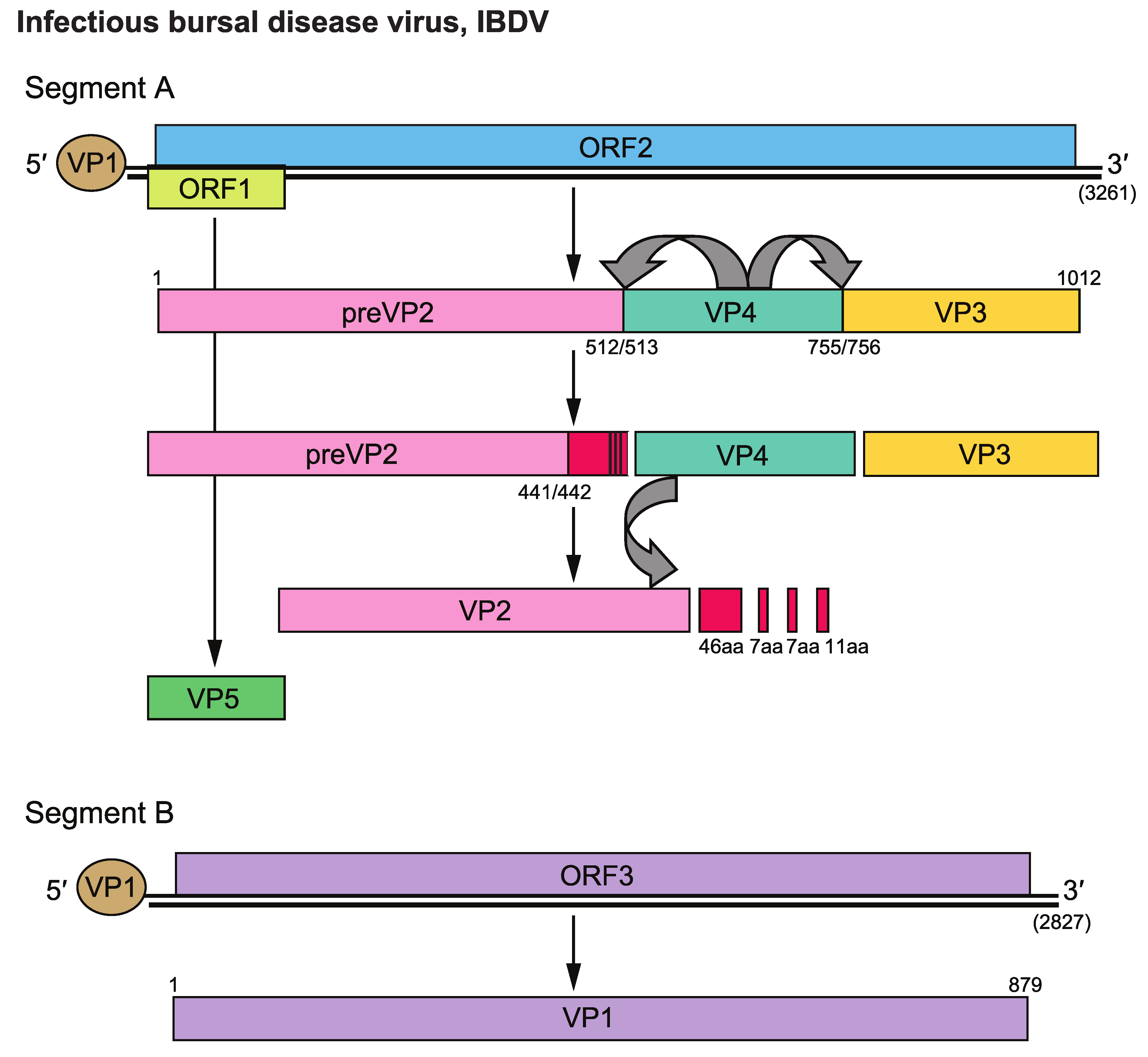 |
| Figure 3. Birnaviridae. Schematic representation of the genome of infectious bursal disease virus illustrating processing of the encoded proteins. Numbers in parentheses indicate the nucleotide lengths of the two genomic segments. |
Segment B mRNA is translated to a 94 kDa polypeptide that represents the viral RdRP (VP1, Figure 3. Birnaviridae). VP1 is found in virions in both a “free” and a genome-linked form. Virus particles assemble and accumulate in the cytoplasm. Encapsidation of the RdRP VP1 is mediated by its interaction with genome-associated protein VP3. The mechanism of virus release is unknown. In tissue culture, about half of the progeny virions remain cell-associated, and, depending on the multiplicity of infection, defective interfering particles are also formed.
Reverse genetics systems have been established for IBDV and IPNV (Mundt and Vakharia 1996, Santi et al., 2005). In vitro transcribed capped viral cRNAs of segment A and B were found to be infectious, facilitating studies of birnavirus replication and virulence.
Biology
While salmonid fish were first recognized as natural hosts of IPNV, viruses antigenically-related to IPNV have been isolated from other freshwater and marine fishes, as well as from bivalve molluscs (Tellina virus 2 (TV-2)). IPNV is transmitted both vertically and horizontally, and there are no known vectors. The geographic distribution is world-wide. IPNV can cause epizootics resulting in high mortality in hatchery-reared salmonid fry and fingerlings. The virus causes necrotic lesions in the pancreas and is also found, without lesions, in other organs such as kidneys, gonads, intestine and brain. It is believed that infected adult fish become lifelong carriers without exhibiting overt signs of infection.
The natural hosts of IBDV are chickens and turkeys. Rarely, IBDV has been isolated from ducks and other domestic fowl. The mode of transmission is horizontal, and there are no known vectors. IBDV has a worldwide distribution. The virus affects the bursa of Fabricius in young chicks, causing B lymphocyte depletion. Genes involved in B cell activation and signalling are down-regulated following infection (Dulwich et al., 2017). Death can occur between 3 and 10 weeks of age, and is then associated with inflammation in the bursa of Fabricius, formation of immune complexes, depletion of complement, clotting abnormalities, and subsequent severe immunosuppression.
Populations of common fruit flies (Drosophila melanogaster) are the natural host of DXV. The mode of transmission is horizontal and there are no known vectors. The geographic distribution is unknown. Infected fruit flies become sensitive to CO2. The target organs and histopathology are not known. DXV has also been isolated from populations of biting midges (Culicoides spp.). BSNV was isolated from a cell line developed from the tropical blotched snakehead fish (Channa lucius), whereas TV-1 was identified in thin tellins (bivalve mollusc of the species Tellina tenuis). Rotifer birnavirus (RBV, species Ronavirus rotiferae) was isolated from a population collapse of a euryhaline rotifer (Brachionus plicatilis), which is cultivated for feeding the fry of marine fish in hatcheries. DBV was identified by sequencing of the small RNAs present in a cultured common fruit fly (Drosophila melanogaster) cell line. Eridge virus was isolated from another common fruit fly (Drosophila immigrans). Espírito Santo virus was recovered in a sample of a virulent strain of dengue virus 2 (Flaviviridae: Flavivirus) and found to be dependent upon co-infection with this virus. Culex Y virus was isolated from Culex pipiens mosquitoes and mosquito X virus was identified from Anopheles sinensis mosquitoes. Culicine-associated Z virus was sequenced from a pool of adult female Ochlerotatus caspius and Oc. detritus mosquitoes from a rice field.
Antigenicity
The capsid protein VP2 is the type-specific antigen and forms the virus-neutralizing epitopes. Anti-VP3 antibodies do not neutralize virus infectivity. There are no serological cross-reactions between the fish, avian, and insect birnaviruses, or between the aquatic birnaviruses IPNV, BSNV, TV-1, and RBV.
Derivation of names
Aquabirnavirus: from Latin aqua, “water”.
Avibirnavirus: from Latin avis, “bird”.
Birnaviridae: from Latin prefix bi, “two”, signifying the bisegmented nature of the viral genome as well as the presence of dsRNA; and rna from ribo nucleic acid, indicating the nature of the viral genome.
Entomobirnavirus: from Greek entomon, “insect”.
Blosnavirus: from blotched snakehead virus.
Genus demarcation criteria
Birnaviridae includes seven genera, members of three of which (Aquabirnavirus, Avibirnavirus, and Blosnavirus) infect vertebrates, members of the two genera (Entomobirnavirus and Dronavirus) infect insects, while other hosts are rotifers (Ronavirus) and molluscs (Telnavirus). Members of the Avibirnavirus genus exclusively infect birds. Members of the Aquabirnavirus and Blosnavirus genera, both defined by viruses infecting fish, reveal less than 60% amino acid identity between their respective RdRP proteins, and also between their respective capsid proteins. More generally, pairwise alignments between members of the four genera are always 30 to 60% identical in their RdRP and capsid protein amino acid sequences.
Relationships within the family
The phylogeny of birnaviruses correlates well with their hosts at the host phylum rank, but less so below that rank (Gibrat et al., 2013). Phylogenetic relationships within the family are shown in Figure 4. Birnaviridae.
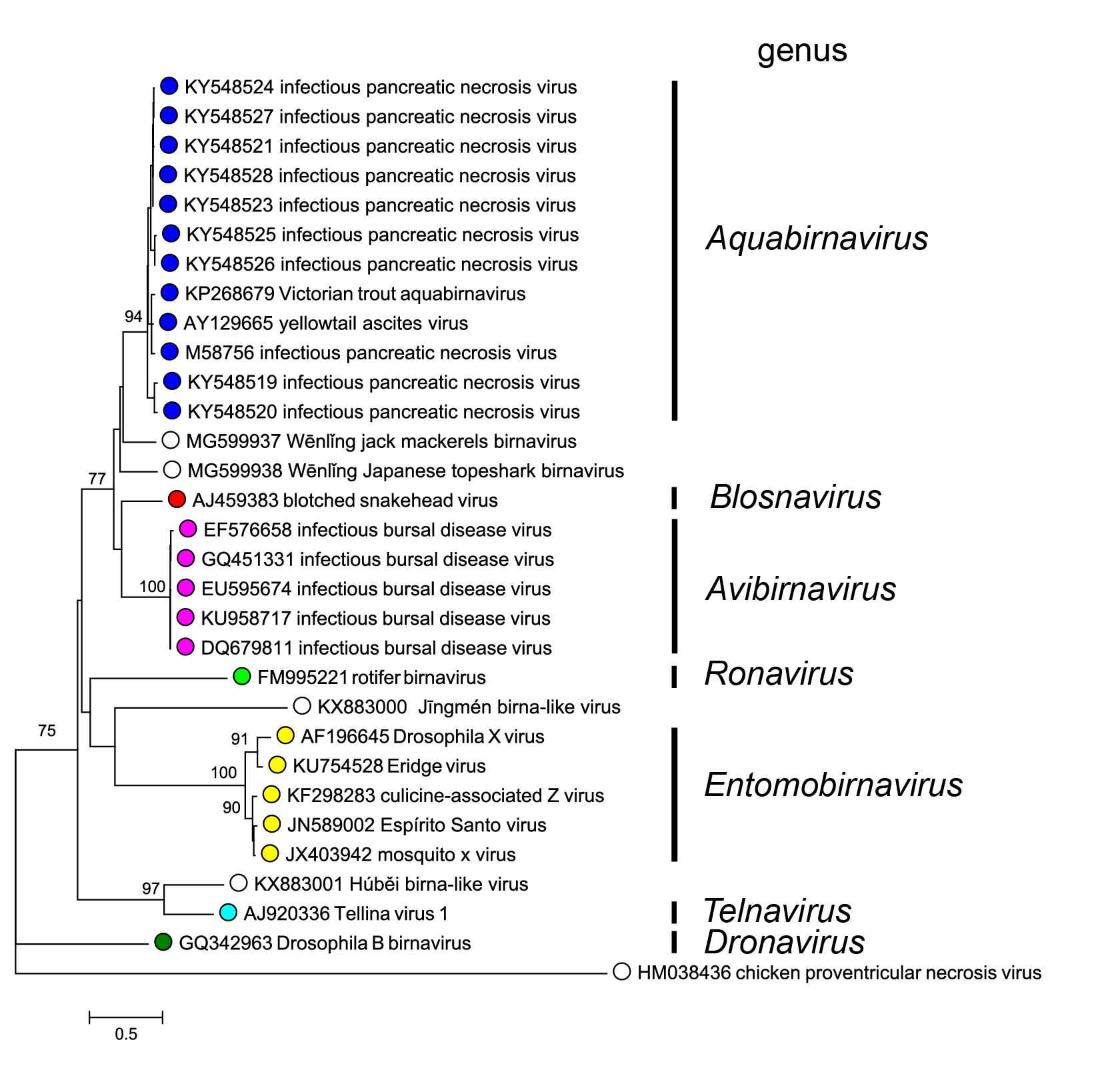 |
| Figure 4. Birnaviridae. A distance tree representing the phylogenetic relationships of VP1 for members of the various genera and genetic clusters in the family (including new birnaviruses described in (Shi et al., 2016, Shi et al., 2018)). Amino acid alignment was done with ClustalW (Larkin et al., 2007) and adjusted visually. Phylogenetic analysis was performed with MEGA7 (Kumar et al., 2016) using the JTT substitution matrix and a gamma distribution of rate variation between sites. This phylogenetic tree and corresponding sequence alignment are available to download from the Resources page. |
Relationships with other taxa
Birnaviruses share no nucleic acid sequence similarity with other taxa, and the birnavirus capsid protein VP2 has no sequence similarities with corresponding capsid proteins of any other virus family. However, the crystal structures of IBDV and IPNV VP2 shows that VP2 is folded into three distinct domains (base, shell, and projection) disposed radially in the virus particle, the base and shell domains displaying high structural similarities with the capsid proteins of viruses belonging to the families Nodaviridae and Tetraviridae, which are positive-sense ssRNA viruses. The VP4 protease has sequence and structural homologies with the Ser-Lys catalytic protease domain of bacterial ATP-dependent Lon proteases. Interestingly, the A-B-C to C-A-B motif rearrangement in the VP1 RdRPs of birnaviruses is also shared by the RdRPs of of Thosea asigna virus and Euprosterna elaeasa virus (genus Alphapermutotetravirus, family Permutotetraviridae), insect viruses that have positive-sense ssRNA genomes, but not by RdRPs of other dsRNA viruses. These unusual RdRPs form a minor and deeply separated cluster in the viral RdRP phylogenetic tree. The structural relationships among the capsid proteins and the RdRPs of birnaviruses and viruses belonging to the Tetraviridae and Permutotetraviridae families suggest evolutionary links between positive-sense ssRNA and dsRNA viruses.
Related, unclassified viruses
|
Virus name |
Accession number |
Virus abbreviation |
|
Wēnlǐng jack macherels birnavirus |
B: MG599937 |
WJMV |
|
Wēnlǐng Japanese topeshark birnavirus |
B: MG599938 |
WJTV |
|
Jīngmén birnavirus |
B: KX883000 |
JBV |
|
Húběi birnavirus |
B: KX883001 |
HuBV |
|
chicken proventricular necrosis virus |
B: HM038436 |
CPNV |
Virus names and virus abbreviations are not official ICTV designations.

