Family: Reoviridae
Chapter Version: ICTV Ninth Report; 2009 Taxonomy Release
Virion properties
Morphology
Virus particles of members of the family Reoviridae (collectively called reoviruses) have icosahedral symmetry but may appear spherical in shape. The protein capsid is organized as one, two or three concentric layers of capsid proteins, which surround the linear dsRNA segments of the viral genome, with an overall diameter of 60–80 nm (Figure 1).
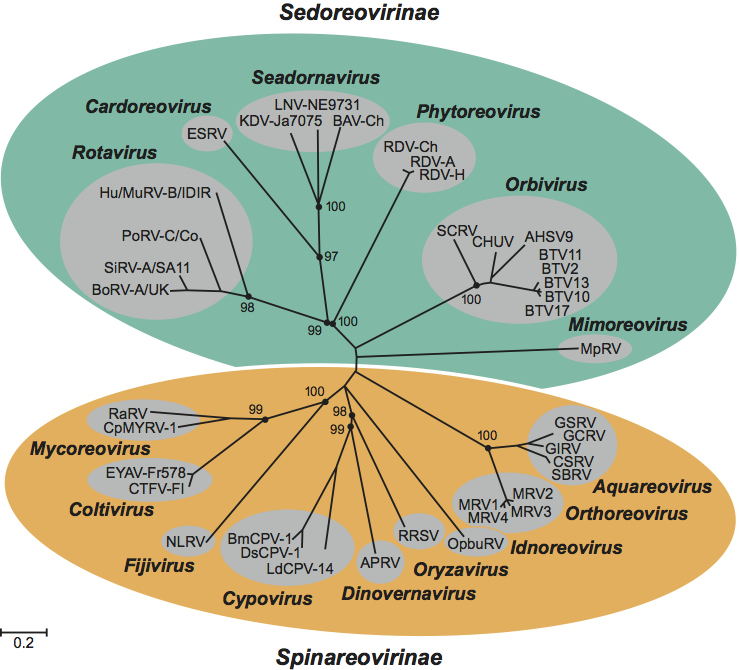
The 15 genera of reoviruses are divided between two subfamilies. The subfamily Spinareovirinae contains viruses that have relatively large spikes or turrets situated at the 12 icosahedral vertices of either the virus or core particle. The subfamily Sedoreovirinae includes viruses that do not have large surface projections on their virions or core particles, giving them an almost spherical or “smooth” appearance.
The terminology that has been used to describe reovirus particles with different numbers of capsid layers varies among the genera. The current nomenclature will therefore be explained in each case. The transcriptionally active core particle of the spiked viruses (subfamily Spinareovirinae) appears to contain only a single complete capsid layer (which has been interpreted as having T=1 or T=2 symmetry), to which the projecting spikes or turrets are attached. In most cases, the core is surrounded (in the complete virion) by an incomplete protein layer (with T=13 symmetry) that forms the outer capsid, which is penetrated by the projections on the core surface. These virus particles are therefore usually regarded as double-shelled.
One exception is the cypoviruses, which have transcriptionally active but fully intact virions with only a single capsid shell that are equivalent to the core particles of viruses from other genera. However, virus particles of most cypoviruses are characteristically occluded (either singly or multiply) within the matrix of proteinaceous crystals called polyhedra. These are composed primarily (>90%) of the viral polyhedrin protein.
In contrast, virions of the non-spiked viruses (subfamily Sedoreovirinae) have an inner protein layer, which may be relatively fragile, having structural similarities to the innermost shell of the spiked viruses (interpreted as having T=2 symmetry). However, in transcriptionally active core particles, the subcore is surrounded and reinforced by a complete core-surface layer, which has T=13 symmetry. These double-layered cores have no surface spikes and (in intact virions) are surrounded by a further outer capsid shell, giving rise to three-layered virus particles that are equivalent to the two-layered particles of members of the subfamily Spinareovirinae.
The innermost protein layer of reovirus particles has an internal diameter of approximately 50–60 nm and surrounds the 9, 10, 11 or 12 linear dsRNA genome segments. In the smooth-cored genera, the enzymatically active minor proteins of the virion are attached to the inner surface of the central space at the five-fold axes of symmetry. These include the RNA-dependent RNA polymerase (RdRp, which functions as a transcriptase and replicase), NTPase, helicase, and capping and transmethylase enzymes. However, in the spiked genera, some of these enzymatic proteins form turrets on the surface of the core. These hollow projections appear to act as conduits for the exit of nascent mRNA synthesized by core-associated enzymes.
Particles of some genera can leave infected cells by budding (e.g. genera Orbivirus and Seadornavirus) or can bud into the endoplasmic reticulum during morphogenesis (genus Rotavirus), acquiring an envelope derived from cellular membranes. However, in most cases, the envelope appears to be transient and is not usually considered to be part of the intact virion. In some genera, the protein components of the outer capsid shell can be modified by proteases (such as trypsin or chymotrypsin) to form infectious or intermediate subviral particles (ISVPs). ISVP formation may occur intracellularly (within endocytic vesicles, which represent an entry route for virus particles taken in from the cell surface), extracellularly (e.g. in the intestinal lumen following ingestion or peroral inoculation, or in the host’s blood stream), or in vitro, following treatment with proteases (including those present in the saliva of insect vector species). The virion-to-ISVP transition can significantly increase, and may even be essential for, infectivity of these viruses.
Physicochemical and physical properties
The virion Mr is about 12×107. The buoyant density in CsCl is 1.36–1.39 g cm−3. Virus infectivity is moderately resistant to heat, organic solvents (e.g. ether) and non-ionic detergents (depending on both virus strain and detergent). The pH stability of virions varies among the genera.
Nucleic acid
Reovirus particles can contain 9, 10, 11 or 12 segments of linear dsRNA, depending on the genus. The individual Mr of these RNA molecules ranges from 0.2 to 3.0×106. The total Mr of the genome is 12–20×106. The RNA constitutes about 15–20% of the virion dry weight. The positive strands of each duplex have a 5′-terminal type 1 cap structure (7mGpppN2’OmpNp…), which was first discovered in the cypoviruses. There are data to suggest that negative strands may have phosphorylated 5′ termini. However, in some cases (e.g. bluetongue virus (BTV), genus Orbivirus), the negative strand has been shown to be poorly labeled (with the same efficiency as the positive strand) by treatment with polynucleotide kinase and [γ-32P]ATP, suggesting that it may also have a blocked 5′ structure. Both RNA strands have a 3′-OH group, and viral mRNAs lack 3′-polyA tails. The viral dsRNA species are present within virus particles in equimolar proportions, representing one copy of each genome segment per virion. Intact virions of some genera also contain significant amounts of short ssRNA oligonucleotides.
Reovirus RNA is usually regarded as non-infectious. However, recent developments involving the introduction of viral mRNAs into susceptible cells have succeeded in recovering fully viable virus particles, thus providing further research opportunities utilizing reverse genetic technologies.
Proteins
At least three internal virion structural proteins have enzyme activities involved in RNA synthesis and capping, including a conservative RdRp or Pol (which may function as a transcriptase, i.e. involved in positive strand synthesis on a dsRNA template, or a replicase, i.e. involved in negative strand synthesis on a positive strand ssRNA template), nucleotide phosphohydrolase, guanylyltransferase, two distinct transmethylases, dsRNA unwinding (i.e. helicase) activity and pyrophosphatase. Some of the minor proteins may also play a structurally significant role as components of the virion, together with at least three major capsid proteins. The virion structural proteins range in size from 15 to 155 kDa and constitute about 80–85% of the dry weight of virions.
Lipids
Mature virions lack a lipid envelope. Depending on the genus, a myristyl residue may be covalently attached to one of the virion proteins. Coltiviruses, rotaviruses and orbiviruses have an intermediate in virus morphogenesis or release, which may have a lipid envelope that is subsequently lost or removed. However, this may help to explain why virus particles are in some cases associated with membrane fractions in infected cell lysates.
Carbohydrates
In some genera, one of the outer capsid proteins can be glycosylated with high mannose glycans or O-linked N-acetylglucosamine. A small non-structural (NS) viral protein may also be glycosylated.
Genome organization and replication
The viral RNA species are mostly monocistronic, although some segments have second functional, in-frame initiation codons or additional protein-coding ORFs. Proteins are encoded on one strand only of each duplex (the mRNA species). The mode of entry of viruses into cells varies between genera but usually results in loss of outer capsid components. Transcriptionally active particles derived from the parental virions (cores, represented by single or double layered particles, from subfamily Spinareovirinae or Sedoreovirinae, respectively) are released into the cell cytoplasm (Figure 2). Repetitive asymmetric transcription of full-length mRNA species from each dsRNA segment occurs within these particles throughout the course of infection. The mRNA products, which are produced in larger copy numbers from the smaller segments, are extruded from the icosahedral apices of these particles. Structures that have been called viroplasms or virus inclusion bodies (VIBs) occur in localized areas of the infected cell cytoplasm. They appear to be the sites of viral mRNA synthesis, genome replication and particle assembly. VIBs have a granular and moderately electron-dense appearance when viewed by electron microscopy, and usually contain nascent subviral particles. Outer capsid components appear to be added to progeny virus particles at the periphery of the VIB and are thought to stop further mRNA synthesis.
The mechanism of genome assembly and synthesis remains largely uncharacterized. For orthoreoviruses and rotaviruses, evidence has been obtained that sets of capped mRNAs and certain NS proteins are incorporated into assortment complexes that are considered to be the precursors of progeny virus particles. These mRNAs are then used as templates for a single round of minus strand synthesis, thereby reforming the dsRNA genome segments of a progeny virus particle. The various species of mRNAs in the cell cytoplasm are present in non-equimolar ratios. However, the dsRNA genome segments are usually packaged in exactly equimolar ratios (i.e. one copy of each genome segment per particle). The selection of viral mRNAs for packaging is therefore thought to be highly specific, involving recognition signals on each mRNA species. Genome segment re-assortment, involving the selection and packaging of mRNAs from different parental strains, occurs readily in cells that are co-infected with different viruses of the same species, which presumably share the same packaging signals.
The RNA segments have conserved terminal sequences at both ends, which may be involved as recognition signals for the viral transcriptase and replicase functions. These sequences may also be essential for selection and incorporation of the RNAs into the nascent progeny particles, and may play some role in efficient initiation of translation of the viral mRNAs. In many cases, sequences near to the 5′ and 3′ termini of the positive sense viral RNAs share extensive complementarity, interrupted by short sequences predicted to form stem loops and other secondary structures. These findings and mounting experimental evidence suggest that the viral mRNAs contain both primary sequences and higher-order structures that are involved in regulation of RNA function (i.e. translation, replication or packaging). A consistent feature of the secondary structures predicted for viral positive sense RNAs is that the conserved 5′- and 3′-terminal sequences remain non-hybridized. Indeed, non-hybridized 3′-terminal sequences have been shown to be required for efficient negative-strand synthesis. The dsRNA within assembled particles has been shown, in at least some genera, to be packaged as a series of concentric and highly organized shells, which also have elements of icosahedral symmetry.
In addition to the parental virus-derived subviral particles (smooth cores), progeny cores (single or double layered particles from subfamily Spinareovirinae or Sedoreovirinae, respectively) also synthesize mRNAs, providing an amplification step in replication. Depending on the genus, some NS proteins are involved in translocation of virus particles within cells or virus egress by budding. Many cypoviruses also form polyhedra, which are large crystalline protein matrices that occlude virus particles (either singly or multiply) and which appear to be involved in transmission between individual insect hosts. The steps involved in virion morphogenesis and virus egress from cells vary according to genus. The only known examples of non-enveloped viruses that induce cell–cell fusion and syncytium formation in virus-infected cells are members of the family Reoviridae. In the case of fusogenic orthoreoviruses, syncytium formation promotes a rapid lytic response and release of progeny virions.
Antigenic properties
The viruses that infect vertebrate hosts generally possess both serogroup- (virus species) specific antigens, and (within each species or serogroup) more variable serotype-specific antigens. The viruses that infect plants and insects only may show greater uniformity and less antigenic variation in their proteins, possibly due to the lack of neutralizing antibodies in the host and therefore the absence of antibody selective pressure on neutralization-specific antigens. No antigenic relationship has been found between the viruses in different genera. Some viruses bind erythrocytes (i.e. hemagglutinate).
Biological properties
The biological properties of the viruses vary according to genus. Some viruses replicate only in certain vertebrate species (orthoreoviruses and rotaviruses) and are transmitted between hosts by respiratory or fecal–oral routes. Other vertebrate viruses (orbiviruses, coltiviruses and seadornaviruses) replicate in both arthropod vectors (e.g. biting midges, mosquitoes or ticks) and vertebrate hosts. Plant viruses (phytoreoviruses, fijiviruses and oryzaviruses) replicate in both plants and arthropod vectors (leafhoppers). Viruses that infect insects (cypoviruses) are transmitted by contact or fecal–oral routes.
Genus and species demarcation criteria in the family
The number of genome segments (usually 9, 10, 11 or 12) is in most cases characteristic of viruses within a single genus, although the genus Mycoreovirus currently contains viruses with both 11 and 12 genome segments. Host (and vector) range and disease symptoms are also important indicators that help to identify viruses from different genera. Capsid structure (number of capsid layers, the presence of spiked or unspiked cores, and the symmetry and structure of the outer capsid) can also be significant. The level of sequence divergence, particularly in the more conserved genome segments and proteins (for example as detected by comparisons of RdRp or inner capsid shell proteins and the segments from which they are translated) can be used to distinguish members of different genera. Available data suggests that isolates from different genera usually have <26% amino acid identity in comparisons between their RdRps, while within a single genus identities are usually >33%. However, the RdRp of Rotavirus B isolates shows a high level of amino acid sequence divergence from that of other rotaviruses (<21% identity).
The prime determinant for inclusion of virus isolates within a single virus species is their ability to exchange genetic information during co-infection, by genome segment re-assortment, thereby generating viable progeny virus strains. However, data providing direct evidence of segment re-assortment between isolates are only available for viruses in a few genera. The following methods are therefore commonly used (preferably in combination) to examine levels of similarity between isolates and to predict their possible compatibility:
- Cross-hybridization assays (northern or dot blot), with probes made from viral RNA or cDNA. Stringency conditions may be selected so that viruses within a species will show hybridization.
- Nucleotide and amino acid sequence analysis (viruses within different species should have low levels of sequence similarity among the cognate genome segments).
- Serological comparisons of antigens or antibodies using either polyclonal antisera or monoclonal antibodies against conserved antigens. Methods used may include ELISA, complement fixation and agar gel immunodiffusion. Closely related isolates and serotypes generally belong to the same species.
- Analysis of electropherotype by agarose gel electrophoresis (AGE) but not by PAGE. Virus isolates within the same species will show a relatively uniform electropherotype. However, a major deletion/insertion event may sometimes result in two distinct electropherotypes within a single species, and similarities can exist between more closely related species.
- Identification of the conserved terminal regions of the genome segments. These are usually conserved across all segments within a species although some closely related species can also have identical terminal sequences on at least some segments.
These criteria apply throughout the family. Additional or more specific criteria are provided in the section for each genus, where applicable.
Subfamily Spinareovirinae
Genus Orthoreovirus
Type species Mammalian orthoreovirus
Distinguishing features
Orthoreoviruses infect only vertebrates and are spread by respiratory or fecal–oral routes. All members of the genus have a well-defined capsid structure, as observed by electron microscopy and negative staining, with 12 spikes or turrets situated on the surface of the core particle at the icosahedral vertices. They also contain 10 linear dsRNA molecules that include three large (L), three medium (M) and four small (S) size-class segments. They have a characteristic protein profile with three λ, three µ and four σ primary translation products, as well as additional small gene products that are encoded by polycistronic segments. Members of all of the five species, except Mammalian orthoreovirus, induce syncytium formation.
Virion properties
Morphology
Virion morphology and construction is illustrated in Figure 3. Virions are icosahedral with a roughly spherical appearance and possess a double-layered protein capsid, the different layers of which are discernible by negative staining and electron microscopy (Figure 3A). Higher-resolution images have been obtained by cryo-electron microscopy (cryoEM) and image reconstruction of mammalian orthoreovirus (MRV) and avian orthoreovirus (ARV) particles. These are similar with a central compartment (about 48 nm in diameter) containing the dsRNA genome segments, surrounded by an inner capsid that has T=1 symmetry (60 nm diameter: composed of 120 copies of protein λ1(Hel)) and an outer capsid (85 nm diameter) that has T=13 (laevo) symmetry. The inner-capsid of the orthoreoviruses is equivalent to the T=1 core-particle of the rotaviruses and the sub-core of the orbiviruses (which is also composed of 120 molecules (of VP3) interpreted as having T=2 pseudo icosahedral symmetry). The surface of the complete orthoreovirus particle is covered by 600 finger-like projections arranged in 60 hexameric and 60 tetrameric clusters that surround solvent channels, which extend radially into the outer capsid layer (Figure 3B).
Intact virions also contain large, open depressions with a flower-shaped structure at the five-fold axes, resulting in an angular capsid profile when viewed in the three-fold orientation (Figure 3A, 3B). ISVPs, which are generated by partial removal of the outer capsid proteins (Figure 3C), are approximately 80 nm in diameter. The flower-shaped structures at the five-fold axes of the ISVPs may contain an extended form of the viral attachment protein, σ1, which protrudes as a 40 nm fibre from the vertices. MRV core particles generated by more extensive removal of the outer capsid proteins (Figure 3D) have also been examined by X-ray crystallography and have 150 ellipsoidal nodules (protein σ2) on their surface and distinctive turrets located at the five-fold axes. These projections, which are altered conformations of the flower-shaped structures observed on intact virions (composed of protein λ2(Cap), the viral capping enzyme) are about 10 nm in length, possessing central channels 5–8 nm in diameter extending into the central compartment.
Physicochemical and physical properties
The virion Mr is about 130×107 with a buoyant density in CsCl of 1.36 g cm−3 (1.38 g cm−3 for ISVPs, 1.43 g cm−3 for core particles). The virion, ISVP and core S20,w values are about 730S, 630S and 470S, respectively. Virions are remarkably stable and withstand extremes of ionic conditions, temperatures up to 55 °C, pH values between 2 and 9, lipid solvents, and detergents. Exposure to UV irradiation reduces infectivity.
Nucleic acid
All orthoreoviruses have 10 linear dsRNA segments that range from 0.60×106 to 2.60×106 Mr. The total Mr of the MRV-3 (strain Dearing) genome is about 1.5×107 (23,549 bp) and constitutes approximately 11.5% of the virion mass. Based on their resolution by gel electrophoresis, the genomic dsRNAs are grouped into three size classes commonly referred to as large (L1–L3, about 3.9–3.8 kbp), medium (M1–M3, about 2.3–2.2 kbp) and small (S1–S4, about 1.6–0.9 kbp). The gel mobilities of certain genome segments are characteristic of the five distinct species of orthoreoviruses. In comparison to isolates of the type species Mammalian orthoreovirus, most isolates of ARV, Nelson Bay orthoreovirus (NBV) and reptilian orthoreovirus (RRV) display retarded genome segment migration of their polycistronic S1 genome segments. Baboon orthoreovirus (BRV) and the Muscovy duck isolates of ARV (ARV-Md) have truncated polycistronic S1 genome segments that migrate as the S4 genome segment by PAGE.
Complete virus particles contain numerous oligonucleotides (2–20 nt) representing approximately 25% of the total RNA content. Three-quarters of these are abortive reiterative 5′-terminal transcripts, produced by the reovirus core-associated transcriptase and capping enzymes, while the remainder are oligoadenylates. The 5′ terminus of the positive-sense RNA strand of each genome segment contains a dimethylated Cap 1 structure (m7GpppGm2'OH). The genomic RNAs lack polyA tails and do not contain covalently linked proteins. Genomic dsRNA segments contain 5′- and 3′-terminal sequences of 4 or 5 bp that are conserved in all 10 genome segments within a particular virus species. The 3′-terminal consensus sequence (UCAUC-3′) is also conserved between orthoreovirus species, at least as assessed from the available sequences of the four S-class genome segments. The 5′-terminal conserved sequences vary and may be useful for assigning new isolates to one of the five species or subgroups thereof (Table 1).
Table 1 Conserved terminal sequences (positive strand) of orthoreovirus genome segments
| Virus species | Serotype or strain | 5′ end | 3′ end |
| Mammalian orthoreovirus | MRV-1La* | 5′-GCUA | UCAUC-3′ |
| Avian orthoreovirus | ARV-138** | 5′-GCUUUUU | UCAUC-3′ |
| Nelson Bay orthoreovirus | NBV | 5′-GCUUUA | UCAUC-3′ |
| Baboon orthoreovirus | BRV | 5′-GUAAAUUU | UCAUC-3′ |
| Reptilian orthoreovirus | RRV-Py | 5′-GUUAUUUU | UCAUC-3′ |
* Also MRV-2Jo, MRV-3De and MRV-4Nd.
** Also ARV-176 and ARV-Md.
Proteins
The orthoreovirus structural proteins are designated in terms of their relative sizes and size classes: λ1, λ2, λ3; µ1, µ2; and σ1, σ2, σ3. In ARV, these proteins are referred to as λA, λB, λC; µA, µB; and σA, σB, σC (Table 2). The following discussion refers to the nomenclature scheme for prototype strain MRV-3.
Table 2 Genome segments and protein products of mammalian orthoreovirus-3De
| Genome segment | Size (bp) | Proteins (structure/function)* | Size (kDa) | Protein copies per particle | Location | Function |
| L1 | 3854 | λ3 (Pol) | 142 | 12 | core | RNA polymerase |
| L2 | 3916 | λ2 (Cap) | 144 | 60 | core spike | Guanylyl transferase, methyl transferase turret protein |
| L3 | 3896 | λ1 (Hel) | 143 | 120 | core | Inner capsid structural protein, binds dsRNA, NTPase, helicase |
| M1 | 2304 | µ2 | 83 | 12 | core | NTPase, required for inclusion body development, probable polymerase subunit, cell tropism, modulation of cellular interferon response |
| M2 | 2203 | µ1 | 76 | 600 | outer capsid | Multimerizes with σ3 and cleaved to µ1C and µ1N during viral entry, assumes T=13 symmetry in the outer capsid |
|
| µ1C (T13) | 72 |
| µ1C further cleaved to δ and φ during the entry process, myristoylated N-terminus, membrane penetration, apoptosis | ||
| δ | 59 |
| ||||
| φ | 13 |
| ||||
| µ1N | 4 |
|
| |||
| M3 | 2235 | µNS / µNSC | 80/75 | 0 | NS | Binds ssRNA and cytoskeleton, nucleates viral inclusion bodies, phosphoprotein, µNSC (unknown function) from alternate translation start site |
| S1 | 1416 | σ1 | 49 | 36 | outer capsid | Viral attachment protein, homotrimer, hemagglutinin, type-specific antigen, cell tropism, pathways of viral spread in vivo, virulence |
|
| σ1s | 16 | O | NS | Viral spread in vivo, cell cycle arrest | |
| S2 | 1331 | σ2 | 47 | 150 | core | Inner capsid structural protein, weak dsRNA-binding, morphogenesis? |
| S3 | 1189 | σNS | 41 | 0 | NS | ssRNA-binding, inclusion body development, genome packaging? |
| S4 | 1196 | σ3 | 41 | 600 | outer capsid | dsRNA-binding, multimerizes with µ1, nuclear and cytoplasmic localization, translation control, modulation of cellular interferon response, zinc-binding |
* Protein structure/function: Pol, RNA polymerase ; Cap, capping enzyme (guanylyltransferase and transmethylase); T13, virus structural protein with T=13 symmetry; Hel, protein with helicase activity.
The stabilizing lattice of the outer capsid is composed of 200 interlocking trimers of the 76 kDa µ1 protein. The µ1 subunits also interact with monomers of the σ3 protein, which represent fingerlike projections on the surface of the virion. Pentameric subunits of the λ2 protein make up the flower-like structures and turrets at the vertices of viral particles and cores, respectively. The λ2 structures interact with subunits of the tetrameric σ3 clusters and with the µ1 lattice and represent essential structural components of the outer capsid. This essentially outer capsid protein (CP) remains associated with core particles, unlike the other outer CPs. The fourth component of the outer capsid, the σ1 protein, exists as 12 homotrimers associated with the vertices of virions and ISVPs. It may assume either a retracted or extended conformation. The λ1 (120 copies) and σ2 proteins (150 copies) represent the major structural proteins of the inner capsid. The final two structural proteins of the virus, λ3 and µ2, are present at about 12 copies per virion and located on the inside of the inner capsid. The λ3 protein forms 7 nm projections that extend toward the interior of the core, underlying the 12 vertices of the capsid. The µ2 protein may be associated with these λ3 structures.
Lipids
Mature virions lack a lipid envelope. The major outer capsid lattice protein, µ1, and its µ1N cleavage product are N-terminally myristoylated. The small NS proteins responsible for syncytium formation induced by the fusogenic orthoreoviruses are either N-terminally myristoylated or palmitoylated at internal cysteine residues. These acylations are essential for the membrane fusion activity of the proteins.
Carbohydrates
Convincing evidence that any of the orthoreovirus proteins are glycosylated has not been reported. Moreover, no carbohydrate has been observed in the structures of any of the mammalian reovirus proteins that have been determined by X-ray crystallography (λ1, λ2, λ3; µ1; and σ1, σ 2, σ3).
Genome organization and replication
The genome consists of ten segments of linear dsRNA, which are packaged in equimolar ratios (one copy of each within each virion). The segments possess terminal non-translated regions (NTRs) that are shorter at the 5′ terminus (12–32 bp for MRV-3De) than at the 3′ terminus (35–85 bp). The major ORFs vary in length from 1059 to 3867 bp. The MRV S1 segment is bicistronic, encoding the 49 kDa σ1 protein and the 14 kDa σ1s protein from a second overlapping ORF (Table 2). The S1 genome segments of ARV and NBV are functionally tricistronic, encoding the viral attachment protein σC, a membrane-associated protein (p17) of unknown function, and a fusion-associated small transmembrane (FAST) protein (p10) responsible for virus-induced syncytium formation. The RRV S1 genome segment is bicistronic, encoding a σC viral attachment protein homolog and a novel FAST protein (p14). The truncated S1 genome segment-equivalent (S4, 1124 bp) of Muscovy duck reovirus (ARV-Md) encodes a σC viral attachment protein homolog and a p10 protein that shares limited sequence similarity to the p10 FAST proteins of NBV and other ARV isolates. The truncated S1 genome segment-equivalent of BRV (S4, 887 bp), contains two sequential 140–141 codon ORFs, one of which encodes a third unique FAST protein (p15) and the other a novel NS protein (p16) of unknown function (Figure 4).
The overall course of infection involves adsorption, low pH-dependent penetration and uncoating to core particles, asymmetric transcription of capped, non-polyadenylated mRNAs via a fully conservative mechanism (the nascent strand is displaced), translation, assembly of positive strands into progeny subviral particles, conversion of positive strands to dsRNA, and further rounds of mRNA transcription and translation. The efficiency of translation of the various orthoreovirus mRNA species varies over a 100-fold range, while the proportions of the mRNA species found in infected cells vary inversely to their proportionate size. The final stage of the replication cycle involves the assembly of the outer capsid onto progeny subviral particles to form infectious virions. Based on studies of MRV replication, virion morphogenesis is thought to proceed along a pathway involving a series of assembly intermediates. Progeny particles accumulate in paracrystalline arrays in the perinuclear region of the cytoplasm and are released when infected cells lyse late in the replication cycle. The exception to the above generalized replication cycle involves the formation of multinucleated syncytia by ARV, BRV, RRV and NBV. Syncytia formation commences 10–12 h post infection, resulting in a more rapid lytic response and enhanced kinetics of virus release.
The functions and properties of specific viral proteins influence various stages of the MRV replication cycle (Table 2). The MRV σ1 viral attachment protein determines the cell and tissue tropism of the virus strain and has hemagglutination activity. The σ1 protein binds cell-surface carbohydrate and junctional adhesion molecule-A. The µ1 protein is N-terminally myristoylated and forms a complex with σ3 in solution that triggers cleavage of µ1 to µ1N and µ1C. The µ1C fragment is further proteolytically cleaved into δ and φ polypeptides during virus entry into cells and is responsible for membrane penetration. The µ1 protein also influences strain-specific differences in capsid stability, transcriptase activation, apoptosis and neurovirulence. In the case of ARV, the µ1 homolog (µB) has been implicated in strain-specific differences in macrophage infection. In addition to interacting with µ1 and forming the outer capsid layer of the virion, the σ3 protein is a dsRNA-binding protein involved in translation regulation, altering the activity of protein kinase R (PKR), and modulating the interferon response. The λ2 core spike is the guanylyl transferase involved in mRNA capping, while the λ1 and σ2 major inner capsid proteins both bind dsRNA. The λ1 protein may also function as a helicase and an RNA triphosphatase. The minor inner CP λ3 is the viral polymerase, while the second minor inner CP µ2, along with the major inner CP λ1, is involved in the NTPase activity associated with core particles.
There are also at least three NS proteins encoded by the MRV genome: µNS, σNS, and σ1s. The µNS and σNS proteins are produced in high abundance during infection and, together with σ3, associate with mRNA to form virus mRNA-containing complexes, which are presumed to be precursors of progeny virus assembly.
The σNS protein binds ssRNA, and the µNS protein associates with the cytoskeleton. Core protein µ2 stabilizes microtubules within viral inclusions and associates with µNS, which is an organizing center for inclusion formation. Co-expression of µNS and σNS proteins in mammalian cells from cloned viral cDNAs yields punctate structures resembling intracytoplasmic inclusions of virally infected cells. The σ1s protein is a small, basic protein expressed in cells infected by all three MRV serotypes. This protein is required for MRV dissemination in infected mice and contributes to cell-cycle arrest during MRV infection. The relationship between these σ1s-mediated properties is currently unknown. It is dispensable for growth in cell culture but is involved in cell cycle arrest at the G2/M checkpoint.
Replication strategies used by ARV, NBV, RRV and BRV are similar to that described for MRV, with some notable exceptions. The truncated viral attachment protein of ARV, RRV and NBV, σC (35 kDa), exists as a multimer with a coiled-coil domain similar to that of MRV but possesses no hemagglutination activity. BRV is unique in that the S-class genome segments encode no homolog of the ARV, NBV, RRV or MRV viral attachment proteins. The dsRNA-binding domain of the MRV major sigma-class outer CP σ3 is not conserved in the homologous σB proteins of ARV, NBV or BRV. As with the MRV σ2 protein, the major sigma-class core protein of ARV, σA, displays dsRNA-binding activity. The ARV σA core protein may function analogously to the σ3 major outer CP of MRV by regulating PKR activity and the interferon response. ARV, NBV, RRV and BRV encode an additional FAST (fusion-associated small transmembrane) protein responsible for syncytium formation. The p10 FAST proteins of ARV and NBV share sequence and structural similarities, but are unrelated to the p15 and p14 FAST proteins of BRV and RRV, respectively. All of these FAST proteins are small, basic, acylated, transmembrane proteins and induce fusion in transfected cells in the absence of other viral proteins.
Antigenic properties
The serotype-specific antigen of the orthoreoviruses is protein σ1 (σC of the avian species), which is recognized by neutralizing antibodies. Antigenic recognition of this protein is the basis for three major serotypes of MRV and 5–11 serotypes of ARV. Ndelle virus was isolated from a mouse and originally classified as an orbivirus. Recent sequence data revealed that Ndelle virus is actually an orthoreovirus and is closely related to MRV-1 and MRV-3. However, neutralizing antibodies against the three major MRV serotypes do not neutralize Ndelle virus, indicating that it represents a fourth MRV serotype. The MRV σ1 and σ1s proteins elicit strain-specific and cross-reactive cytotoxic T-cell activities. The MRV proteins λ2 and σ3 are species-specific antigens, similar to the λB and σB proteins of ARV (Figure 3). The considerable sequence similarity that exists between different isolates in the same orthoreovirus species, but not among species, is reflected by the limited antigenic cross-reactivity detected among species. The most extensive antigenic similarity between species subgroups occurs between ARV and NBV, which is in accordance with the increased amino acid sequence identity between these species.
Biological properties
Transmission is by an enteric or respiratory route, no arthropod vectors are involved, and infection is restricted to a variety of vertebrate species (baboons, bats, birds, cattle, humans, monkeys, sheep, snakes, swine and rodents). Orthoreovirus distribution is worldwide. Human orthoreoviruses generally do not produce symptoms, but may cause upper respiratory tract illness and possibly enteritis in infants and children (albeit rare). In mice, orthoreovirus infection can cause diarrhea, runting, oily hair syndrome, hepatitis, jaundice, myocarditis, myositis, pneumonitis, encephalitis and hydrocephalus. A variety of symptoms may be associated with orthoreovirus infection of domestic animals including upper and lower respiratory illnesses and diarrhea. In monkeys, orthoreoviruses cause hepatitis, extrahepatic biliary atresia, meningitis and necrosis of ependymal and choroid plexus epithelial cells. The prototype BRV isolate was obtained from baboons with meningoencephalomyelitis. RRV isolates from snakes were obtained from animals displaying neurological symptoms. The outcome of ARV infection in birds may range from inapparent to lethal, depending on the virus strain and the age of the host. Systemic infection results in virus dissemination to numerous tissues. Disease presentations in chickens include feathering abnormalities, gastroenteritis, hepatitis, malabsorption, myocarditis, paling, pneumonia, stunted growth and weight loss. In turkeys, ARVs cause enteritis. Birds that survive an acute systemic infection may develop obvious joint and tendon disorders (tenosynovitis) that resemble the pathology of rheumatoid arthritis in humans. ARVs do not infect mammals.
MRV and ARV induce the biochemical and morphologic hallmarks of apoptosis in cultured cells. MRV infection leads to activation of nuclear factor kappa B (NF-κB), a family of transcription factors known to play important roles in regulating cellular stress responses, including apoptosis. The µ1 cleavage fragment φ, which is released following disassembly, is an important trigger of NF-κB activation, but the precise mechanism is unclear. Apoptosis induced by reovirus requires both extrinsic (death-receptor) and intrinsic (mitochondrial) signaling pathways linked by the small Bcl-2 family member, Bid. As with MRV, ARV-induced apoptosis requires virus disassembly but not viral transcription.
Recent studies indicate that MRV preferentially replicates in a lytic manner in transformed cells. The basis for this cell tropism has been suggested to result from an activated Ras pathway in transformed cells on modulation of PKR activity and regulation of the translation machinery. These observations have led to the development of orthoreoviruses as an oncolytic agent for cancer therapy.
Species demarcation criteria in the genus
The orthoreoviruses include five species. The classification is supported by experiments showing re-assortment of genome segments between isolates of the same species but not between those of different species. In addition to the other general criteria used throughout the family, members of a species in the genus Orthoreovirus may be identified by:
- Extensive sequence identity between the proteins encoded by homologous genome segments (for conserved core proteins, >85% amino acid sequence identity within a species versus <65% identity between species; for the more divergent outer CPs, >55% identity within a species and <35% between species)
- Extensive sequence identity between homologous genome segments (for most genome segments, >75% nucleotide sequence identity within a species versus <60% between species)
- Analysis of electropherotype by agarose gel electrophoresis but not by PAGE (some similarities can exist between closely related species)
- Similar organization of the polycistronic genome segment
- Identification of host species and clinical signs
List of species in the genus Orthoreovirus
| Mammalian orthoreovirus |
|
|
| Mammalian orthoreovirus 1 Lang | [L1: M24734, L2: AF378003, L3: AF129820, M1: AF461682, M2: AF490617, M3: AF174382, S1: M14779, S2: M17598, S3: M14325, S4: M13139 ] | (MRV-1La)
|
| Mammalian orthoreovirus 2 D5/Jones
| [L1: P17377, L2: Q91RA4, L3: Q9WAB1, M1: Q91PK4, M2: M19355, M3: Q9PY82, S1: M10261, S2: P32922, S3: P12002, S4: P30211] | (MRV-2Jo)
|
| Mammalian orthoreovirus 3 Dearing | [L1: M24734, L2: J03488, L3: M13139, M1: M27261, M2: M19408, M3: M27262, S1: M10262, S2: M25780, S3: X01627, S4: K02739] | (MRV-3De) |
| Mammalian orthoreovirus 4 Ndelle | [L1: AF368033, M2: AF368034, S1: AF368035, S2: AF368036, S4: AF368037] | (MRV-4Nd) |
| Avian orthoreovirus |
|
|
| Avian orthoreovirus S1133 {chicken isolate} | [S1: L39002, S2: AF104311, S3: U20642, S4: U95952] | (ARV-1133) |
| Avian orthoreovirus 176 {chicken isolate} | [S1: AF218358, S2: AF059716, S3: AF059720, S4: AF059724] | (ARV-176) |
| Avian orthoreovirus SK138a {chicken isolate} | [S1: AF218359, S2: AF059717, S3: AF059721, S4: AF059725] | (ARV-138) |
| Avian orthoreovirus 1733 {chicken isolate} | [L3: AF384171] | (ARV-1733) |
| Avian orthoreovirus 89026 {Muscovy duck isolate} | [S1: AJ278102, S2: AJ006476, S3: AJ133122, S4: AJ310525, AJ310526] | (ARV-Md89026) |
| Avian orthoreovirus 89330 {Muscovy duck isolate} | [M3: AJ293969] | (ARV-Md89330) |
| Avian orthoreovirus D15/99 {goose isolate} | [S1: AY114138] | (ARV-Go) |
| Avian orthoreovirus NC98 {turkey isolate} | [S3: AF465799] | (ARV-Tu) |
| Baboon orthoreovirus |
|
|
| Baboon orthoreovirus | [S1: AF059719, S2: AF059723, S3: AF059727, S4: AF406787] | (BRV) |
| Nelson Bay orthoreovirus |
|
|
| Nelson Bay orthoreovirus | [S1: AF218360, S2: AF059718, S3: AF059726, S4: AF059722] | (NBV) |
| Reptilian orthoreovirus |
|
|
| Reptilian orthoreovirus - Python | [S1: AY238887, S3: AY238886] | (RRV-Py) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ] and assigned abbreviations ( ) are also listed.
Mammalian orthoreovirus includes all the nonfusogenic orthoreoviruses, with three major serotypes (MRV-1, MRV-2 and MRV-3) representing numerous isolates, and a fourth serotype with only one isolate, Ndelle reovirus (MRV-4Nd). Amino acid sequence identities of the sigma-class major outer CPs and core proteins of various MRV serotypes range from 90 to 97%.
Avian orthoreovirus contains numerous isolates from commercial poultry flocks, including chickens, Muscovy ducks, turkeys and geese, and includes several different serotypes. Sequence diversity is more extensive among the various ARV isolates than among MRV isolates (54–95% in the sigma-class major outer CP).
Nelson Bay orthoreovirus contains an atypical syncytium-inducing mammalian reovirus isolated from a flying fox. The sequence similarity between NBV and ARV exceeds that between NBV and the other species subgroups. ARV and NBV also share more extensive antigenic similarity than other species, possess more similar conserved terminal genome segment sequences, display a similar gene organization of the polycistronic S1 genome segment, and encode homologous p10 fusion proteins (Table 2). These observations indicate that NBV is more closely related to ARV isolates than to other mammalian or reptilian reovirus isolates. Although ARV and NBV clearly share a more recent evolutionary past than the other reovirus species, in view of the extent of sequence divergence (59–61% identity in the sigma-class core protein and only 29–36% identity in the sigma-class major outer CP) and the absence of evidence for re-assortment between the ARV and NBV isolates, these isolates are considered as two separate species.
Baboon orthoreovirus contains a single isolate, BRV. This atypical mammalian isolate induces syncytium formation but shares little sequence (16–32% amino acid sequence identity between homologous S-class gene products) or antigenic similarity with the other fusogenic species. BRV contains a truncated, fusion-inducing, polycistronic S1 genome segment-equivalent (the S4 genome segment) with a distinct gene organization, a fusion protein (p15) with no sequence or sequence-predicted structural similarity to the fusion proteins of ARV or NBV, and a unique 5′-terminal consensus sequence. This isolate clearly represents a distinct species of the orthoreoviruses.
Reptilian orthoreovirus represents the RRVs. Sequence information is available for the polycistronic S1 genome segment and the S-class genome segment encoding the sigma-class major outer CP of an RRV isolate from a python (RRV-Py). Several additional isolates have been obtained from other snakes and iguanas, but no sequence information is currently available. RRV contains the conserved 3′-terminal pentanucleotide sequence of the orthoreoviruses (UCAUC-3′) but possesses a unique 5′-terminal conserved sequence (5′-GUUA) (Table 1). The S1 genome segment of RRV-Py is bicistronic, encoding a viral attachment protein homolog and a novel p14 FAST protein that induces syncytium formation. Amino acid sequence identities between the RRV sigma-class major outer CP and the homologous protein of other species subgroups are 16–25%, clearly indicating that RRV represents a distinct species of orthoreoviruses.
List of other related viruses which may be members of the genus Orthoreovirus but have not been approved as species
None reported.
Phylogenetic relationships within the genus
The five species of orthoreoviruses represent evolutionarily distinct lineages, as illustrated by phylogenetic analysis using the amino acid sequences of the sigma-class major outer CP, for which the greatest number of sequences from diverse isolates is available (Figure 5). Identical phylogenetic relationships are generated by comparison of the NS proteins (data not shown).
Genus Aquareovirus
Type species Aquareovirus A
Distinguishing features
Aquareoviruses physically resemble orthoreoviruses but possess 11 dsRNA genome segments. They infect a variety of aquatic animals, including finfish and crustacea. Aquareoviruses replicate in cell cultures of piscine and mammalian origins, at temperatures between 15 and 25 °C. Large syncytia are produced as a typical cytopathic effect of infection by a majority of aquareoviruses.
Virion properties
Morphology
Aquareovirus particles are spherical in appearance with diameter of about 80 nm composed of multiple capsid layers (Figure 6, upper and middle panels). The outermost layer, formed by VP5–VP7 heterodimers, consists of 600 subunits (200 trimers) arranged on an incomplete T=13 icosahedral lattice, with an overall structural organization identical to those of MRV and ARV. A distinguishing feature on the outer layer is the five-fold proximal depressions, resulting from missing peripentonal trimers (Figure 6, upper left panel, P1 position indicated by arrows).
The shaded surface view of the aquareovirus core structure (Figure 6, upper right panel), shows the innermost capsid shell, which is about 600 Å in diameter. Twelve VP1 pentameric turrets decorate the shell of 120 VP3 monomers, which are arranged with icosahedral symmetry that is interpreted as T=1 (comparable to the sub-core of the orbiviruses and the innermost capsid shell of the rotaviruses), and are clamped together by 120 VP6 monomers.
Removal of VP7 generates ISVPs, which have a smooth surface formed by a network of VP5 trimers (Figure 6, bottom panel). The atomic model of ISVPs contains six conformers of four proteins: two of VP3, two of VP6, one VP1 on the core, and one VP5 on the coat.
The aquareovirus particle morphology is strikingly similar to that of the orthoreovirus ISVP. A noticeable morphology distinction between aquareovirus and orthoreoviruses is that aquareovirus particles lack the hemagglutinin spike protein σ1 observed in orthoreoviruses.
Physicochemical and physical properties
The virion buoyant density in CsCl is 1.36 g cm−3 with a sedimentation coefficient of about 550S. Virus infectivity is stable between pH 3 and pH 10. Virus infectivity is not affected by treatment with ether or chloroform. Exposure to UV irradiation reduces infectivity. None of the viral proteins is removed from the particle by treatment with 3 mM EDTA or cesium salts. Aquareoviruses held at 4, 16 or 23 °C in minimal essential medium (MEM) with 5% serum showed no significant reduction in infectivity over a period of 28 days. However, all virus infectivity is lost after incubation at 45 °C for 7 days. Virus infectivity is rapidly inactivated by heating to 56 °C.
Nucleic acid
The aquareovirus genome is composed of 11 segments of dsRNA that are packaged in equimolar ratios. The Mr of the dsRNA segments range from 0.4 to 2.6×106. The total Mr of the golden shiner reovirus (GSRV) is about 1.5×107 (23,695 bp). The genomic RNAs are named segment 1 (Seg1) to segment 11 (Seg11) in order of increasing electrophoretic mobility in 1% agarose gels. The genome segments migrate as three size classes. There are three large (Seg1 to Seg3, about 3.9–3.8 kbp), three medium (Seg4 to Seg6, about 2.3–2.0 kbp) and five small segments (Seg7 to Seg11, about 1.4–0.8 kbp). Six distinct species (Aquareovirus A to Aquareovirus F) were originally identified by reciprocal RNA–RNA hybridization studies, but can also be distinguished by nucleotide sequence analyses. The genome segment migration pattern (electropherotype), as analyzed by electrophoresis in 1% agarose gel, is consistent within a single species but shows significant variation among species. However, viruses within a single species can show variations in electropherotype, when their dsRNA genome segments are analyzed by electrophoresis in high percentage (>6%) polyacrylamide gels.
The G+C content of aquareoviruses ranges between 52 and 60%. The complete genomic sequences of GSRV and grass carp reovirus (GCRV) have been determined from cloned cDNAs, along with several genome segments of other aquareovirus isolates. Genomic dsRNA segments contain 7 nt at the 5′ terminus and 6 nt at the 3′ terminus, which are conserved in all 11 genome segments within a particular virus species (Table 3). The 5′- and 3′-conserved terminal sequences of isolates of Aquareovirus C are 5′-GUUAUUU/G-3′ and 5′-A/UUCAUC-3′, compared to 5′-GUUUUAU/G-3′ and 5′-A/UUCAUC-3′ in Aquareovirus A.
Table 3 Conserved terminal sequences (positive strand) of aquareovirus genome segments
| Virus species | Serotype or strain | 5′ end | 3′ end |
| Aquareovirus A | CHSRV | 5′-GUUUUAU/G | A/UUCAUC-3′ |
| Aquareovirus C | GSRV/GSRV | 5′-GUUAUUU/G | A/UUCAUC-3′ |
| Aquareovirus G | AGCRV | 5′-GUUUUAU/A | U/AU/AUCAUC-3′ |
Proteins
Virions of Aquareovirus A isolates contain seven structural proteins: VP1, 130 kDa; VP2, 127 kDa; VP3, 126 kDa; VP4, 73 kDa; VP5, 71 kDa; VP6, 46 kDa; VP7, 35 kDa. VP1, VP2, VP3 and VP6 form the core of the virus particle. VP3 and VP6 are more abundant than VP1 and VP2. VP1 is present in greater copy numbers than VP2. VP6 and VP3 probably form nodules and the spherical shell of the core, respectively. VP1 is thought to form turret-like structures present at the five-fold axis. VP2 is present in very small amounts per virion and is thought to be present beneath the five-fold axis.
VP7, VP4 and VP5 are present in the outer coat of the virion. All three proteins are removed by prolonged trypsinization, resulting in release of core particles. VP7 is the most external protein. VP5 is the next most accessible protein after VP7. Removal of VP7 by trypsin may expose some regions of VP5 critical for efficient entry into cells.
Lipids
Aquareoviruses have no known lipid components.
Carbohydrates
VP7 of Aquareovirus A isolates may be glycosylated.
Genome organization and replication
Twelve primary gene products have been identified for isolates of Aquareovirus A (Table 4). However, observed variations in dsRNA electropherotype suggest that viruses from different species may have proteins with significant differences in size. Each genome segment of Aquareovirus A isolates encodes only one primary translation product, with the exception of Seg11, which encodes two primary translation products. In addition to the seven structural proteins, five non-structural proteins of unknown function are encoded. In isolates of Aquareovirus C and Aquareovirus G, it is Seg7 that encodes two proteins, from non-overlapping and out-of-phase ORFs (Figure 7).
Table 4 Genome segments and protein products of striped bass reovirus (species Aquareovirus-A)
| Genome segment | Size (kbp) | Protein nomenclature | Protein size (kDa) | Protein location |
| Seg1 | 3.8 | VP1 | 130 | Inner capsid (core) |
| Seg2 | 3.6 | VP2 | 127 | Inner capsid (core) |
| Seg3 | 3.3 | VP3 | 126 | Inner capsid (core) |
| Seg4 | 2.5 | VP4 | 97 | Non-structural |
| Seg5 | 2.4 | VP5 | 71 | Inner capsid (core) |
| Seg6 | 2.2 | VP4 | 73 | Inner capsid (core) |
| Seg7 | 1.5 | NS4 | 28 | Non-structural |
| Seg8 | 1.4 | VP6 | 46 | Inner capsid (core) |
| Seg9 | 1.2 | NS2 | 39 | Non-structural |
| Seg10 | 0.9 | VP7 | 34 | Major outer capsid |
| Seg11 | 0.8 | NS3 | 29 | Non-structural |
|
|
| NS5 | 15 | Non-structural |
Antigenic properties
Aquareovirus outer CPs lack hemagglutinating activity. Viruses possess type-specific and group-specific antigenic determinants. Members within a single species may be antigenically related. Members of different species are antigenically distinct. Minor antigenic cross-reactivity has only been demonstrated between members of Aquareovirus A and Aquareovirus B. Distinct serotypes probably exist within each species. The major outer CP of isolates of Aquareovirus A (VP7) is not the major neutralizing antigen. There is no antigenic relationship between aquareoviruses and MRVs.
Biological properties
Host range
Aquareoviruses have been isolated from poikilothermic vertebrates as well as invertebrates (hosts include fish, molluscs, etc.) obtained from both fresh and sea water. The viruses replicate efficiently in fish and mammalian cell lines at temperatures ranging from 15 °C to 25 °C. They produce a characteristic cytopathic effect consisting of large syncytia. Generally, the viruses are of low pathogenicity in their host species. However, GCRV is highly pathogenic in grass carp. The infectivity of aquareoviruses is enhanced by treatment with trypsin or chymotrypsin, which correlates with digestion of the outer capsid protein VP7. The most infectious stage of the virus is produced by a 5-min treatment with trypsin. However, prolonged trypsin treatment almost completely abolishes infectivity, reflecting release of core particles.
Species demarcation criteria in the genus
In addition to the other general criteria used throughout the family, members of a species in the genus Aquareovirus may be identified by:
- Northern hybridization assays under conditions (stringency) that do not allow >17% mismatch. Only isolates within the same species will show hybridization.
- Sequence analysis: In genome segment 10 which encodes the major outer CP (VP7), viruses from different species have <55% nucleotide identity (36% amino acid identity in the VP7). In the RdRp isolates of the same species have >95% aa identity, while the corresponding values between species are 57–74%.
Seven species (Aquareovirus A to Aquareovirus G) and some unassigned viruses have been recognized on the basis of RNA-RNA hybridization.
List of species in the genus Aquareovirus
| Aquareovirus A |
|
|
| American oyster reovirus 13p2 |
| (13p2RV) |
| Angel fish reovirus |
| (AFRV) |
| Atlantic salmon reovirus HBR |
| (HBRV) |
| Atlantic salmon reovirus AS |
| (ASRV) |
| Atlantic salmon reovirus TS |
| (TSRV) |
| Chinook salmon reovirus DRC |
| (DRCRV) |
| Chum salmon reovirus CS | [Seg 1: AF418294, Seg 2: AF418295, Seg 3: AF418296, Seg 4: AF418297, Seg 5: AF418298, Seg 6: AF418299, Seg 7: AF418300, Seg 8: AF418301, Seg 9: AF418302, Seg 10: AF418303, Seg 11: AF418304 ] | (CSRV) |
| Masou salmon reovirus MS |
| (MSRV) |
| Smelt reovirus |
| (SRV) |
| Striped bass reovirus | [Seg 2: AF450318, Seg 3: AF450319, Seg 4: AF450320, Seg 8: AF450321, Seg 10: AF450322, U83396] | (SBRV) |
| Aquareovirus B |
|
|
| Chinook salmon reovirus B |
| (GRCV) |
| Chinook salmon reovirus LBS |
| (LBSV) |
| Chinook salmon reovirus YRC |
| (YRCV) |
| Chinook salmon reovirus ICR |
| (ICRV) |
| Coho salmon reovirus CSR | [Seg 10: U90430] | (CSRV) |
| Coho salmon reovirus ELC |
| (ELCV) |
| Coho salmon reovirus SCS |
| (SCSV) |
| Aquareovirus C |
|
|
| Golden shiner reovirus | [Seg 1: AF403398, Seg 2: AF403399, Seg 3: AF403400, Seg 4: AF403401, Seg 5: AF403402, Seg 6: AF403403, Seg 7: AF403404, Seg 8: AF403405, Seg 9: AF403406, Seg 10: AF403407, Seg 11: AF403408]
| (GSRV) |
| Grass carp reovirus | [Seg 1: AF260511, Seg 2: AF260512, Seg 3: AF260513, Seg 4: AF403390, Seg 5: AF403391, Seg 6: AF403392, AF239175, Seg 7: AF403393, Seg 8: AF403394, Seg 9: AF403395, Seg 10: AF403396, Seg 11: AF403397] | (GCRV) |
| Aquareovirus D |
|
|
| Channel catfish reovirus |
| (CCRV) |
| Aquareovirus E |
|
|
| Turbot reovirus |
| (TRV) |
| Aquareovirus F |
|
|
| Chum salmon reovirus PSR |
| (PSRV) |
| Coho salmon reovirus SSR |
| (SSRV) |
| Aquareovirus G |
|
|
| American grass carp reovirus | [EF589098-108] | (AGCRV) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ] and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Aquareovirus but have not been approved as species
| Chub reovirus |
| (CHRV) |
| Golden ide reovirus | [Seg2: AF450323, Seg5: AF450324] | (GIRV) |
| Hard clam reovirus |
| (HCRV) |
| Landlocked salmon reovirus |
| (LSRV) |
| Tench reovirus |
| (TNRV) |
Relationship with orthoreoviruses
The highest level of amino acid sequence identity detected between the RdRp of aquareoviruses and a member of a distinct reovirus genus was 41% (to MRV, a member of the genus Orthoreovirus), supporting the hypothesis that these genera are closely related (derived from a common ancestor, estimated ca. 510 million years ago [MYA]). Although this value of amino acid sequence identity is higher than that separating most genera (usually <30%), classification of the aquareoviruses and orthoreoviruses as members of two distinct genera is based on multiple parameters and not simply genetic relatedness. For example, the aquareoviruses can infect many marine and freshwater species, whereas the orthoreoviruses primarily infect mammals, birds and reptiles. The common origin of these viruses, and of their respective hosts, suggests co-speciation of the viruses with their respective hosts.
Genus Oryzavirus
Type species Rice ragged stunt virus
Virion properties
Morphology
Intact rice ragged stunt virus (RRSV) particles appear to be icosahedral in symmetry and double-shelled. The particle diameter is in the range of 75–80 nm and surface A-spikes (approximately 10–12 nm wide and 8 nm in length) are attached to the end of B-spikes situated at the five-fold axes of the viral core. The subviral or core particles have an estimated diameter of 57–65 nm (Figure 8) and possess 12 B-type spikes, 8–10 nm in height, 23–26 nm wide at the base and 14–17 nm wide at the top. In negatively-stained preparations of RRSV, B-spiked subviral particles have been seen but intact double-shelled particles are not seen without pretreatment with fixative. Echinochloa ragged stunt virus (ERSV) particles are slightly larger than RRSV particles.
Physicochemical and physical properties
RRSV particles sediment as a single component and are stable at pH 6.0–9.0. They are stable in 0.1 M MgCl2. The B spikes dissociate from the core particle in 0.5 M MgCl2 and the entire particle is disrupted in 2 M MgCl2. The particles retain infectivity after 7 days at 4 °C and after 10 min at 50 °C but lose their infectivity after 10 min at 60 °C. They retain infectivity after three cycles of freezing and thawing. The particles contain an RdRp.
Nucleic acid
The oryzavirus genome consists of 10 linear dsRNA segments. The genomes of RRSV and ERSV have similar sizes and segment profiles (RRSV Mr 18.15×106 (26,066 bp); ERSV Mr 17.78×106), with segments ranging in size from 1,162 to 3,849 bp. The genomic dsRNAs are termed Seg1 to Seg10, in order of increasing electrophoretic mobility in 7.5% polyacrylamide gels. The entire genome of RRSV has been sequenced; Seg4 and Seg10 are larger than they appear from migration in polyacrylamide gels, suggesting that they may migrate in the position 3 and 9 respectively during agarose gel electrophoresis (AGE). The conserved terminal sequences of the ERSV genome segments are identical to those of RRSV (5′-GAUAAA…(G)GUGC-3′) and differ from those of phytoreoviruses or fijiviruses. RRSV RNAs hybridize weakly with their counterparts in ERSV but not with segments of the phytoreovirus rice dwarf virus (RDV).
Proteins
RRSV particles are composed of five major, highly immunoreactive structural proteins, with estimated sizes of 33, 39, 43, 70 and 120 kDa, and at least five minor structural proteins (49, 60, 76, 90 and 94 kDa). Three more proteins (31, 63 and 88 kDa) have also been identified by in vitro translation of RRSV genomic dsRNA, and designated as non-structural proteins. RRSV S5, S8 and S9, respectively, encode a 90 kDa minor structural protein (possibly a guanylyltransferase), a 67 kDa major structural protein, which is further self-processed to 46, 43 and 26 kDa proteins, and a 38 kDa major structural protein. P9 is thought to be involved in vector transmission. RRSV segments S7 and S10 encode non-structural proteins of about 68 and 32 kDa, respectively. RRSV S4 probably encodes an RdRp and a second protein of unknown function. ERSV particles have four major structural proteins (127, 123, 63 and 34 kDa) and three minor proteins (103, 50 and 49 kDa). The reported differences in morphology of the outer capsids of RRSV and ERSV could be at least partially due to differences in the sizes of these structural proteins.
Lipids
None reported.
Carbohydrates
There is no evidence for the glycosylation of oryzavirus proteins.
Genome organization and replication
The genome organization is well characterized only for RRSV (Table 5). The dsRNA genome segments contain a single large ORFs (in one strand of the pair) except S4, which contains two large ORFs. The proteins encoded by S3, S8 and S9 are major components of the RRSV particle, but those encoded by segments S7 and S10 are not found in the virion. Seg8 codes for a polyprotein that appears to autocatalytically cleave into at least two polypeptides one of which is a major structural protein. The larger protein encoded by Seg4 appears to be an RdRp. The tentative functions of the proteins encoded by the other segments are shown in Table 5. The viruses induce viroplasms in the cytoplasm of infected cells.
Table 5 Genome segments and protein products of rice ragged stunt virus
| Genome segment | Size (bp) | Protein nomenclature | Protein Mr predicted (kDa) | Protein Mr apparent (kDa) | Function (location) |
| Seg1 | 3849 | P1 | 137.7 | 137 | Virus core associated (B Spike) |
| Seg2 | 3810 | P2 | 133.1 | 118 | (Inner core capsid) |
| Seg3 | 3699 | P3 | 130.8 | 130 | (Major core capsid) |
| Seg4 | 3823 | P4A (Pol) | 141.4 | 145 | RDR polymerase |
|
|
| P4B | 36.9 |
| (Unknown) |
| Seg5 | 2682 | P5 (Cap) | 91.4 | 90 | Capping enzyme/guanyltransferase |
| Seg6 | 2157 | P6 | 65.6 |
|
|
| Seg7 | 1938 | NS7 | 68 | 66 | (Nonstructural) |
| Seg8 | 1814 | P8 | 67.3 | 67 | Precursor Protease (major capsid) |
| P8A/ P8B | 25.6/41.7 | 47/44 |
| ||
| Seg9 | 1132 | P9 | 38.6 | 37 | Vector transmission (spike) |
| Seg10 | 1162 | NS10 | 32.3 | 32 | Non-structural |
Antigenic properties
RRSV and ERSV cross-react in serological tests. Polyclonal antisera raised against RRSV particle preparations react most strongly with P3, P8 and P9 (both the native state and the state resulting from in vitro production), suggesting that they are highly immunogenic. P5 is weakly immunogenic. Glutathione-S-transferase-NSP7 fusion protein is highly immunogenic, and antibodies against this protein are useful in ELISA for the detection of RRSV in infected plants and insects.
Biological properties
Oryzaviruses infect plants in the family Gramineae, causing diseases in rice (RRSV) and species of Echinochloa (barnyard grasses and millets; ERSV). They are transmitted by, and replicate in, phloem-feeding, viruliferous delphacid planthoppers (RRSV: Nilaparvata lugens; ERSV: Sogatella longifurcifera and S. vibix). RRSV is ingested when the hopper feeds on rice plants, usually at the seedling stage. The minimum acquisition access period for the vector is about 3 h, the latent period is about 9 days, and the minimum inoculation access time is about 1 h. Planthopper nymphs are more efficient vectors than adults, but all forms of the insect can act as vectors. Any individual viruliferous hopper gives intermittent transmission. The virus is not passed though the egg.
Oryzaviruses appear to replicate in fibrillar viroplasms within the cytoplasm of phloem, or phloem-associated, plant cells and in cells of the salivary glands, fat body, gut and brain of the planthopper. The phloem cells proliferate to form galls on the plant. RRSV has been reported in southeastern and far-eastern Asian countries, where it affects rice yields (generally 10–20% loss, but up to 100% in severely affected areas). ERSV has been reported in Taiwan.
Species demarcation criteria in the genus
In addition to the general criteria used throughout the family, species in the genus Oryzavirus differ in vector (planthopper) and host plant species.
List of species in the genus Oryzavirus
| Echinochloa ragged stunt virus |
|
|
| {Sogatella longifurcifera, S. vibix} |
|
|
| {Graminae: Echinochloa} |
|
|
| Echinochloa ragged stunt virus |
| (ERSV) |
| Rice ragged stunt virus |
|
|
| {Nilaparvata lugens} |
|
|
| {Graminae: Rice} |
|
|
| Rice ragged stunt virus - Thailand | [Seg1: AF020334; Seg2: AF020335; Seg3:AF020336; Seg4: U66714; Seg5: U33633; Seg6: AF020337, Seg7: U66713; Seg8: U46682; Seg9: L38899; Seg10: U66712] | (RRSV-Tai) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ], insect vector and host names { } and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Oryzavirus but have not been approved as species
None reported.
Phylogenetic relationships within the genus
No information.
Genus Fijivirus
Type species Fiji disease virus
Distinguishing features
Fijivirus particles have a double-shelled, icosahedral structure, with a spherical rather than angular appearance and short surface spikes (A spikes) on each of the 12 vertices of the icosahedron. The outer shell is fragile and easily breaks down, leaving the inner shell bearing 12 B spikes. There are 10 genome segments. The viruses replicate in delphacid planthoppers. Nilaparvata lugens reovirus (NLRV) has the above properties but replicates only in insects, whereas other fijiviruses can also replicate in phloem cells of susceptible plants of the families Gramineae (in which they induce small tumors or enations), or Liliaceae.
Virion properties
Morphology
Virions are double-shelled, spherical, 65–70 nm in diameter with A spikes of about 11 nm in length and breadth, at the 12 vertices on the icosahedra (Figure 9, left). Unless pre-fixed, viruses readily break down in vitro to give cores, about 55 nm in diameter, with 12 B spikes, about 8 nm long and 12 nm in diameter (Figure 9, right). Some treatments (shaking with butan-1-ol or incubation with 1.9 M MgCl2) produce smooth subcores (Figure 9, center).
Physicochemical and physical properties
The physicochemical properties of the virions have not been determined.
Nucleic acid
Fijiviruses have 10 dsRNA segments that are numbered in order of increasing electrophoretic mobility during PAGE. Some segments do not migrate in order of their Mr and may migrate in a reverse order during (1%) agarose gel electrophoresis (AGE). Examples include segments 2 and 3 of NLRV and Mal de Rio Cuarto virus (MRCV) and segments 8 and 9 of oat sterile dwarf virus (OSDV). The conserved terminal sequences are shown in Table 6. Within the genus, only the 3′-terminal sequence …GUC-3′ is conserved. Adjacent to the conserved terminal oligonucleotide sequences, each genome segment possesses inverted repeats, which are several bases long, similar to those in phytoreovirus and oryzavirus RNAs, although the sequences involved differ in these other genera. Characteristic of the genus is the low G+C content of the genomic RNAs, mostly around 34–36%. The sizes and groupings of the 10 dsRNA species are characteristic and distinctive for the five groups of fijiviruses that are recognized.
Table 6 Conserved terminal sequences (positive strand) of Fijivirus genome segments
| Virus species | Strain | 5′ end | 3′ end |
| Fiji disease virus | FDV | 5′-AAGUUUUU | CAGCNNNNGUC-3′ |
| Maize rough dwarf virus | MRDV | 5′-AAGUUUUUU | UGUC-3′ |
| Mal de Rio Cuarto virus | MRCV | 5′-AAGUUUUU | CAGCUNNNGUC-3′ |
| Nilaparvata lugens reovirus | NLRV | 5′-AGU | GUUGUC-3′ |
| Oat sterile dwarf virus | OSDV | 5′-AACGAAAAAAA | UUUUUUUUAGUC-3′ |
| Rice black streaked dwarf virus | RBSDV | 5′-AAGUUUUU | AGCUNN(C/U)GUC-3′ |
| (not classified) | SRBSDV | 5′-AAGUUUUU | CAGCUGAUGUC-3′ |
Proteins
Six polypeptides, numbered respectively I to VI (139, 126, 123, 111, 97 and 64 kDa), can be detected by SDS PAGE of purified MRDV. The B-spiked cores contain peptides I, II and III, while the smooth core contains peptides I and II. The B spikes should therefore be composed of peptide III. Peptides IV–VI form the outer capsid. During infection of most (possibly all) fijiviruses, tubules about 90 nm in diameter accumulate in the cytoplasm. Sometimes these are incompletely closed and form scrolls. They are presumably composed of a non-structural protein whose function and genome segment assignment are unknown.
Three major proteins (130, 120 and 56 kDa) and three minor ones (148, 65 and 51 kDa) can be detected by SDS PAGE of purified virions of Rice black streaked dwarf virus (RBSDV). The 120 kDa protein is the B spike protein. Smooth subcore particles consist of 148, 130 and 65 kDa proteins. The 56 kDa protein is the major component of the outer capsid shell and the 51 kDa protein is a partial degradation of it. In NLRV virions, three major proteins (140, 135 and 65 kDa), three intermediate (160, 110 and 75 kDa), and one minor protein (120 kDa) can be resolved. The 135 kDa protein is the B spike. The 65 kDa protein is the major component of the outer capsid shell and the 140 kDa protein is the major core protein. In addition to the above structural proteins, there is an A spike but its protein has not yet been identified.
The Fiji disease virus (FDV) VP9a equivalent in MRCV is designated P9-1. This protein was shown to establish cytoplasmic inclusion bodies resembling viral inclusion bodies, after transfection of Spodoptera frugiperda insect cells. P9-1 self-associates, giving rise to high molecular weight complexes when expressed in bacteria. P9-1 binds ssRNA and possesses an ATPase activity.
Lipids
Not known.
Carbohydrates
Not known.
Genome organization and replication
Genome organizations and coding assignments of fijiviruses are summarized in Table 7. Most of the genome segments are monocistronic. Some segments possess two ORFs but expression of the second ORF has not been demonstrated in vivo in insect or plant cells. For viruses other than NLRV, replication occurs in the cytoplasm of phloem-related cells in association with viroplasms composed partly of fine filaments. NLRV does not have a counterpart to the ORF2 present in FDV Seg7 (and the corresponding segments of other plant-infecting fijiviruses), and this may reflect its inability to replicate in plant hosts.
Table 7 Genome segments and protein products of Fiji disease virus. The equivalent segment numbers for other members of the genus Fijivirus are also shown
| Genome segment | Size (bp) | Protein Mr predicted (kDa) | Location (function)* | Homologous segment in other members of the genus | ||||
| MRCV | MRDV | OSDV | RBSDV | NLRV | ||||
| Seg1 | 4532 | 170.6 | Core (RNA polymerase) | 1 | na | na | 1 | 1 |
| Seg2 | 3820 | 137.0 | Major core | 3 | na | na | 2 | 3 |
| Seg3 | 3623 | 135.5 | Outer shell (possible B spike) | 2 | na | na | 4 | 2 |
| Seg4 | 3568 | 133.2 | Unknown | 4 | na | na | 3 | 4 |
| Seg5 | 3150 | 115.3 | Unknown | 5 | na | na | 5 |
|
| Seg6 | 2831 | 96.8 | Unknown | 6 | na | na | 6 |
|
| Seg7 | 2194 | 41.7 | Non-structural (possible tubule protein) | 7 | 6 | 7 | 7 | 10** |
| 36.7 | Unknown |
|
|
|
|
| ||
| Seg8 | 1959 | 68.9 | Core protein (possible NTP-binding) | 8 | 7 | 9 | 8 | 7 |
| Seg9 | 1843 | 38.6 | Viroplasm | 9 | 8 | 10 | 9 |
|
| 23.8 | Non-structural protein? |
|
|
|
|
| ||
| Seg10 | 1819 | 63.0 | Major outer capsid | 10 | 10 | 8 | 10 | 8 |
* The probable function of some of the proteins has been deduced from the equivalent genome segment of other virus species.
** Genome Seg10 of NLRV does not contain a second ORF.
Antigenic properties
Some proteins of the viruses in group 2 (MRCV, MRDV, Pangola stunt virus (PaSV) and RBSDV) are distantly related but homologous proteins from viruses of other species in the genus are serologically unrelated.
Biological properties
All the plant-infecting fijiviruses induce hypertrophy of the phloem (both expansion and multiplication of cells), leading to vein swellings and sometimes galls (enations or tumors) derived from phloem cells, especially on the backs of leaves. MRDV in maize induces longitudinal splitting of the roots. Other effects include the suppression of flowering, plant stunting, increased production of side shoots, and induction of a dark green coloring. In insect hosts, no particular tissue tropism or severe disease is recognized. Viruses are transmitted propagatively by delphacid planthoppers (Hemiptera, Delphacidae, e.g. Perkinsiella, Laodelphax, Toya, Sogatella, Javesella, Ribautodelphax, Dicranotropis, Delphacodes, Sogatella and Unkanodes). Following virus acquisition from infected plants, the latent period is about two weeks, and leads to a lifelong capacity for virus transmission to plants. No transovarial or seed transmission of virus has been identified. Mechanical transmission from plant to plant can be demonstrated only with difficulty. Virus is spread by offsets in vegetatively propagated crops (e.g. pangolagrass and sugarcane). Viruses can overwinter in diapausing planthoppers, in certain weed species and in autumn-sown cereals.
Generally, fijiviruses are widespread in nature, although they are apparently absent from North America and have not been reported from Africa or confirmed from India. FDV has been reported from Australia and the Pacific islands. RBSDV occurs in Japan, Korea and China. PaSV occurs in northern countries of South America, Oceania, Taiwan and northern Australia, and OSDV occurs in northern Europe. Garlic dwarf virus (GDV) has been found only in southern France. MRDV is found in Scandinavia and in areas bordering the northern and eastern Mediterranean. MRCV occurs in Argentina.
NLRV was found in the planthopper Nilaparvata lugens, which occurs in south-east Asia. Experimentally it infects a second hopper, Laodelphax striatellus. There is no evidence that NLRV can multiply in rice plants, a natural host of N. lugens, but the virus is transmitted from hopper to hopper through contaminated rice plants and moves through the phloem and/or xylem of rice plants once injected by the viruliferous hoppers.
Species demarcation criteria in the genus
Of the seven fijivirus species, the four members of group 2 are relatively closely related to one another. Further information about these viruses may eventually necessitate a revision of their species status. In particular, MRDV and RBSDV may be considered sufficiently closely related to constitute a single species.
The conserved terminal sequences of genome segments do not differ greatly between fijivirus species (Table 6). In addition to the other general criteria used throughout the family, members of a species in the genus Fijivirus may be identified by:
- Sequence analysis: members of different species usually have <40% amino acid identity in comparisons of proteins corresponding to those encoded by RBSDV segments 7, 8, 9 and 10). In comparisons among the genome segments coding for the major capsid protein, viruses from different groups have <55% nucleotide identity (but identities are much higher within group 2).
- Cross-hybridization of less conserved genome segments (Segs-10 of MRDV and RBSDV, encoding a highly conserved major outer shell protein, share 94% nucleotide sequence and so are not suitable). Hybridization using RBSDV Seg5 and Seg6 cDNA probes to detect the homologous sequences is more than 20 times more sensitive than hybridization using their counterparts from MRDV.
- Serological cross-reactions: viruses in different groups do not cross-react; those in group 2 do so to a limited extent that is dependent on the proteins being compared.
- The identity or family of the plant host species (if any) together with the insect vector and its host.
List of species in the genus Fijivirus
| Fijivirus group 1 |
|
|
| Fiji disease virus |
|
|
| Fiji disease virus {Perkinsiella saccharicida, P. vastatrix, P. vitiensis: Graminae} | [Seg1: AY029520, Seg2: AF049704, Seg3: AF359556, Seg4: AF049705, Seg5: AY029521, Seg6: AF356083, Seg8: AY297693, Seg9: AF050086, Seg10: AY297694] | (FDV) |
| Fijivirus group 2 |
|
|
| Rice black streaked dwarf virus |
|
|
| Rice black streaked dwarf virus - ZJ {Laodelphax striatellus, Ribautodelphax albifascia, Unkanodes sapporona: Graminae} | [Seg1: AJ294757, Seg2 AJ409145, Seg3: AJ293984, Seg4: AJ409146, Seg5: AJ409147, Seg6: AJ409148, Seg7: AJ297427, Seg8: AJ297431, Seg9: AJ297430, Seg10: AJ297433] | (RBSDV-ZJ) |
| Maize rough dwarf virus |
|
|
| Maize rough dwarf virus - Italy {Ribautodelphax notabilis: Graminae} | [Seg6: X55701, Seg7: L76562, Seg8: L76561, Seg10: L76560] | (MRDV -IT) |
| Mal de Rio Cuarto virus |
|
|
| Mal de Rio Cuarto virus - Argentina {Delphacodes kuscheli: Graminae} | [Seg1: AF499925, Seg2: AF499926, Seg3: AF499928, Seg4: AF395873, Seg6: AF499927, Seg 7: AY923115, Seg8: AF395872, Seg9: DQ023312, Seg10: AY607586] | (MRCV-ARG) |
| Pangola stunt virus |
|
|
| Pangola stunt virus {Sogatella furcifera S. kolophon: Graminae} |
| (PaSV) |
| Fijivirus group 3 |
|
|
| Oat sterile dwarf virus |
|
|
| Oat sterile dwarf virus {Javesella pellucida, J. discolor, J. dubia, J. obscurella, Dicranotropis hamata: Graminae} | [Seg7: AB011024, Seg8: AB011025, Seg9: AB011026, Seg10: AB011027] | (OSDV) |
| Fijivirus group 4 |
|
|
| Garlic dwarf virus |
|
|
| Garlic dwarf virus {unknown: Liliaceae} |
| (GDV) |
| Fijivirus group 5 |
|
|
| Nilaparvata lugens reovirus |
|
|
| Nilaparvata lugens reovirus {Nilaparvata lugens, Laodelphax striatellus: no plant hosts} | [Seg1: D49693, Seg2: D49694, Seg3: D49695, Seg4: D49696, Seg5: D49697, Seg6: D49698, Seg7: D49699, Seg8: D26127, Seg9: D49700, Seg10: D14691] | (NLRV) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ], insect vector and host names { }
and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Fijivirus but have not been approved as species
| Southern rice black streaked dwarf virus | [Seg1: FN563983, Seg2: FN563984, Seg3: FN563985, Seg4: FN563986, Seg5: FN563987, Seg6: FN563988, Seg7: EU784841, Seg8: EU784842, Seg9: EU784843, Seg10: EU784840] | (SRBSDV) |
Phylogenetic relationships within the genus
Phylogenetic relationships within the genus are shown in Figure 10.
Genus Mycoreovirus
Type species Mycoreovirus 1
Distinguishing features
Virions have a relatively featureless outer capsid as viewed by negative staining and electron microscopy, whereas the core particles have 12 icosahedrally arranged surface turrets or spikes. The genome is composed of 11 or 12 segments of dsRNA. The members of the genus that have been described all infect fungi.
Virion properties
Morphology
Particles are non-enveloped. Electron microscopy and negative staining of mycoreovirus virions with aqueous uranyl acetate indicates that they are double shelled, spherical in appearance (icosahedral symmetry) and approximately 80 nm in diameter. The viral core (estimated as 50 nm in diameter) has 12 icosahedrally arranged surface projections (turrets or B-spikes) (Figure 11). Particles are disrupted by 2% phosphotungstic acid (pH 7.0).
Physicochemical and physical properties
Not determined.
Nucleic acid
The genome consists of 11 (group 1) or 12 (group 2) dsRNA segments that are numbered in order of reducing molecular weight or increasing electrophoretic mobility following agarose gel electrophoresis. The total genome of Cryphonectria parasitica mycoreovirus-1 (CpMRV-1) contains 23,436 bp, with the length of individual segments ranging from 732 bp to 4127 bp, showing a 3-3-2-3 electrophoretic profile following either 11% PAGE or 1% agarose gel electrophoresis (AGE). In contrast, the genomic RNA of Rosellinia necatrix mycoreovirus-3 (RnMRV-3) shows a 3-3-6 electrophoretic profile following 5% PAGE. As with other members of the family Reoviridae, the genome segment migration patterns during AGE (or low percentage (<5%) PAGE) are considered likely to be characteristic of each virus species. Terminal sequences of the genome segments are shown in Table 8.
Table 8 Conserved terminal sequences (positive strand) of mycoreovirus genome segments
| Virus species | Serotype or strain | 5′ end | 3′ end |
| Mycoreovirus 1 | CpMYRV-1-9B21 | 5′-GAUCA | CGCAGUCA-3′ |
| Mycoreovirus 3 | RnMYRV-3-W370 | 5′-ACAAUUU | UGCAGAC-3′ |
Proteins
Protein sequences of Cryphonectria parasitica mycoreovirus-1 (CpMYRV-1) were deduced from sequences of the viral genomic RNAs. Their putative functions are shown in Table 9. Proteins of the 11 and 12 segmented mycoreoviruses are currently named as VP1 to VP11 or VP12, based on the molecular weight of the genome segment (segment number) from which they are translated. Proteins of CpMYRV-1 were expressed in baculovirus, and the capping enzyme was identified by an autoguanylation assay as VP3 (encoded by Seg3). A series of progressive N-terminal and C-terminal deletion mutants were also made to localize the auto-guanylation active-site of VP3 to amino acid residues 133–667. Within this region, a sequence was identified (residues 170–250) that has relatively high sequence similarity to homologues in the two other mycoreoviruses, CpMYRV-2 and Rosellinia necatrix mycoreovirus-3 (RnMYRV-3), as well as two coltiviruses, Colorado tick fever virus and Eyach virus. Site-directed mutagenesis of conserved residues revealed that H233, H242, Y243, F244 and F246, but not K172 or K202, play critical roles in guanylyltransferase activities.
Table 9 Genome segments and protein products of Cryphonectria parasitica mycoreovirus-1
| Genome segment | Size (bp) | Protein nomenclature | Protein size aa (kDa) | Structure/function |
| Seg1 | 4127 | VP1 | 1354 (151.8) | RdRp (Pol); sequence similarity to coltivirus VP1 |
| Seg2 | 3846 | VP2 | 1238 (138.5) | Sequence similarity to coltivirus VP2 |
| Seg3 | 3251 | VP3 | 1065 (120.8) | Guanylyltransferase (CaP) |
| Seg4 | 2269 | VP4 | 721 (79.8) | Sequence similarity with RnMYRV-1-W370 Seg4 and coltivirus Seg4 |
| Seg5 | 2023 | VP5 | 648 (72.8) |
|
| Seg6 | 2056 | VP6 | 650 (73.4) | Sequence similarity with RnMYRV-1-W370 Seg6 and coltivirus Seg10 |
| Seg7 | 1536 | VP7 | 482 (54.1 |
|
| Seg8 | 1539 | VP8 | 470 (51.2 |
|
| Seg9 | 1072 | VP9 | 298 (32.9) | Sequence similarity with RnMYRV-1-W370 Seg11 |
| Seg10 | 975 | VP10 | 248 (27.8 |
|
| Seg11 | 732 | VP11 | 102 (11.5) |
|
Table 10 Genome segments and protein products of Rosellinia necatrix mycoreovirus-3
| Genome segment | Size (bp) | Protein nomenclature | Protein size aa (kDa) | Structure/function |
| Seg1 | 4143 | VP1 | 1360 (153.4) |
|
| Seg2 | 3773 | VP2 | 1226 (138.5) | Shows some sequence similarity to coltivirus VP2 |
| Seg3 | 3310 | VP3 | 1086 (121.9) |
|
| Seg4 | 2259 | VP4 | 725 (78.7) |
|
| Seg5 | 2089 | VP5 | 646 (72.3) |
|
| Seg6 | 2030 | VP6 | 634 (71.5) |
|
| Seg7 | 1509 | VP7 | 482 (55.1) |
|
| Seg8 | 1299 | VP8 | 325 (36.5) |
|
| Seg9 | 1226 | VP9 | 380 (41.6) |
|
| Seg10 | 1171 | VP10 | 310 (33.6) |
|
| Seg11 | 1003 | VP11 | 282 (31.1) |
|
| Seg12 | 943 | VP12 | 265 (29.2) |
|
Lipids
None reported.
Carbohydrates
None reported.
Genome organization and replication
The sizes and predicted ORFs for the 11 segments of CpMYRV-1 and the 12 segments of RnMYRV-3 are shown in Tables 9 and 10, respectively. On the basis of the available sequence data for several of the genome segments and the overall similarity of mycoreoviruses to other members of the family Reoviridae, it is assumed that many aspects of the genome organization and replication are also similar. On this basis it is likely that the viral core contains transcriptase complexes that synthesize mRNA copies of the individual genome segments, which are exported and translated to produce viral proteins within the host cytoplasm. These positive sense RNAs also are likely to form templates for negative strand synthesis during progeny virus assembly and maturation. As with other reoviruses, most of the mycoreovirus genome segments appear to represent single genes, with a large ORF and relatively short terminal non-coding regions.
Antigenic properties
Not available.
Biological properties
RnMYRV-1 is found in the mycelium of a strain of the white root rot fungus Rosellinia necatrix. The virus itself appears to make the fungus hypovirulent and may represent a useful biological control for the damage caused by the wild-type fungus. The uninfected fungus can be regenerated by hyphal tip culture. CpMYRV-1 and CpMYRV-2 are found in the mycelium of the filamentous fungus that causes chestnut blight disease (Cryphonectria parasitica). Purified particles of CpMYRV-1 can be used to infect protoplasts of virus-free mycelium. Infection with CpMYRV-1 greatly reduces virulence of the fungal strain and may represent a useful biological control for the disease.
Species demarcation criteria in the genus
See the general criteria used throughout the family.
List of species in the genus Mycoreovirus
| Group 1 (11 genome segments) |
|
|
| Mycoreovirus 1 |
|
|
| Cryphonectria parasitica mycoreovirus-1 (9B21) | [Seg1: AY277888, Seg2: AY277889, Seg3: AY277890, Seg4:AB179636, Seg5: AB179637, Seg6: AB179638, Seg7: AB179639, Seg8: AB179640, Seg9: AB179641, Seg10: AB179642, See11: AB179643] | (CpMYRV-1 / 9B21) |
| Mycoreovirus 2 |
|
|
| Cryphonectria parasitica mycoreovirus-2 (C18) | [Seg3: DQ902580] | (CpMYRV-2 / C18) |
| Group 2 (12 genome segments) |
|
|
| Mycoreovirus 3 |
|
|
| Rosellinia necatrix mycoreovirus-3 (W370) | [Seg1: AB102674, Seg2: AB098022, Seg5: AB098023, Seg6: AB073277, Seg7: AB073278, Seg8: AB073279, Seg9: AB073280, Seg10: AB073281, Seg11: AB073282, Seg12: AB073283] | (RnMYRV-3 / W370) |
| (Rosellinia anti-rot virus) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ] and assigned abbreviations ( ) are also listed
List of other related viruses which may be members of the genus Mycoreovirus but have not been approved as species
None reported.
Phylogenetic relationships within the genus and with other genera of family Reoviridae
Both C. parasitica mycoreoviruses (CpMYRV-1 and CpMYRV-2) contain 11 genome segments (Group 1), but only have approximately 29% amino acid sequence identity in the capping enzyme, confirming their identity as distinct species.
There are also some clear indications of homology in the larger genome segments of the Group 1 mycoreoviruses to RnMYRV-3 (Group 2, 12 segment genome). The highest amino acid sequence identity was detected in the polymerase sequence (39% identity).
Previous phylogenetic analyses, using the RdRp amino acid sequence (see section on the family Reoviridae), have shown that, in most cases, members of different reovirus genera exhibit amino acid sequence identities of less than 30% although aquareoviruses and orthoreoviruses can show up to 42% aa identity.
CpMYRV-1 shows about 29% amino acid sequence identity with the polymerase of members of the genus Coltivirus.
Genus Cypovirus
Type species Cypovirus 1
Distinguishing features
Cypovirus particles may be singly or multiply occluded by a virus-coded polyhedrin protein, which forms polyhedra within the cytoplasm of infected cells. Cypoviruses only infect, and are pathogenic for, arthropods. Virions have a single capsid shell with surface spikes, and have transcriptase and capping enzymes that are active without particle modification. They can retain RNA polymerase activity despite particle disruption into 10 distinct RNA protein complexes, each representing a single genome segment and a transcriptase complex. Consequently, transcriptase activity is resistant to repeated freeze–thawing, which disrupts the particle structure. The transcriptase activity may show very pronounced dependence on the presence of S-adenosyl-L-methionine or related compounds, although this dependence may be reduced by repeated freeze–thawing.
Virion properties
Morphology
Virus particles have a single-layered capsid, composed of a central capsid shell of 57 nm diameter, which extends to 71.5 nm (determined by cryoEM) when the 12 surface spikes or turrets that are situated on the icosahedral five-fold vertices are included. These surface projections are hollow and have previously been estimated to be up to 20 nm in length and 15–23 nm wide (by conventional microscopy and negative staining). They also appear to have a section near the tip that can be lost or removed. The virus particle has a central compartment about 35 nm in diameter. Cypovirus virions are structurally comparable to the core particles of members of other genera within the family Reoviridae, particularly genera containing viruses spiked cores (Orthoreovirus, Aquareovirus, Idnoreovirus and Oryzavirus) (Figures 12 and 13). The virus particles contain three major structural proteins that have been identified as: the capsid shell protein (CSP), 120 copies per particle, equivalent to the VP3(T2) protein of bluetongue virus, and orthoreovirus lambda 1; large protrusion protein (LPP, 120 copies, comparable to orthoreovirus lambda 3) and turret protein (TP, 60 copies, comparable to orthoreovirus lambda 2). The virion also contains transcriptase enzyme complexes attached to the inner surface of the capsid shell at the icosahedral five-fold vertices.
Cypovirus particles may be occluded by a crystalline matrix of polyhedrin protein, forming a polyhedral inclusion body. These polyhedra have a symmetry (e.g. cubic, icosahedral or irregular) that is influenced by both the virus strain (polyhedrin sequence) and the host. The polyhedrin protein appears to be arranged as a face-centered cubic lattice with center-to-center spacing varying between 4.1 and 7.4 nm.
Physicochemical and physical properties
The virion Mr is about 5.4×107. The buoyant density in CsCl is 1.44 g cm−3 for virions, approximately 1.30 g cm−3 for empty particles, and 1.28 g cm−3 for polyhedra. The S20,W is approximately 420S for virions and 260S for empty particles. Polyhedra vary considerably in size and Mr and do not have a single characteristic S value. Polyhedra may occlude many virus particles or only single particles depending on the virus strain. Large empty polyhedra (apparently containing no virions) have also been observed.
Cypoviruses retain infectivity for several weeks at −15 °C, 5 °C or 25 °C. The virus retains full enzymatic activity (dsRNA-dependent ssRNA polymerase and capping activity) after repeated freeze–thawing (up to 60 cycles). However, it appears likely that this results in the breakdown of the virus particle into ten active and distinct enzyme/template complexes. Each complex contains one genome segment and a complete transcriptase complex, derived from the virion capsid and including one of the spike structures from the vertices of the icosahedron. Polymerase activity is therefore a poor indicator of virion integrity. Within the family Reoviridae, the ability to retain enzyme function despite particle breakdown may be unique to the cypoviruses.
Cations have relatively little effect on the virus structure. Heat treatment of virions at 60 °C for 1 h leads to degradation and release of genomic RNA. Virus particles are relatively resistant to treatment with trypsin, chymotrypsin, ribonuclease A, deoxyribonuclease or phospholipase. Virion enzyme functions also show some resistance to treatment with proteinase K. However, this may reflect the retention of enzyme activities despite particle disruption, particularly during the early stages of digestion. Cypovirus particles are resistant to detergents such as sodium deoxycholate (0.5–1%) but are disrupted by 0.5–1% SDS, which releases the genomic dsRNA. Treatment with Triton X-100, NP40 or urea also causes disruption of the virus particle structure. One or two fluorocarbon treatments have little effect on virus infectivity, and treatment with ethanol leads to release of RNA from virions. Viruses and polyhedra are readily inactivated by UV-irradiation. It has been reported that UV also releases the dsRNA template from individual genome segment/transcriptase complexes. Polyhedra remain infectious for years at temperatures below 20 °C. Virions can be released from polyhedra by treatment with carbonate buffer at pH >10.5 but are disrupted at pH <5. High pH treatment completely dissolves the polyhedral protein matrix, as in the mid-guts of permissive insects. This process is partly due to increased solubility of polyhedrin at high pH but is also aided by alkaline-activated proteases associated with polyhedra.
Nucleic acid
Polyhedra (but not virions) contain significant amounts of adenylate-rich oligonucleotides. In the majority of cases, cypovirus particles contain ten linear dsRNA genome segments. However there is evidence to indicate that in some cases the virus particles may also contain an eleventh small segment (e.g. Trichoplusia ni cypovirus 15, TnCPV-15). In Bombyx mori cypovirus 1 (BmCPV-1), the genome segments vary in size from 4190 to 944 bp with a total genome size of 24,809 bp. In other isolates that have not yet been sequenced, the sizes of the genome segments have been estimated by electrophoretic comparisons and have calculated Mrs that vary from 0.42×106 to 3.7×106 (0.6 to 5.6 kbp) and a total genome Mr that varies from 19.3×106 to 22.0×106 (29.2 to 33.3 kbp).
The pattern of size distribution of the genome segments (Table 11) varies widely between different cypoviruses (e.g. the smallest dsRNA has an estimated size that varies between 530 and 1440 bp). These size differences have formed a basis for the recognition and classification of distinct species (electropherotypes) of cypoviruses (with patterns of dsRNA migration, which differ significantly in the migration of at least three genome segments, and frequently in the majority of segments, as analyzed by electrophoresis using 1% agarose or 3% SDS-PAGE). The genome segment migration patterns of members of species Cypovirus 1, Cypovirus 12 and Cypovirus 14 have some overall similarity, although in each case at least three segments show significant migrational differences during agarose gel electrophoresis. These viruses also show significant serological cross-reactions. More recently, it has been shown that members of different cypovirus species can also be distinguished on the basis of RNA sequence comparisons (e.g. by comparison of genome segment 10: the polyhedrin gene).
The termini of the coding strands are common or very closely related for each of the different genome segments within members of the species Cypovirus 1, but differ from those reported for other cypovirus species (Table 12). Choristoneura fumiferana cypovirus 16 (CfCPV-16) shows high levels of overall sequence variation when compared to members of species Cypovirus 1, Cypovirus 2, Cypovirus 5, Cypovirus 14 or Cypovirus 15 and is therefore considered to be in a different species (Cypovirus 16), although it has a similar 5′ and different 3′ end to representatives of species Cypovirus 5. These data demonstrate that different cypovirus electropherotypes are likely to, but may not always, have different conserved RNA terminal sequences.
Table 11 Cypovirus genome segment size distribution (kbp) determined by sequence analyses or estimated from electrophoretic comparisons of the genomic dsRNA of cypoviruses 1 to 20
| Genome segment number | Cypovirus types (1 to 20) | |||||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | |
| Total genome | 24.8 | 25.5 | 26.7 | 27.5 | 26.3 | 27.2 | 25.6 | 27.0 | 24.1 | 27.6 | 25.5 | 26.1 | 25.2 | 25.3 | 24.9 |
| 24.6 | 24.7 | 23.9 | 22.0 |
| 1 | 4.19 | 4.06 | 4.29 | 4.17 | 4.17 | 4.17 | 4.32 | 4.54 | 4.32 | 4.31 | 4.60 | 4.43 | 4.26 | 4.33 | 4.36 |
| 3.87 | 4.17 | 4.17 | 3.70 |
| 2 | 3.86 | 4.06 | 4.12 | 4.17 | 4.17 | 4.06 | 4.15 | 4.54 | 4.18 | 4.31 | 4.40 | 4.12 | 4.26 | 4.06 | 4.19 |
| 3.75 | 3.79 | 3.76 | 3.65 |
| 3 | 3.85 | 3.83 | 4.12 | 4.17 | 4.17 | 4.00 | 4.02 | 4.40 | 4.07 | 4.02 | 4.40 | 4.12 | 4.03 | 3.92 | 3.88 |
| 3.58 | 3.79 | 3.64 | 3.60 |
| 4 | 3.26 | 3.65 | 3.69 | 3.90 | 3.69 | 3.72 | 3.81 | 3.92 | 3.62 | 4.02 | 3.83 | 3.67 | 3.60 | 3.34 | 3.31 |
| 3.30 | 3.25 | 3.27 | 3.10 |
| 5 | 2.85 | 2.21 | 3.60 | 2.43 | 3.22 | 2.73 | 2.54 | 3.69 | 2.34 | 2.50 | 1.98 | 3.30 | 3.20 | 3.16 | 2.26 |
| 2.40 | 2.88 | 2.11 | 2.20 |
| 6 | 1.80 | 1.93 | 2.29 | 2.17 | 2.17 | 2.36 | 2.27 | 1.90 | 1.72 | 2.29 | 1.98 | 2.00 | 1.60 | 1.78 | 1.86 |
| 1.90 | 1.80 | 1.89 | 1.75 |
| 7 | 1.50 | 1.79 | 2.15 | 1.95 | 2.06 | 2.23 | 2.02 | 1.30 | 1.72 | 2.29 | 1.35 | 1.44 | 1.40 | 1.39 | 1.78 |
| 1.85 | 1.47 | 1.70 | 1.40 |
| 8 | 1.33 | 1.56 | 1.08 | 1.72 | 1.21 | 1.63 | 1.08 | 1.19 | 0.78 | 1.69 | 1.27 | 1.27 | 1.14 | 1.25 | 1.23 |
| 1.50 | 1.42 | 1.28 | 1.40 |
| 9 | 1.19 | 1.38 | 0.83 | 1.47 | 0.88 | 1.40 | 0.85 | 0.88 | 0,69 | 1.21 | 0.98 | 1.13 | 0.98 | 1.14 | 1.16 |
| 1.50 | 1.18 | 1.18 | 1.25 |
| 10 | 0.99 | 0.98 | 0.60 | 1.44 | 0.88 | 0.90 | 0.53 | 0.65 | 0.69 | 0.99 | 0.71 | 0.64 | 0.78 | 0.96 | 0.90 | 1.171 | 0.90 | 0.93 | 0.87 | 0.85 |
| 11 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| 0.20* |
|
|
|
|
|
Sizes in bold are derived from sequence analysis of the genome segment. Previously published estimates of genome segment sizes for members of Cypovirus 2 to Cypovirus 13 have been adjusted in line with base pair values derived from sequencing studies of cDNA copies of genome segments from BmCPV-1.
* TnCPV-15 has been reported to contain an 11th small genome segment (200 bp).
Table 12 Conserved terminal sequences (positive strand) of cypovirus genome segments
| Virus species | Strain | 5′ end | 3′ end |
| Cypovirus 1 | BmCPV-1 | 5′-AGUAA | GUUAGCC-3′ |
|
| DpCPV-1 | 5′-AGUAA | GUUAGCC-3′ |
|
| LdCPV-1 | 5′-AGUA/GA/G | GU/CUAGCC-3′ |
| Cypovirus 2 | IiCPV-2 | 5′-AGUUUUA | UAGGUC-3′ |
| Cypovirus 4* | ApCPV-4 | 5′-AAUCGACG | GUCGUAUG-3′ |
| Cypovirus 5 | OpCPV-5 | 5′-AGUU | UUGC-3′ |
| Cypovirus 14 | LdCPV-14 | 5′-AGAA | CAGCU-3′ |
| Cypovirus 15 | TnCPV-15 | 5′-AUUAAAAA | GC-3′ |
| Cypovirus 16** | CfCPV-16 | 5′-AGUUUUU | UUUGUGC-3′ |
| (not classified) | UsCPV-17 | 5′-AGAACAAA | UACACU-3′ |
| (not classified) | ObCPV-18 | 5′-AGUAAAG/U/AC/U | U/CA/CGUUAGCU-3′ |
| (not classified) | ObCPV-19 | 5′AACAAAA/UA/U | A/UGA/UUUUGC-3′ |
| (not classified) | SuCPV-20 | 5′-AGAAAAC | CAUGGC-3′ |
| (not classified) | MvCPV-21 | 5′-AUAUAAUU | AGUUAGU-3′ |
* Based on genome Seg9 only.
** Based on genome Seg10 only.
Proteins
Cypovirus particles generally contain five to six distinct proteins, two to three with sizes of more than 100 kDa. For BmCPV-1, the structural proteins have sizes in kDa (and are identified as): 148 (VP1), 136 (VP2), 140 (VP3), 120 (VP4), 64 (VP6) and 31 kDa (VP7) (see Table 13). Polyhedra also contain a 25–37 kDa polyhedrin protein (28.5 kDa for BmCPV-1) that constitutes about 95% of the polyhedra protein dry weight. Due to the very high level of variation between different cypoviruses, it is unlikely that their homologous proteins will be identifiable simply by their migration order during PAGE.
Table 13 Genome segments and protein products of Bombyx mori cypovirus 1 (strain I)
| Genome segment | Size (bp) | Protein1 nomenclature (2) | Size (kDa) | Function (location) |
| Seg1 | 4190 | VP1 (VP1) | 148 | Major core CP (virion) |
| Seg2 | 3854 | VP2 (VP2) | 136 | RdRp (virion) |
| Seg3 | 3846 | (VP3) | 140 | (virion) |
| Seg4 | 3262 | VP3 (VP4) | 120 | Possible Mtr (virion) |
| Seg5 | 2852 | NS1 (NS5) | 101 | Non-structural, contains auto cleavage aa sequence, similar to FMDV 2Apro
|
| NS2 (NS5a) | 80* | |||
| NS6 (NS5b) | 23* | |||
| Seg6 | 1796 | VP4 (VP6) | 64 | Leucine zipper ATP/GTP binding protein (virion) |
| Seg7 | 1501 | NS3 | 50 | Non-structural, with “structural” cleavage products
|
| NS4 | (61*) | |||
|
|
| (VP7) | 58* | |
| 31* | ||||
| Seg8 | 1328 | VP5 or P44 (NSP8) | 44 | Unknown (shows anomalous migration during PAGE, with apparent size 55 kDa) |
| Seg9 | 1186 | NS5 (NSP9) | 36 | Non-structural, dsRNA binding |
| Seg10 | 944 | Polyhedrin (Pod) | 28.5 | Polyhedron matrix protein (Pod) |
Size of genome segments and encoded proteins determined by sequence analysis of the genome segments.
* Sizes of some proteins estimated from electrophoretic migration.
1 Protein nomenclature suggested by McCrae and Mertens (McCrae, M.A. and Mertens, P.P.C. (1983). In vitro translation studies on and RNA coding assignments for cytoplasmic polyhedrosis viruses. In: Compans, R.W. and Bishop, D.H.L. (eds.), Double-stranded RNA Viruses, NewYork: Elsevier Science, pp. 35–41.)
2 Alternative nomenclature suggested by Hagiwara et al. (Hagiwara, K., Rao, S., Scott, S.W. and Carner, G. R. (2002). Nucleotide sequences of segments 1, 3 and 4 of the genome of Bombyx mori cypovirus 1 encoding putative capsid proteins VP1, VP3 and VP4, respectively. J. Gen. Virol., 83, 1477–1482.)
Lipids
Cypoviruses are not known to contain any lipids in either virus particles or polyhedra.
Carbohydrates
The polyhedrin protein is glycosylated.
Genome organization and replication
For BmCPV-1, the coding assignments are indicated in Table 13. The cognate genes of other cypoviruses are not known. The large variations in the sizes of genome segments between most cypoviruses (apart from members of Cypovirus 1, Cypovirus 12 and Cypovirus 14) indicate that these assignments will not apply to other cypovirus species. Genome segment coding assignments generated by in vitro translation of individual denatured genome segment RNAs have been published for members of Cypovirus 1 and Cypovirus 2. These data and subsequent sequencing studies indicate that, in many cases, polyhedrin may be encoded by the smallest segment.
Unlike orthoreoviruses, cell entry and initiation of cypovirus replication in insect cells does not require modification of virions for activation of core-associated transcriptase enzymes. Uptake appears to be a relatively inefficient process in cell culture, which can be very significantly improved by the use of liposomes. Virus replication and assembly occur in the host cell cytoplasm, although there is some evidence that viral RNA synthesis may also occur in the nucleus. Replication is accompanied by the formation of viroplasms (viral inclusion bodies or virogenic stroma) within the cytoplasm. Viroplasms contain large amounts of virus proteins and virus particles. How genome segments are selected for packaging and assembly into progeny particles is not known. The importance of the terminal regions in this process is indicated by the packaging and transcription of a mutant Seg10 from an isolate of Cypovirus 1 that contained only 121 bp from the 5′ end and 200 bp from the 3′ end. Particles are occluded within polyhedra apparently at the periphery of the virogenic stroma, from about 15 h post infection onwards. The polyhedrin protein is produced late in infection and in large excess compared to other viral proteins. It is not known how polyhedrin synthesis is regulated.
Antigenic properties
Serological cross-comparisons of cypovirus structural and polyhedrin proteins support the use of genomic dsRNA electropherotypes as one of the species parameters for the genus Cypovirus. Virus isolates within a single electropherotype exhibit high levels of antigenic cross-reaction (in both polyhedrin and virion structural proteins), as well as efficient cross-hybridization of denatured genomic RNA, even under high-stringency conditions. In contrast, there is evidence of little or no serological cross-reaction between viruses representing different electropherotypes. Exceptions are members of Cypovirus 1 and Cypovirus 12, which show low level serological cross-reactions but have some overall similarity in electropherotype patterns and show a low level of cross-hybridization of their genome segments. Cypovirus 14 members also show some similarity in RNA electropherotype patterns to viruses in both Cypovirus 1 and Cypovirus 12 and may therefore also show some antigenic relationship and RNA sequence similarity with these viruses.
Biological properties
Cypoviruses have only been isolated from arthropods. Attempts to infect vertebrates or vertebrate cell lines have failed. In addition, cypovirus replication is inhibited at 35 °C. Even susceptible insect larvae treated with cypoviruses fail to develop infections at temperatures ≥35 °C. Cypoviruses are normally transmitted by ingestion of polyhedra on contaminated food materials. The polyhedra dissolve within the high pH environment of the insect gut and release occluded virus particles, which then infect the cells lining the gut wall. Virus infection in larvae is generally restricted to the columnar epithelial cells of the midgut, although goblet cells may also become infected. Cypovirus replication in the fat body has been reported. In larvae, the virus infection spreads throughout the midgut region. In some species the entire gut is occasionally infected. The production of very large numbers of polyhedra gives the gut a characteristically creamy-white appearance. In infected cells the endoplasmic reticulum is progressively degraded, mitochondria enlarge and the cytoplasm becomes highly vacuolated. In most cases, the nucleus shows few pathological changes. An exception is one little-studied cypovirus strain that produces inclusion bodies within the nucleus. In the later stages of infection, cellular hypertrophy is common and microvillae are reduced or completely absent. Very large numbers of polyhedra are released by cell lysis into the gut lumen and excreted. The gut pH is lowered during infection and this prevents dissolution of progeny polyhedra in the gut fluid.
The majority of cypovirus infections produce chronic disease, often without extensive larval mortality. Consequently, many individuals reach the adult stage even though heavily diseased. However, cypovirus infections produce symptoms of starvation due to changes in the gut cell structure and reduced adsorptive capacity. Infected larvae stop feeding as early as 2 days post infection. Larval body size and weight are often reduced and diarrhea is common. The larval stage of the host can be significantly increased (about by 1.5 times the normal generation time). The size of infected pupae is frequently reduced, and the majority of diseased adults are malformed. They may not emerge correctly, and may be flightless. Infected females may exhibit a reduced egg-laying capacity.
Virus can be transmitted on the surface of eggs, producing high levels of infection in the subsequent generation. However, provided the egg surface is disinfected, no transovarial transmission has been observed. The infectious dose increases dramatically in the later larval instars. Different virus strains vary significantly in virulence. Larvae can recover from cypovirus infection, possibly because the gut epithelium has considerable regenerative capacity and because infected cells are shed at each larval molt.
Species demarcation criteria in the genus
Cypoviruses are currently classified within 16 species, most of which were initially characterized by their distinctive dsRNA electropherotype patterns. Cross-hybridization analyses of the dsRNA, serological comparisons of cypovirus proteins and, more recently, comparison of RNA sequences have confirmed the validity of this classification and have identified new virus species. However, relatively few cypoviruses have been characterized, suggesting that there may be many more distinct species that are as yet unidentified.
The system of nomenclature currently used to identify different cypovirus isolates takes account of both the virus species and the host species from which the virus was originally isolated (e.g. BmCPV-1). The relationships between different cypoviruses within a single electropherotype or species, or with other cypovirus types, are not fully understood at the molecular level. Sequence analyses of genome segments from distinct isolates have shown high levels of identity within a single species. For example, the different isolates of Cypovirus 5 that have been analyzed show >98% identity in genome Seg10 (the polyhedrin gene), whereas isolates of Cypovirus 1 show 80–98% nt sequence identity in this gene. In contrast, comparisons of unrelated types showed only low levels of sequence identity (20–23%). Studies of genomic RNA from different cypovirus isolates have demonstrated that, although there may be slightly higher conservation in the largest genome segments (possibly as a result of functional constraints), the level of variation is relatively uniform across the whole genome. This contrasts with reoviruses that infect vertebrates, perhaps because there is no neutralizing antibody response in the host insects of cypoviruses and consequently no selective pressure to promote variation in outer capsid proteins and the genome segments from which they are translated.
In addition to the other general criteria used throughout the family, members of a species in the genus Cypovirus may be identified by:
- Similar electrophoretic migration of at least seven genome segments, as analyzed using either an agarose, or a low percentage (3%) polyacrylamide gel system. Viruses of different species have significant migrational differences in at least three genome segments.
- A high degree of nucleotide or amino acid sequence conservation (estimated >80% for the former).
- Cross-hybridization of genome segments under high stringency conditions (designed to detect >90% identity).
List of species in the genus Cypovirus
| Cypovirus 1 |
|
|
| Bombyx mori cypovirus 1 | [Seg1: AF323781, Seg2: AF323782, Seg3: AF323783, Seg4: AF323784, Seg5: AB035733, Seg6: AB030014, Seg7: AB030015, Seg8: AB016436, Seg9: AF061199, Seg10: D37768] | (BmCPV-1) |
| Dendrolimus punctatus cypovirus 1 | [Seg1: AY163247, Seg2: AY147187, Seg3: AY167578, Seg4: AF542082, Seg5: AY163248, Seg6: AY163249, Seg7: AY211091, Seg8: AF513912, Seg9: AY310312, Seg10: AF541985] | (DpCPV-1) |
| Dendrolimus spectabilis cypovirus 1 |
| (DSCPV-1) |
| Lymantria dispar cypovirus 1 | [Seg1: AF389462, Seg2: AF389463, Seg3: AF389464, Seg4: AF389465, Seg5: AF389466, Seg6: AF389467, Seg7: AF389468, Seg8: AF389469, Seg9: AF389470, Seg10: AF389471] | (LdCPV-1) |
| Cypovirus 2 |
|
|
| Aglais urticae cypovirus 2 |
| (AuCPV-2) |
| Agraulis vanillae cypovirus 2 |
| (AvaCPV-2) |
| Arctia caja cypovirus 2 |
| (AcCPV-2) |
| Arctia villica cypovirus 2 |
| (AviCPV-2) |
| Boloria dia cypovirus 2 |
| (BdCPV-2) |
| Dasychira pudibunda cypovirus 2 |
| (DpCPV-2) |
| Eriogaster lanestris cypovirus 2 |
| (ElCPV-2) |
| Hyloicus pinastri cypovirus 2 |
| (HpCPV-2) |
| Inachis io cypovirus 2 |
| (IiCPV-2) |
| Lacanobia oleracea cypovirus 2 |
| (LoCPV-2) |
| Malacosoma neustria cypovirus 2 |
| (MnCPV-2) |
| Mamestra brassicae cypovirus 2 |
| (MbCPV-2) |
| Operophtera brumata cypovirus 2 |
| (ObCPV-2) |
| Papilio machaon cypovirus 2 |
| (PmCPV-2) |
| Phalera bucephala cypovirus 2 |
| (PbCPV-2) |
| Pieris rapae cypovirus 2 |
| (PrCPV-2) |
| Cypovirus 3 |
|
|
| Anaitis plagiata cypovirus 3 |
| (ApCPV-3) |
| Arctia caja cypovirus 3 |
| (AcCPV-3) |
| Danaus plexippus cypovirus 3 |
| (DpCPV-3) |
| Gonometa rufibrunnea cypovirus 3 |
| (GrCPV-3) |
| Malacosoma neustria cypovirus 3 |
| (MnCPV-3) |
| Operophtera brumata cypovirus 3 |
| (ObCPV-3) |
| Phlogophera meticulosa cypovirus 3 |
| (PmCPV-3) |
| Pieris rapae cypovirus 3 |
| (PrCPV-3) |
| Spodoptera exempta cypovirus 3 |
| (SexmCPV-3) |
| Cypovirus 4 |
|
|
| Actias selene cypovirus 4 |
| (AsCPV-4) |
| Antheraea assamensis cypovirus 4 | [Seg9: AF374299] | (AaCPV-4) |
| Antheraea mylitta cypovirus 4 | [Seg9: AF374298] | (AmCPV-4) |
| Antheraea pernyi cypovirus 4 |
| (ApCPV-4) |
| Antheraea proylei cypovirus 4 | [Seg9: AF374300] | (AprCPV-4) |
| Cypovirus 5 |
|
|
| Euxoa scandens cypovirus 5 | [Seg10: J04338] | (EsCPV-5) |
| Heliothis armigera cypovirus 5 | [Seg10: U06196] | (HaCPV-5) |
| Orgyia pseudosugata cypovirus 5 | [Seg10: U06194] | (OpCPV-5) |
| Spodoptera exempta cypovirus 5 |
| (SexmCPV-5) |
| Tichoplusia ni cypovirus 5 |
| (TnCPV-5) |
| Cypovirus 6 |
|
|
| Aglais urticae cypovirus 6 |
| (AuCPV-6) |
| Agrochola helvolva cypovirus 6 |
| (AhCPV-6) |
| Agrochola lychnidis cypovirus 6 |
| (AlCPV-6) |
| Anaitis plagiata cypovirus 6 |
| (ApCPV-6) |
| Anti xanthomista cypovirus 6 |
| (AxCPV-6) |
| Biston betularia cypovirus 6 |
| (BbCPV-6) |
| Eriogaster lanestris cypovirus 6 |
| (E1CPV-6) |
| Lasiocampa quercus cypovirus 6 |
| (LqCPV-6) |
| Cypovirus 7 |
|
|
| Mamestra brassicae cypovirus 7 |
| (MbCPV-7) |
| Noctua pronuba cypovirus 7 |
| (NpCPV-7) |
| Cypovirus 8 |
|
|
| Abraxas grossulariata cypovirus 8 |
| (AgCPV-8) |
| Heliothis armigera cypovirus 8 |
| (HaCPV-8) |
| Malacosoma disstria cypovirus 8 |
| (MdCPV-8) |
| Nudaurelia cytherea cypovirus 8 |
| (NcCPV-8) |
| Phlogophora meticulosa cypovirus 8 |
| (PmCPV-8) |
| Spodoptera exempta cypovirus 8 |
| (SexmCPV-8) |
| Cypovirus 9 |
|
|
| Agrotis segetum cypovirus 9 |
| (AsCPV-9) |
| Cypovirus 10 |
|
|
| Aporophyla lutulenta cypovirus 10 |
| (AlCPV-10) |
| Cypovirus 11 |
|
|
| Heliothis armigera cypovirus 11 |
| (HaCPV-11) |
| Heliothis zea cypovirus 11 |
| (HzCPV-11) |
| Lymantria dispar cypovirus 11 |
| (LdCPV-11) |
| Mamestra brassicae cypovirus 11 |
| (MbCPV-11) |
| Pectinophora gossypiella cypovirus 11 |
| (PgCPV-11) |
| Pseudaletia unipuncta cypovirus 11 |
| (PuCPV-11) |
| Spodoptera exempta cypovirus 11 |
| (SexmCPV-11) |
| Spodoptera exigua cypovirus 11 |
| (SexgCPV-11) |
| Cypovirus 12 |
|
|
| Autographa gamma cypovirus 12 |
| (AgCPV-12) |
| Mamestra brassicae cypovirus 12 |
| (MbCPV-12) |
| Pieris rapae cypovirus 12 |
| (PrCPV-12) |
| Spodoptera exempta cypovirus 12 |
| (SexmCPV-12) |
| Cypovirus 13 |
|
|
| Polistes hebraeus cypovirus 13 |
| (PhCPV-13) |
| Cypovirus 14 |
|
|
| Heliothis armigera cypovirus 14 (‘A’ strain) |
| (HaCPV-14) |
| Lymantria dispar cypovirus 14 | [Seg1: AF389452, Seg2: AF389453, Seg3: AF389454, Seg4: AF389455, Seg5: AF389456, Seg6: AF389457, Seg7: AF389458, Seg8: AF389459, Seg9: AF389460, Seg10: AF389461] | (LdCPV-14) |
| Cypovirus 15 |
|
|
| Trichoplusia ni cypovirus 15 | [Seg1: AF291683, Seg2: AF291684, Seg3: AF291685, Seg4: AF291686, Seg5: AF291687, Seg6: AF291688, Seg7: AF291689, Seg8: AF291690, Seg9: AF291691, Seg10: AF291692, Seg11*: AF291693] | (TnCPV-15) |
| Cypovirus 16 |
|
|
| Choristoneura fumiferana cypovirus 16 | [Seg10, U95954] | (CfCPV-16) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ] and assigned abbreviations ( ) are also listed.*TnCPV-15 has been reported as having 11 distinct genome segments.
List of other related viruses which may be members of the genus Cypovirus but have not been approved as species
In addition to many other lepidopteran cypoviruses that have been described (but are otherwise uncharacterized), there are hymenopteran cypoviruses. One isolate from a freshwater daphnid has been reported. In total, more than 230 cypoviruses have been described. The total potential number of species is unknown but, based on the number and diversity of insect species, is thought likely to be considerably greater than 16.
| Uranotaenia sapphirina cypovirus 17 | [Seg10: AY876384] | (UsCPV-17) |
| Culex restuans cypovirus 17 | [Seg10: DQ212785] | (CrCPV-17) |
| Operophtera brumata cypovirus 18 | [Seg5: DQ192245, Seg6: DQ192246, Seg7: DQ192247, Seg8: DQ192248, Seg9: DQ192249, Seg10: DQ192250] | (ObCPV-18) |
| Operophtera brumata cypovirus 19 | [Seg2: DQ192251, Seg5: DQ192252, Seg9: DQ192253, Seg10: DQ192254] | (ObCPV-19) |
| Simulium ubiquitum cypovirus 20 | [Seg10: DQ834386] | (SuCPV-20) |
| Maruca vitrata cypovirus 21 |
| (MvCPV-21) |
| Heliothis armigera cypovirus (“B” strain) |
| (HaCPV-B) |
| Maruca vitrata cypovirus (A strain) |
| (MvCPV-A) |
| Maruca vitrata cypovirus (B strain) |
| (MvCPV-B) |
| Plutella xylostella cypovirus |
| (PxCPV) |
Phylogenetic relationships within the genus
The available sequence data for members of Cypovirus 1, Cypovirus 2, Cypovirus 5, Cypovirus 14, Cypovirus 15 and Cypovirus 16 allow a comparison of some genes of these viruses, showing not only that different cypovirus species are quite distantly related but also (at least for the viruses analyzed) that there is a high level of conservation within a single species. For example, a comparison of polyhedrin genes shows only 20–23% sequence identity between the members of different cypovirus species, but 89–98% identity between different isolates of a single species (Figure 14). These data indicate that sequence analyses and comparisons are effective methods for distinguishing and identifying the members of cypovirus species.
Genus Idnoreovirus
Type species Idnoreovirus 1
Distinguishing features
Idnoreovirus virions have a roughly spherical outer capsid, which may have small icosahedrally arranged surface projections when viewed by negative staining and electron microscopy. The core particles have 12 icosahedrally arranged surface turrets or spikes, which appear similar to those of the cypoviruses. The genome is composed of 10 segments of linear dsRNA. All of the members of the genus that have been described infect insects.
Virion properties
Morphology
Particles are non-enveloped. Unlike the cypoviruses, there are no polyhedra, and the virus particles have a clearly defined outer capsid layer (Figure 15). Electron microscopy and negative staining of virions (e.g. with aqueous uranyl acetate) shows that they are double shelled, roughly spherical in appearance (with icosahedral symmetry), with an estimated diameter of about 70 nm. Core particles (estimated diameter of ca. 60 nm) display 12 icosahedrally arranged, prominent surface projections (ca. 15 nm in length – identified as turrets or spikes), which may lose a portion near the tip and at least in some cases appear to be tubular. In particles where stain has entered the central space, there appears to be material (considered likely to be protein) associated with the spike structure on the inside of the inner capsid shell (Figure 15, far-right panel).
Physicochemical and physical properties
Limited studies of some viruses in the genus indicate that particles are resistant to freon (trichlorotrifluoroethane) and CsCl. They may also be resistant to chymotrypsin. Intact particles and cores of Diadromus pulchellus idnoreovirus-1 (DpIRV-1) have densities of 1.370 g cm−3 and 1.385 g cm−3, respectively, and intact virions and empty particles of Dacus oleae idnoreovirus-4 (DoIRV-4) have a density of about 1.38 g cm−3 and 1.28 g cm−3, respectively, as determined by CsCl gradient centrifugation. The outer capsid layer of Hyposoter exiguae idnoreovirus-2 (HeIRV-2) was disrupted by brief exposure to 0.4% sodium sarcosinate, releasing the virus core.
Nucleic acid
The genome usually consists of 10 dsRNA segments that are numbered in order of reducing molecular weight (or increasing electrophoretic mobility) during agarose gel electrophoresis (AGE). By analogy with other members of the family Reoviridae, the genome segment migration patterns during AGE are likely to be characteristic for each idnoreovirus species.
The total genome of DpIRV-1 (the current type member) contains an estimated 25.15 kbp of dsRNA, with individual segments that range between about 4.8 to 0.98 kbp, showing a 5-5 electrophoretic migration pattern by 1% AGE. However, the virions of DpIRV-1 may be unusual in the genus, since they can sometimes also contain an eleventh, 3.33 kbp dsRNA segment, the presence of which is related to, and may help determine, the sex and ploidy of the individual wasp host. This additional dsRNA (migrating between Seg3 and Seg4) contains sequences similar to, and therefore possibly derived by, duplication of an incomplete Seg3 (3.8 kbp).
The genome segments of HeIRV-2 range in estimated size from about 3.9 to 1.35 kbp, with a 4-6 electrophoretic migration pattern by 12.5% polyacrylamide gel electrophoresis (PAGE). DoIRV-4 contains an estimated 23.4 kbp of dsRNA, with the estimated lengths of individual segments ranging from about 3.8 to 0.7 kbp and a 5-3-2 electrophoretic migration pattern by 7% PAGE. Ceratitis capitata idnoreovirus-5 (CcIRV-5) has a 3-3-4 genome segment migration pattern by 6% PAGE, and has clear similarities to Drosophila melanogaster idnoreovirus-5 (DmIRV-5), as analyzed by 0.5% agarose-2% polyacrylamide gels, suggesting that, despite some serological differences, these viruses belong to the same species (Idnoreovirus – 5). It is unclear how closely Drosophila S virus (which causes the “S” phenotype in D. simulans) is related to the other Drosophila-derived idnoreoviruses. It is therefore currently listed as a possible member of the genus.
The total genome of Operophtera brumata idnoreovirus (ObIRV) contains 23,647 kbp, with genome segments that range in size from 4.17 to 1.51 kbp, giving an electrophoretic migration pattern of 4-2-4. This is the only complete genome sequence available and appears to represent a new species. All 10 genome segments of ObIRV contain five fully-conserved bases at the 3′ termini of their positive sense RNA strands, which are different from those found in other species that have been characterized within the family Reoviridae. There is also significant conservation at the 5′ termini (four of the six terminal bases). In each case, the two terminal bases are complementary (5′AA to 3′TT).
In contrast, initial sequencing studies suggest that the 3′ termini of DpIRV-1 genome segments are more variable than those of other species in the family Reoviridae, with little sign of conservation. However, conserved sequences were detected at the 5′ termini (Table 14), and these are different from those found in other reoviruses. No sequence data are currently available for other members of this genus.
Table 14 Conserved terminal sequences (positive strand) of idnoreovirus genome segments
| Virus species | Serotype or strain | 5′ end | 3′ end |
| Idnoreovirus - 1 | DpIRV-1 | 5′-(a/u/g)CAAUUU | (variable)-3′ |
| (not classified) | ObIRV | 5′-AA(A/C)(A/U)AA | AGGUU-3′ |
Proteins
Native viral proteins of idnoreoviruses have not been characterized extensively. However, sequencing of the ObIRV genome indicates that it encodes a total of 10 proteins, ranging in size from 49.6 to 155.7 kDa. The composition of these proteins was deduced from the sequence of the viral genome, as indicated in Table 15, where they are named as VP1 to VP10 based on the molecular weight of the genome segment (segment number) from which they are translated.
The genome of DpIRV-1 encodes 11 proteins with size ranging from 21 to 140 kDa. Three of these proteins appeared to be glycosylated (ca. 21, 15 and 35 kDa).
Table 15 Genome segments and protein products of Operophtera brumata idnoreovirus
| Genome segment | Size (bp) | Protein nomenclature | Protein size aa (kDa) | Structure/putative function |
| Seg1 | 4170 | VP1 | 1358 (155.7) | RNA-dependent RNA polymerase |
| Seg2 | 3780 | VP2 | 1207 (137) |
|
| Seg3 | 3595 | VP3 | 1161 (133) | T2 subcore shell |
| Seg4 | 3362 | VP4 | 1091 (122.7) |
|
| Seg5 | 2106 | VP5 | 620 (69.2) |
|
| Seg6 | 1935 | VP6 | 594 (68.4) |
|
| Seg7 | 1606 | VP7 | 499 (57.1) |
|
| Seg8 | 1584 | VP8 | 467 (51.6) |
|
| Seg9 | 1547 | VP9 | 437 (49.6) |
|
| Seg10 | 1509 | VP10 | 467 (53.9) |
|
The sizes of dsRNA segments and their putative translation products have been determined by sequence analyses.
Lipids
None reported.
Carbohydrates
Three proteins from DpIRV-1 appeared to be glycosylated (ca. 21, 15 and 35 kDa).
Genome organization and replication
On the basis of the overall similarity of idnoreoviruses to other members of the family Reoviridae, it is assumed that many aspects of the genome organization and replication are similar. Thus, it is likely that the virus core contains transcriptase complexes that synthesize mRNA copies of the individual genome segments. These mRNAs are likely to be exported and translated to produce viral proteins in the host cytoplasm. These positive sense RNAs are also likely to form templates for negative strand synthesis during progeny virus assembly and maturation. Each of the genome segments that has been sequenced represents a single gene, with a single large ORF and relatively short terminal NCRs (Table 15).
Antigenic properties
Unknown.
Biological properties
The idnoreoviruses can infect insect species, where in many cases they appear to cause few pathological effects. However, they may significantly alter the biological properties of the individual host. Drosophila S virus appears to be associated with the S phenotype in D. simulans. The presence of an additional 3.33 kbp-dsRNA segment in DpIRV-1 is related to, and may help determine, the sex and ploidy of the host. This segment may play a role in the biology of this wasp species, possibly by providing information necessary for larval development.
Species demarcation criteria in the genus
See the general criteria used throughout the family.
List of species in the genus Idnoreovirus
| Idnoreovirus 1 |
|
|
| Diadromus pulchellus idnoreovirus-1 | [Seg2: X82049; Seg3: X80481; Seg7: X82048; Seg8: X82047; Seg9: X82046; Seg10: X82045; Additional segment: X80480] | (DpIRV-1) |
| (Diadromus pulchellus reovirus) |
| |
| Idnoreovirus 2 |
|
|
| Hyposoter exiguae idnoreovirus-2 |
| (HeIRV-2) |
| (Hyposoter exiguae reovirus) | ||
| Idnoreovirus 3 |
|
|
| Musca domestica idnoreovirus-3 |
| (MdIRV-3) |
| (Musca domestica reovirus) | ||
| (Housefly virus) | ||
| Idnoreovirus 4 |
|
|
| Dacus oleae idnoreovirus-4 |
| (DoIRV-4) |
| (Dacus oleae reovirus) | ||
| Idnoreovirus 5 |
|
|
| Ceratitis capitata idnoreovirus-5 |
| (CcIRV-5) |
| (Ceratitis capitata I virus) | (DmIRV-5) | |
| Drosophila melanogaster idnoreovirus-5 | ||
| (Drosophila F virus) |
Species names are in italic script; names of isolates are in roman script; names of synonyms are in roman script and parentheses. Sequence accession numbers [ ] and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Idnoreovirus but have not been approved as species
| Drosophila S virus |
| (DSV) |
| Operophtera brumata idnoreovirus | [Seg1: DQ192235; Seg2: DQ192236; Seg3: DQ192237; Seg4: DQ192238; Seg5: DQ192239; Seg6: DQ192240; Seg7: DQ192241; Seg8: DQ192242; Seg9: DQ192243; Seg10: DQ192244] | ObIRV |
Similarity with other taxa
There is no evidence of significant sequence homology between idnoreovirus genes and those of the other members of the family Reoviridae, other than that of the RNA-dependent RNA polymerase. The RdRp of ObIRV has 20–27% amino acid sequence identity to those of viruses in other genera of the subfamily Spinareovirinae.
Genus Dinovernavirus
Type species Aedes pseudoscutellaris reovirus
Distinguishing features
The dinovernavirus genome consists of nine segments of dsRNA. Aedes pseudoscutellaris reovirus (APRV) is currently the only member of this genus and was isolated from persistently infected Aedes pseudoscutellaris cells (AP61).
Virion properties
Morphology
Purified APRV particles, isolated using an Iodixanol (Optiprep®) gradient and analyzed by TEM, had the morphology typical of cores of turreted reoviruses (Figure 16). In particular, the morphology of APRV appears similar to that observed for cypovirus particles (which are single-shelled). It is therefore considered likely that APRV is also single-shelled. The mean diameter of the particle is approximately 49–50 nm, with a central section that is 36–37 nm. Turrets were visible projecting from the particle surface. These also appear to have sections near the tip that can become detached (Figure 16) in a manner similar to that observed previously with cypoviruses.
Physicochemical and physical properties
At 4 °C, APRV is stable for long periods, even non-purified in cell culture lysate, which is a convenient way for medium-term storage. Heating to 55 °C significantly reduces infectivity. The virus is stable upon treatment with freon, which can be used for purification of virus particles from cell lysate. Virion infectivity is not affected by treatment with 1% deoxycholate, but is abolished by treatment with sodium dodecyl sulfate. Viruses can be stored for long periods at −80 °C, and infectivity can be further protected by addition of 50% fetal calf serum. Infectivity is retained at pH values between 6 and 8. Between pH 4 and 5 or between pH 9 and 10, infectivity is reduced by a factor of 10. Virion morphology (observed by electron microscopy) was considerably distorted at pH values lower than 5 and virions were completely disrupted at pH values lower than 3.5.
Nucleic acid
The dinovernavirus genome consists of nine linear segments of dsRNA, which numbered in order of reducing Mr, or increasing electrophoretic mobility during agarose gel electrophoresis (AGE). The genome comprises 23,355 bp, with segment lengths that range between 3817 and 1147 bp. Analysis of genomic RNAs by 1% AGE shows a 5-1-3 migration pattern (electropherotype) (Figure 17).
The positive strands of all nine segments of the APRV genome have conserved sequences in the 5′ and 3′ non-coding regions (NCRs) (5′-AGUUA/UAAA/CA/C----------U/GUUnnnC/UnnA/UAGU-3′, where n = any nucleotide; Table 16). Comparisons of these conserved termini with those of viruses in the genera Cypovirus, Oryzavirus and Fijivirus (Table 16) showed that only the first three nucleotides in the 5′ NCR (AGU) are conserved between APRV, cypoviruses and NLRV (species Nilaparvata lugens reovirus, genus Fijivirus). The 3′ termini of APRV differ from those of the cypoviruses, NLRV and RRSV (species Rice ragged stunt virus, genus Oryzavirus).
In contrast to those of cypoviruses and fijiviruses, the first and last nucleotides of the APRV genome segments are complementary (A and U). The mean G+C content of the APRV genome is 34.4%, compared to 34.8% for NLRV, 44.7% for RRSV and 43% for cypoviruses.
Table 16 Conserved terminal sequences (positive strand) of dinovernavirus genome segments and comparison to those of cypoviruses, fijiviruses, oryzaviruses and idnoreoviruses
| Genus | Virus | 5′ end | 3′ end |
| Dinovernavirus | APRV | 5′-AGUUA/UAAA/CA/C | A/UAGU-3′ |
| Cypovirus | CPV1 | 5′-AGUAAA | GUUAGCC-3′ |
|
| CPV2 | 5′-AGUUU | GAGUUUGC-3′ |
|
| CPV15 | 5′-AUUAAAAA | GC-3′ |
|
| CPV4 | 5′-AAUCGACG | GUCGUAUG-3′ |
| Fijivirus | NLRV | 5′-AGU | GUUGUC-3′ |
|
| MRCV | 5′-AAGUUUUUU | GUC-3′ |
|
| FDV | 5′-AAGUUUUUU | GUC-3′ |
|
| RBSDV | 5′-AAGUUUUUU | GUC-3′ |
| Oryzavirus | RRSV | 5′-GAUAAA | GUGC-3′ |
| Idnoreovirus | ObIRV | 5′-AAA/CA/UAA | AGGUU-3′ |
Proteins
APRV proteins were inferred from the ORFs in the dsRNA segments. Each segment encodes a single protein (Table 17).
Table 17 Genome segments and protein products of Aedes pseudoscutellaris reovirus
| Genome segment | Size (bp) | Protein nomenclature | Protein size aa (kDa) | Structure/putative function |
| Seg1 | 3817 | VP1 | 134 | Unknown |
| Seg2 | 3752 | VP2 | 143 | RdRp |
| Seg3 | 3732 | VP3 | 136 | Unknown |
| Seg4 | 3375 | VP4 | 116 | Unknown |
| Seg5 | 3227 | VP5 | 121 | Unknown |
| Seg6 | 1775 | VP6 | 62 | Unknown |
| Seg7 | 1171 | VP7 | 39.4 | Unknown |
| Seg8 | 1151 | VP8 | 39.8 | Unknown |
| Seg9 | 1147 | VP9 | 32 | Unknown |
Lipids
None reported.
Carbohydrates
None reported.
Genome organization and replication
APRV persistently infects the AP61 cell line, with an estimated 6–8 particles per cell. Treatment of cells with 2-aminopurine showed a 10-fold increase in the number of viral particles, as shown by qPCR. The virus replicates to high titres in C6/36 cells and considerable amounts (over 40% of progeny) are liberated into the culture medium.
Extracts of mammalian cells inoculated with APRV fail to support replication, indicating that this virus does not grow in mammalian cells. The RNA extracted from the blood of mice inoculated with APRV also remained negative (by RT-PCR) from 0 to 12 days post-injection.
Antigenic properties
None reported.
Biological properties
APRV was isolated from persistently infected AP61 cells. AP61 cells collected from different sources were consistently found to contain the same APRV strain. Virus-like particles were also identified in the original AP61 line by electron microscopy. Care should be taken when using AP61 cells to propagate any other mosquito-borne arboviruses, particularly dsRNA viruses, as these readily become contaminated with APRV. Upon infection with other dsRNA viruses, the dormant or persistent state of APRV is activated to a productive infection, releasing APRV in larger amounts into the cell culture supernatant, along with other mosquito-borne dsRNA viruses (such as orbiviruses and seadornaviruses) that were used to infect the cells.
Species demarcation criteria in the genus
Not applicable.
List of species in the genus Dinovernavirus
| Aedes pseudoscutellaris reovirus |
|
|
| {Aedes pseudoscutellaris mosquitoes} | ||
| Aedes pseudoscutellaris reovirus | [Seg1: DQ087276., Seg2: DQ087277., Seg6: DQ087278., Seg7: DQ087279., Seg8: DQ087280., Seg9: DQ087281] | (APRV) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numberss [ ], arthropod vector and host names { } and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Dinovernavirus but have not been approved as species
None reported.
Phylogenetic relationships with other taxa
The morphology of purified APRV is very similar to that of the single shelled cypoviruses. Based on electron microscopy analysis, it was initially thought that APRV would represent a new cypovirus from mosquitoes. However, unlike the cypoviruses, APRV particles are non-occluded and full-length sequence analyses confirmed that it only has nine genome segments. These analyses also demonstrated a genetic distance between APRV and the cypoviruses that is consistent with membership of distinct genera. Indeed, a similar relationship was also demonstrated with the oryzaviruses and fijiviruses (Table 18).
It is interesting to note that the VP1 of APRV (1189 amino acid residues) shows a significant match (22% identity, with a probability of 2e26) over a sequence between residues 756 and 1173 (a 418 residue fragment) to a minor structural protein of a ssDNA parvo-like virus isolated from the silkworm. Observation of significant similarities between dsRNA viruses and DNA viruses is rather unusual. APRV VP3 also shows some similarity to the histidine kinase of Heliothis zea virus, which is in genus Nudivirus.
Table 18 Amino acid identity values between APRV, cypoviruses, fijiviruses and oryzaviruses
| APRV proteins | CPV1 - proteins | RRSV- proteins | NLRV- proteins | |||
| (position) | [% identity] | (position) | [% identity] | (position) | [% identity] | |
| VP1 | P138 (768-1157) | [23] | P1 (963-1208) | [22] |
|
|
| VP2(Pol) | V2 (23-1206) | [26] | P4 (68-1030) | [23] | P165.9 (599–624) | [22] |
| VP3 | V1 (6-1302) | [21] | P3 (398-645) | [21] | P106.4 (339–698) | [21] |
| VP4 | NS1 (795-874) | [28] | P5 (727-789) | [31] |
|
|
| VP5 | V3 (38-1045) | [22] | P2 (589-1102) | [19] |
|
|
| VP6 | V4 (1-555) | [22] | NS7 (399-591) | [23] | P73.5 (399–591) | [23] |
| VP7 |
|
|
|
|
|
|
| VP8 |
|
|
|
|
|
|
| VP9 |
|
|
|
| NS35.2 (169–218) | [31] |
Genus Coltivirus
Type species Colorado tick fever virus
Distinguishing features
The coltivirus genome consists of 12 segments of dsRNA. During replication, viruses are found in the cell cytoplasm, associated with granular matrices (viral inclusion bodies: VIB), arrays of filaments or tubules and fine kinky threads. Immunofluorescent staining reveals nucleolar fluorescence. Viruses are transmitted to vertebrate hosts by tick vectors.
Virion properties
Morphology
Coltivirus particles are 60–80 nm in diameter having two concentric capsid shells with a core that is about 50 nm in diameter. Electron microscopic studies, using negative staining, have shown that particles have a relatively smooth surface capsomeric structure and icosahedral symmetry (Figure 18). Particles are found intimately associated with filamentous structures and granular matrices in the cytoplasm. The majority of the viral particles are non-enveloped, but a few acquire an envelope structure during their passage through the endoplasmic reticulum.
Physicochemical and physical properties
The buoyant density of the virus in CsCl is 1.38 g cm−3. Viruses are stable between pH 7 and 8, but lose infectivity at pH 3.0. At 4 °C, the virus is stable for long periods when stored in presence of 50% fetal calf serum in 0.2 M Tris-HCl pH 7.8. Heating to 55 °C considerably decreases the viral infectivity. Coltiviruses are fairly stable upon treatment with non-ionic detergents, sodium lauroyl sarcosine, or freon but the viral infectivity is abolished by treatment with sodium deoxycholate or sodium dodecyl sulfate. Moderate ultrasonic oscillation treatment does not destroy infectivity and can be used in virus purification. Viruses can be stored for long periods at −80 °C, and infectivity is further protected by addition of 50% fetal calf serum.
Nucleic acid
The genome consists of 12 dsRNA segments that are numbered in order of decreasing size, or increasing electrophoretic mobility during agarose gel electrophoresis. The genome comprises approximately 29,000 bp, with segment lengths that range between 4350 and 675 bp. The genomic RNA of Colorado tick fever virus (CTFV) migrates in three size classes (Figure 19) during 1% agarose gel electrophoresis (AGE): the large (long) or L-segments (Seg1 to Seg4), the medium length or M-segments (Seg5 to Seg10) and the small (short) or S-segments (Seg11 and Seg12). The dsRNA genome of Eyach virus (EYAV) has not yet been analyzed by AGE. The terminal 5′- and 3′-sequences of the coltivirus genome are conserved (Table 19).
RNA cross-hybridization analysis shows that CTFV isolates have remained relatively homogenous, and distinct CTFV serotypes have been difficult to distinguish (although some sequence variation does occur, for example in Seg4 and Seg6). The overall nt sequence similarity between genome segments of different CTFV isolates, ranges between 90% and 100%. However, the degree of similarity between homologous segments from CTFV and EYAV isolates ranges from 55% to 86%. Seg7 from EYAV is partially homologous to Seg6 of CTFV, although the protein it encodes also shows similarities to a sarcolemmal-associated protein from the European rabbit Oryctolagus cunniculis, which is thought to be one of its major hosts. Coltivirus mRNAs are usually regarded as non-infectious. However fully functional and infectious viruses have been recovered by the introduction of all 12 mRNAs into BSR cells.
Table 19 Conserved terminal sequences (positive strand) of coltivirus genome segments
| Virus species | Strain | 5′ end | 3′ end |
| Colorado tick fever virus | CTFV-Fl | 5′-G/CACAUUUUGU | UGCAGUG/C-3′ |
| Eyach virus | EYAV-Fr578 | 5′-GACAA/UUU | A/UUGC/UAGUC-3′ |
Lipids
None reported.
Carbohydrates
None reported.
Genome organization and replication
CTFV genome segments 1 to 8 and 10 to 11, each encodes a single protein. However, Seg9 contains a leaky stop codon and produces two proteins when translated in a cell-free system or in cells transfected with a plasmid containing the full length Seg9. The shorter 38 kDa protein, (VP9ter) is the early-termination translation product, while the read-through protein (VP9rdt) is 67.3 kDa (Table 20). The proteins of EYAV have not been characterized by translation.
In cells infected by CTFV, granular matrices are produced which contain virus-like particles. These structures appear similar to VIBs produced during orbivirus infections. In addition, bundles of filaments (tubules), characterized by cross-striations, are found in the cytoplasm and, in some cases, in the nucleus of infected cells. These may also be comparable to the tubules found in orbivirus infected cells. There is no evidence for virus release prior to cell death and lysis, after which more than 90% of virus particles remain cell associated. Immuno-fluorescence staining shows that viral proteins accumulate in the cytoplasm and could be detected from 12 h post infection. Nucleolar fluorescence was also observed. Mosquito cells infected by EYAV show syncitial foci. Electron microscopy of EYAV-infected mouse brain shows similar intracellular structures to those observed in CTFV-infected cells.
Table 20 Genome segments and protein products of Colorado tick fever virus
| Genome segment | Size (bp) | Protein nomenclature | Protein size (kDa)* | Protein structure/function |
| Seg1 | 4350 | VP1 | 125 (163) | RdRp |
| Seg2 | 3909 | VP2 | 117 (136) | Methyltransferase, cell-receptor |
| Seg3 | 3586 | VP3 | 113 (135) | RNA replication factors |
| Seg4 | 3157 | VP4 | 100 (112) |
|
| Seg5 | 2432 | VP5 | 90 (84) | Guanylyl transferase |
| Seg6 | 2141 | VP6 | 82 (78) | Nucleotide binding, NTPase |
| Seg7 | 2133 | VP7 | 75 (76) | RNA replication factors |
| Seg8 | 2029 | VP8 | 60 (74) |
|
| Seg9 | 1884 | VP9ter/VP9rdt | 42 and 60 (38 and 67) |
|
| Seg10 | 1880 | VP10 | 55 (69) | Kinase, helicase |
| Seg11 | 998 | VP11 | 34 (28.5) |
|
| Seg12 | 675 | VP12 | 25 (20.4) | RNA replication factors |
* As determined by translation, or calculated from nt sequences (in brackets).
Antigenic properties
There is little cross-reaction in virus neutralization tests between isolates of CTFV (from North America) and EYAV (from Europe). CTFV-Ca from a hare, collected in California in 1976, shows some one-way cross-reaction in serum neutralization tests with EYAV, but is clearly distinguishable. Distinct serotypes of both CTFV and EYAV have been reported.
Biological properties
Coltiviruses have been isolated from several mammalian species (including humans), as well as ticks and mosquitoes which serve as arthropod vectors. The tick species that have been implicated include Dermacentor andersoni, D. occidentales, D. albipictus, D. parumapertus, Haemaphysalis leporispalustris, Otobius lagophilus, Ixodes sculptus, I. spinipalpis, I. ricinus and I. ventalloi.
Ticks can become infected with CTFV after ingestion of a blood meal from an infected vertebrate host. Both adult and nymphal ticks become persistently infected and provide an overwintering mechanism for the virus. CTFV is transmitted trans-stadially but not trans-ovarially. Some rodent species have prolonged viraemia (more than 5 months) which may also facilitate overwintering and virus persistence. Humans most frequently become infected with CTFV when bitten by the adult wood tick D. andersoni but probably do not act as a source of re-infection for other ticks. Transmission from person to person has been recorded as the result of blood transfusion. The prolonged viraemia observed in humans and rodents is thought to be due to the intra-erythrocytic location of virions, protecting them from immune clearance.
CTFV is characterized in humans by an abrupt onset of fever, chills, headache, retro-orbital pains, photophobia, myalgia and generalized malaise. Abdominal pain occurs in about 20% of patients. Rashes are uncommon (less than 10%). A diphasic, or even triphasic, febrile pattern has been observed, usually lasting for 5–10 days. Severe forms of the disease, involving infection of the central nervous system, or hemorrhagic fever, or both, have been infrequently observed (nearly always in children under 12 years of age). A small number of such cases are fatal. Congenital infection with CTFV may occur, although the risk of abortion and congenital defects remains uncertain. Antibodies to EYAV have been found in patients with meningoencephalitis and polyneuritis but a causal relationship to the virus has not been established.
CTFV causes leucopoenia in adult hamsters and in about two-thirds of infected humans. Suckling mice, which usually die at 6–8 days post infection, suffer myocardial necrosis, necrobiotic cerebellar changes, widespread focal necrosis and perivascular inflammation in the cerebral cortex, degeneration of skeletal myofibers, hepatic necrosis, acute involution of the thymus, focal necrosis in the retina and in brown fat. The pathologic changes in mice due to CTFV infection (in skeletal muscle, heart and brain), are consistent with the clinical features of human infection, which may include meningitis, meningo-encephalitis, encephalitis, gastro-intestinal bleeding, pneumonia and myocarditis.
CTFV occurs in forest habitats at 4000–10,000 ft elevation in the Rocky Mountain region of North America. Antibodies to the virus have been detected in hares in Ontario and a virus isolate has been reported from Long Island, New York. EYAV appears to be widely distributed in Europe. An Eyach-like virus has been recently also been identified from ticks in the USA.
Species demarcation criteria in the genus
In addition to the other general criteria used throughout the family, members of a species in the genus Coltivirus may be identified by:
- RNA cross-hybridization assays: within a single species, RNA sequence that exhibit more than 74% similarity will hybridize at 36°C below the Tm of the fully base-paired duplex.
- Sequence analysis: Nucleotide identity of >89% in the conserved Seg12; amino acid identities of >55%, >57% and >60% respectively in VP6, VP7 and VP12 (the most variable proteins).
List of species in the genus Coltivirus
| Colorado tick fever virus (2 serotypes) {Ixodidae ticks : Rodents, humans } |
|
|
| Colorado tick fever virus - Florio | [Seg1: AF133428, Seg2: AF139758, Seg3: AF139759, Seg4: AF139760, Seg5: AF139761, Seg6: AF139762, Seg7: AF139763, Seg8: AF139764, Seg9: AF000720, Seg10: AF139765, Seg11: U72694, Seg12: U53227] | (CTFV-Fl) |
| California hare virus - California s6-14-03 | [Seg1: AF343051, Seg6: AF343054, Seg7: AF343057, Seg12: AF343060] | (CTFV-Ca) |
| Eyach virus {Ixodidae ticks : Possibly humans } |
|
|
| Eyach virus - Germany | [Seg1: AF343053, Seg6: AF343056, Seg7: AF343059, Seg12: AF343062] | (EYAV-Gr) |
| Eyach virus - France-577 | [Seg1: AF343052, Seg6: AF343055, Seg7: AF343058, Seg12: AF343061] | (EYAV-Fr577) |
| Eyach virus - France-578 | [Seg 1: AF282467, Seg 2: [AF282468] Seg3: AF282469, Seg4: AF282470, Seg5: AF282471, Seg6: AF282472, Seg7: AF282473, Seg8: AF282474, Seg9: AF282475, Seg10: AF282476, Seg11: AF282477, Seg12: AF282478] | (EYAV-Fr578) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ], arthropod vector and host names { } and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Coltivirus but have not been approved as species
| Salmon River virus* |
| (SRV) |
* Has been reported as a serotype distinct from CTFV.
Phylogenetic relationships within the genus and evolutionary links between coltiviruses and aquareoviruses
The level of aa identity between homologous proteins of CTFV and EYAV ranges from 55% to 88%, with similarity ranging between 72 and 93%. The most divergent proteins are those encoded by Seg6, Seg7 and Seg12 with aa identities of 55%, 57% and 63%, respectively. Highest identity values were found in VP1 (86–99%), which is the viral RNA-dependent RNA polymerase (Pol or RdRp).
The amino acid sequence (residues 370–490) of EYAV VP7 showed similarities (identity: 24%, similarity: 50%) to the sarcolemmal-associated protein of the European rabbit Oryctolagus cuniculus, a major host of EYAV. A corresponding region of the homologous VP6 protein of CTFV shows no significant match with this rabbit protein. This suggests that a recombination event has occurred between viral and host RNAs leading to the formation of a novel genome segment with mixed ancestry. However, the level of similarity (ca. 50%) to sarcolemmal-associated protein, suggests that this event occurred a very long time ago, possibly involving an ancestor of the European rabbit. Sequence analysis and phylogenetic studies suggest that EYAV is derived from an ancestral virus that was introduced into Europe during the migration of ancestors of the lagomorphs (hares, rabbits) from North America through Asia. Lagomorph ancestors first appeared during the Eocene epoch (57.8–36.6 MYA) in what was then North America. They are thought to have first migrated into Asia during the Oligocene epoch (34–23 MYA) and by the high Miocene epoch (23–25 MYA) they were common in Europe.
Sequence comparisons to other members of the family Reoviridae suggest that there has been an evolutionary jump, involving a change in the number of genome segments, between the aquareoviruses (11 segments) and coltiviruses (12 segments). Segments 7 of Aquareovirus C and G encode two proteins, from two distinct ORFs, which are homologues of two coltivirus proteins encoded separately by genome segments 9 and 12.
Subfamily Sedoreovirinae
Genus Orbivirus
Type species Bluetongue virus
Distinguishing features
Virions have a relatively featureless outer capsid as viewed by negative staining and electron microscopy and a genome composed of 10 segments of dsRNA. Core particles have characteristic ring-shaped capsomers. Replication is accompanied by production of viral tubules and viral inclusion bodies (VIB), and may be accompanied by formation of flat hexagonal crystals/arrays of the major outer core protein (VP7 (T13)) in the cytoplasm of infected cells. Viruses are transmitted between vertebrate hosts by a variety of hematophagous arthropods.
Virion properties
Morphology
Virions of bluetongue virus (BTV) are approximately 90 nm in diameter. Core particles have a maximum diameter of 73 nm, and sub-cores have a maximum diameter of 59 nm and an internal diameter of 46 nm (Figure 20). The virion is spherical in appearance but has icosahedral symmetry. Although mature virions lack a lipid envelope, they can leave the host cell by budding through the plasma membrane. During this process, they transiently acquire an unstable membrane envelope. Unpurified virus is often associated with cellular membranes. By conventional electron microscopy, the surface of intact virions is indistinct (Figure 20). However, the outer capsid does have an ordered structure, with icosahedral symmetry and sail-shaped surface projections that can be observed on virions where the particle structure is maintained (e.g. using cryoEM; Figure 21). When the outer capsid layer is removed, it is possible to view the surface layer of the core particle, which is composed entirely of capsomeres of VP7 (T=13) arranged as hexameric rings (pentameric at the five-fold axes; Figures 20 and 21). These rings, which are readily observed by conventional electron microscopy, give rise to the name of this genus. The core particle also contains a complete inner capsid shell (the subcore layer), which surrounds the 10 dsRNA genome segments and minor structural proteins. The minor core proteins (the transcriptase complexes) are attached to the inner surface of the subcore at the 5-fold symmetry axes (Figure 20). Assembly of the subcore layer appears to control the overall assembly, size and symmetry of the particle.
Physicochemical and physical properties
The virion Mr is about 10.8×107, and the core Mr is about 6.7×107. Their buoyant densities in CsCl are 1.36 g cm−3 (virions) and 1.40 g cm−3 (cores). The S20,W is 550S (virions) and 470S (cores). Virus infectivity is stable at pH 8–9 but virions exhibit a marked decrease in infectivity outside the pH range 6.5–10.2. In part, this may be related to the loss of outer coat proteins, particularly at the lower pH range. The sensitivity of the outer capsid proteins and their removal by cation treatment (e.g. by treatment with MgCl2, or CsCl) varies markedly with both pH and virus strain. At low pH values (less than 5.0), virions and cores are both disrupted. Unlike orthoreoviruses, virus infectivity is abolished at pH 3.0. In blood samples, serum or albumin, viruses held in vitro at less than 15 °C may remain infectious for decades. Purified BTV-1 virions held at 4 °C in 0.1 M Tris/HCl pH 8.0 showed no significant reduction in infectivity after 1 year. Crystals of core particles are very stable when kept at 29 °C. Virus infectivity is rapidly inactivated on heating to 60 °C. In general, orbiviruses are considered to be relatively resistant to treatment with solvents or detergents, although the sensitivity to specific detergents varies with virus species. However, sodium dodecyl sulfate will disrupt the particle and destroy its infectivity. Freezing reduces virus infectivity by about 90%, possibly due to particle disruption. However, once virus is frozen and held at −70 °C, infectivity remains stable.
Nucleic acid
The genomic RNA represents 12% or 19.5% of the total molecular mass of virus particles or cores, respectively. The genome is composed of 10 linear dsRNA segments that are packaged in exactly equimolar ratios, one of each segment per particle. The genomic RNA is packaged as a series of ordered concentric shells within the VP3 layer of the subcore (Figure 21). Four layers of RNA, each of which has elements of icosahedral symmetry, can be detected by X-ray crystallography of the BTV core. Within the central space of the subcore, there appears to be an association between the dsRNA molecules and the protein density at the five-fold axes of symmetry (at the vertices of the icosahedron), which is thought to represent the transcriptase complexes (TCs). From the five-fold axis, the RNA, in the outmost layer, appears to spiral away from the five-fold axes outward around the TC for two turns until it clashes with an icosahedrally related neighbor. At this point, it is thought to move inward forming the next concentric shell of RNA. The genomic RNA contains 5′-terminal Cap 1 structures (7mGpppG(2-Om)…).
For BTVs, the genome segments range in size from 3954 to 822 bp (total size is 19.2 kbp, total Mr of 13.1×106). There is no evidence for short ssRNA oligonucleotides in intact virions. The genomic RNAs are numbered in order of increasing electrophoretic mobility in 1% agarose gels and in order of decreasing Mw. For BTVs, the segments migrate as three size classes: three large (Seg1–3: 3.9–2.8 kbp), three medium (Seg4–6: 2.0–1.6 kbp) and four small segments (Seg6–10: 1.2–0.8 kbp). For other members of the genus, different sizes and size classes exist. For an individual virus species, the dsRNA sizes from different isolates or serotypes are usually comparable, such that a uniform segment migration pattern is observed when the genomic RNAs of normal isolates are analyzed by agarose gel electrophoresis. However, variations in primary sequence cause significant variations in rate and order of migration of genome segments during polyacrylamide gel electrophoresis (PAGE), particularly in high percentage gels (>5% polyacrylamide). Earlier BTV genome segment nomenclature based on PAGE is inconsistent, and the migration of Seg5 and Seg6 is often reversed. In the majority of the orbivirus genome segments that have been analyzed, there is only a single major ORF, which is always on the same strand (see conserved terminal sequences below). However, the ORF may have more than one functional initiation site near to the 5′ end of the RNA, resulting in production of two related proteins.
For BTV-10, the 5′-NTRs of the positive RNA strands range from 8 to 34 bp, and the 3′-NTRs are 31 to 116 bp in length. For other serotypes and viruses, the lengths may be different. However, in general the 5′-NTRs are shorter than the 3′-NTRs. The NTRs of almost all the orbivirus genome segments that have been sequenced (Table 21) contain two conserved base pairs at either terminus (+ve 5′-GU…AC-3′). The NTRs of BTV include terminal sequences of 6 bp that are usually identical for all 10 dsRNA segments (although some variation does occur) and that are conserved between different BTV isolates. Other orbiviruses have terminal sequences that are comparable to those of BTVs, but that are not always identical and that may not be conserved in all 10 segments (Table 21).
BTV mRNAs are usually regarded as non-infectious. However, fully functional and infectious viruses have been recovered by the introduction of all 10 mRNAs into BSR cells.
Table 21 Conserved terminal sequences (positive strand) of orbivirus genome segments
| Virus species | Strain | 5′ end | 3′ end |
| African horse sickness |
| 5′-GUU(A/U)A(A/U) | AC(A/U)UAC-3′ |
| Bluetongue virus |
| 5′-GUUAAA | (A/G)CUUAC-3′ |
| Chobar Gorge virus |
| 5′-GUUUA(A/U) | (A/G)(G/C)(A/C)UAC-3′ |
| Epizootic hemorrhagic disease virus |
| 5′-GUUAAA | (A/G)CUUAC-3′ |
| Equine encephalosis virus |
| 5′-GUU(U/A) | A (U/A/G)(A/U/C)GUUAC-3′ |
| Eubenangee virus |
| 5′-GU(U/A)(A/U)AA | (A/C)(U/A/C)UAC-3′ |
| Great Island virus |
| 5′-GUAAA(A/U)(A/U/C) | (A/G)(A/U/C)(C/G)(C/A/G)AC-3′ |
| Ieri virus* |
| 5′-GUU(U/A)AA | (A/G/C)(G/A/C)NUAC-3′ |
| Palyam virus |
| 5′-GU(A/U)AAA | (A/G)CUUAC-3′ |
| Peruvian horse sickness virus |
| 5′-GUUAAAA | (A/G)(C/G)(A/G)UAC-3′ |
| St Croix River virus |
| 5′-(A/G)UAAU(G/A/U) | (G/A/U)(C/U)(C/A)UAC-3′ |
| Umatilla virus |
| 5′-GUUU(A/U)A | A(G/A)GAUAC-3′ |
| Wad Medani virus |
| 5′-GU(A/U)(A/U)AA | N(G/A/C)CUAC-3′ |
| Yunnan orbivirus |
| 5′-GUUAAA(A/U) | N(G/A/C)(A/G)UAC-3′ |
| Warrego virus | MRV | 5′-GUAAA(A/U) | (A/C/U)C(U/A)UAC-3′ |
| Wallal virus | MUDV | 5′-GUAUA(A/U) | A(C/A)(A/G)C(U/A/C)UAC-3′ |
| (not classified) | ANDV** | 5′-GUUAAA | (A/U)CUUAC-3′ |
* Based on genome segments 3 to 10.
** Based on genome segments 2 to 10.
Proteins
There are seven virus structural proteins (VP1 to VP7; Table 22). These proteins constitute 88% and 80.5% of the dry weight of virions and cores, respectively. In BTV, the outer capsid consists of 180 copies of the 111 kDa sail-shaped VP2 protein arranged as trimeric triskelion structures, and 360 copies of an interdispersed and underlying VP5 protein (59 kDa), arranged as 120 trimers (Figures 20 and 21). The electrophoretic migration order and nomenclature of proteins may vary in members of other Orbivirus species. Both VP2 and VP5 of BTV are attached to VP7. The surface of the core particle consists entirely of 780 copies of VP7, which are arranged with T=13 l symmetry, as a network of hexameric and pentameric rings (in a near-perfect example of quasi-equivalence; Figure 21). The VP7 trimers of the core surface can bind dsRNA molecules, although the functional significance of this binding remains undetermined. Beneath the VP7 layer, the subcore capsid shell is composed of 120 copies of VP3 arranged with T=2 symmetry, displaying geometrical or pseudo quasi-equivalence (Figure 21). The VP3 (T2) capsid shell encloses the 10 dsRNA segments (Figure 21), as well as the three minor structural proteins. The latter include: the 150 kDa VP1(Pol), which is the RdRp; the 76 kDa VP4(Cap), which forms functional dimers and has both guanylyl-transferase and two methyl-transferase (Mtr) activities [Mtr 1 (forming the 7-methyl guanosine of the cap structure) and Mtr 2 (forming the 2-O-methyl guanosine, as the terminal nucleotide of the RNA chain)]; and the 36 kDa VP6/VP6a(Hel), which binds ssRNA or dsRNA and has both helicase and NTPase activities.
A 7 Å resolution of the structure of the infectious BTV virion (BTV-1SA) by cryoEM revealed structural information concerning the VP2 and VP5 outer coat proteins. The VP2 triskelion is composed of three tip domains branching from a central hub domain. The hub domain contains three putative sialic acid-binding pockets. Experimental data indicate that sugar-moiety-binding is important for BTV infection. The VP5 membrane penetration trimer, located between the VP2 trimers, has a central coiled-coil α-helical bundle, similar to the fusion proteins of many enveloped viruses. Weak interactions between the VP5 trimer and the VP2 trimer were detected in the cryoEM density map. Similar interactions were also detected with the underlying core surface layer of VP7 trimers. It has been suggested that the surface of VP5 could unfurl, like an umbrella, during penetration and shedding of the coat to release the transcriptionally active core particle.
X-ray diffraction studies indicate that the minor proteins are attached as a TC to the inner surface of the subcore layer [VP3 (T=2)] at the five-fold symmetry axes (at the vertices of the icosahedron). However, because there is only a single TC at each position, they do not have full icosahedral symmetry and it has not yet been possible to determine their organization at the atomic level.
The VP7 protein of some viruses, such as African horse sickness virus (AHSV), can also form flat hexagonal crystals/arrays, typically up to 5 μm in diameter, within the cytoplasm of the infected cell. These are composed of flat sheets of hexameric rings, which appear similar to the rings of trimers seen in the core-surface layer.
There are three distinct non-structural viral proteins produced in cells infected with BTV or other orbiviruses. The 64 kDa NS1(TuP) protein forms tubules that vary in length up to 4 μm. Although NS1 tubules have an unknown function, they are regarded as a characteristic feature of orbivirus replication. These tubules may have a ladder-like structure, as observed for BTV and epizootic hemorrhagic disease virus (EHDV) (68 and 52 nm in diameter), or they may be finer (23 nm in diameter) and have a reticular cross-weave pattern, as for AHSV.
The 41 kDa NS2(ViP) protein can be phosphorylated and is an important component of the matrix of VIBs, which are the site of virus replication and assembly. VIBs also contain relatively large amounts of the virus core proteins. NS2(ViP) consists of two domains joined by a hinge region and assembles into large multimeric complexes. NS2(ViP) has ssRNA-binding activity, suggesting that it plays an active role in replication. In conjunction with other virus proteins, it is believed to be involved in the recruitment of viral mRNA for encapsidation. The NS3/NS3a proteins are two small, non-structural membrane proteins (25 and 24 kDa) translated from different in-frame initiation sites on a single ORF, and are involved in the release of virus particles from cells. This function may be essential for dissemination of progeny virus, particularly from insect vector cells, which can become persistently infected and do not show cytopathic effect or high levels of cell death. In the process of particle release, the NS3 proteins are also released from the cell.
Table 22 Genome segments and protein products of bluetongue virus serotype 10
| Genome segment | Size (bp) | ORF (bp) | Protein* | Protein size (kDa) | Protein copy number/particle | Function (location) |
| Seg1 | 3954 | 12-3917 | VP1(Pol) | 149.6 | 10 | RdRp |
| Seg2 | 2926 | 20-2887 | VP2 | 111.1 | 180 | Outer layer of the outer capsid, controls virus serotype, cell attachment protein, involved in determination of virulence, readily cleaved by proteases. Most variable protein. Reacts with neutralizing antibodies. Trimer |
| Seg3 | 2770 | 18-2720 | VP3(T2) | 103.3 | 120 | Forms the innermost protein capsid shell subcore capsid layer, T=2 symmetry, controls overall size and organization of capsid structure, RNA binding, interacts with minor internal proteins |
| Seg4 | 2011 | 9-1970 | VP4(Cap) | 76.4 | 20 | Dimer, Mtr 1 and Mtr 2, capping enzyme (guanylyl-transferase) |
| Seg5 | 1769 | 35-1690 | NS1(TuP) | 64.4 | 0 | Forms tubules of unknown function in the cell cytoplasm. These are characteristic of orbivirus replication |
| Seg6 | 1638 | 30-1607 | VP5 | 59.2 | 360 | Inner layer of the outer capsid, may be glycosylated, helps determine virus serotype, variable protein. Trimer |
| Seg7 | 1156 | 18-1064 | VP7(T13) | 38.5 | 780 | Forms outer core surface, which can bind dsRNA, T=13 symmetry, in some species (AHSV) it can form flat hexagonal crystals, involved in cell entry and core particle infectivity in adults and cells of vector insects, reacts with “core neutralizing” antibodies, immuno dominant virus species specific antigen. Trimer |
| Seg8 | 1124 | 20-1090 | NS2(ViP) | 41.0 | 0 | Important viral inclusion body matrix protein, ssRNA binding, phosphorylated. May be associated with outer capsid |
| Seg9 | 1046 | 16-999 | VP6(Hel)VP6a | 35.8 | 60 | ssRNA and dsRNA binding, Helicase, NTPase |
| Seg10 | 822 | 20-706 | NS3 | 25.6 | 0 | Glycoproteins, membrane proteins, involved in cell exit. In some species (AHSV) these are variable proteins and are involved in determination of virulence |
|
|
|
| NS3a | 24.0 | 0 |
|
* Protein structure/function: Pol, RNA polymerase ; Cap, capping enzyme (guanylyltransferase and transmethylase); T2, inner virus structural protein with T=2 symmetry; T13, inner virus structural protein with T=13 symmetry; Hel, protein with helicase activity; ViP, viral inclusion body or viroplasm matrix protein; TuP, virus tubule protein. Viruses from other species within the genus may have proteins with significant differences in sizes.
Lipids
Orbivirus particles may be intimately associated with membraneous cell debris. Mature virions can acquire a membrane envelope by budding through the cell membrane during the process of cell exit, producing membrane enveloped virus particles (MEVPs). However, this membrane is thought to be transient or unstable and the virus particles are usually considered to be non-enveloped.
Carbohydrates
The BTV VP5 protein may be glycosylated. NS3 and NS3a synthesized in mammalian cells can become glycosylated, forming high molecular weight products.
Genome organization and replication
BTV genome segments are usually monocistronic but the Seg9 and Seg10 mRNAs are translated from either of two in-frame AUG codons. Coding assignments are shown in Table 22. The significance of the two forms of the Seg9 and Seg10 gene products (NS3, NS3A; VP6, VP6A) is not known. In some cases, other virus proteins form morphologically defined structures in infected cells [e.g. the flat hexagonal crystals/arrays formed of VP7 of AHSVs], but these are of unknown functional significance. Great Island virus was found to have two overlapping, out-of-phase ORFs, the longest of which codes for VP6(Hel). The second and shorter ORF codes for a 22 kDa protein (identified as VP6[dBP]), which has similarities to known dsRNA-binding proteins.
Virus adsorption involves components of the outer capsid, although cell entry may also involve VP7(T13). VP2 (and possibly also VP5) is involved in determination of virulence. VP5 may be involved in penetration of the cell membrane (release from endosomes into the cytoplasm), and the expressed protein can induce cell fusion. The outer capsid layer is lost during the early stages of replication. The transcription frequency of mRNA from individual genome segments varies, with more copies produced from the smaller segments.
Some details of the processes of virus replication are lacking. VIBs are considered to be the sites of morphogenesis of transcriptionally active virus cores containing dsRNA. The smallest particles containing RNA, which are observed in VIBs, appear to represent progeny subcore particles. The outer core protein [VP7] is added within the VIB and the outer-capsid proteins at the periphery of the VIB.
Virus particles are transported within the cell by specific interaction with the cellular cytoskeleton and can be released from the cell prior to lysis through interaction with membrane-associated NS3 proteins (Figure 2). There is also some evidence of specific association between NS1 tubules and intact virions in the cell cytoplasm. In most mammalian cells, replication of orbiviruses leads to shut-off of host protein synthesis and usually results in cell lysis and the release of virus particles. However, in persistently infected insect cells (or gamma delta T cells), there is no evidence for shut-off of host protein synthesis, extensive cell lysis or cytopathic effect. In some viruses (such as AHSV), NS3 is involved in determination of virulence for the mammalian host and, by controlling virus dissemination within the insect, may at least partially determine their ability to transmit the virus (vector competence). Virus particles can leave viable mammalian cells by two distinct mechanisms: extrusion (involving cell membrane damage) and budding. Only budding has been observed in cells of the BTV vector Culicoides variipennis, resulting in particles that have a membrane envelope, although this is unstable and is rapidly lost. Continuous release of virus particles from infected cells and re-infection appear to be features of orbivirus replication.
Antigenic properties
The main virus serogroup- (species-) specific antigen is the immunodominant outer core protein VP7. Monoclonal or polyclonal antibodies against VP7 can neutralize core particle infectivity, but do not attach to, or neutralize, undamaged virus particles or ISVPs in aqueous suspension, indicating that VP7 is not exposed on the intact virion surface. Other viral proteins are also conserved between virus species (in particular core proteins, NS1 and NS2). Some of these antigens may also show cross-reactions with viruses in other species, particularly those regarded as closely related. These cross-reactions are usually at a significantly lower level than with other viruses from the same virus species and may be one-way. Such relationships between species are also demonstrated by comparisons of the RNA sequences of conserved segments (for example, homologs of BTV Seg3, coding for inner core protein VP3. These data indicate that orbiviruses may be divided into at least four groups. Group A contains: AHSV, BTV, EHDV, equine encephalosis virus (EEV), Eubenangee virus (EUBV), Palyam virus (PALV), Wallal virus (WALV) and Warrego virus (WARV). Group B contains: Chenuda virus (CNUV), Ieri virus (IERIV), Wad Medani virus (WMV) and Great Island virus (GIV). Group C contains Corriparta virus (CORV). Group D contains Wongorr virus (WGRV). Insufficient comparisons have been made to conclusively assign all of the species of the genus Orbivirus to these groups.
Each species of the genus Orbivirus includes a number of serotypes that can be identified and distinguished in serum neutralization assays of intact virus particles (primarily via the specificity of interactions between neutralizing antibodies and the outer CPs). VP2 is the main neutralization antigen of BTV. VP5 is also involved in determination of virus serotype, possibly by imposing conformational constraints on VP2. The VP2 and VP5 proteins of BTV exhibit the greatest antigenic and sequence variation (Figure 22). In other viruses (such as GIV), the relative sizes of the outer CPs (VP4 and VP5) are very different and their individual roles may also be different. There is evidence that VP2 of BTV and AHSV (particularly in association with VP5) and VP7 can act as protective antigens.
In AHSV, the small nonstructural proteins, NS3 and NS3a, are also variable and may be divided into three groups (α, β and γ) based on sequence analysis. Preliminary serological evidence suggests that NS3 cross-reacts poorly between these groups. NS3 can also be involved in determination of virulence (for AHSV), possibly as a result of its involvement in release of virus particles from cells (budding) and its consequent effect on virus dissemination. Recent sequencing studies of BTV NS3 also indicate that it can be highly variable. NS3 variation does not correlate with virus serotype.
Biological properties
The specific infectivity of purified (disaggregated) BTV particles is equivalent to a particle infectivity ratio of approximately 1000:1 in both mammalian and insect cell systems. However, core particle infectivity varies from being 1000-fold less than that of intact virions (baby hamster kidney cells) to non-infectious (Chinese hamster ovary cells) in mammalian systems, depending on the cell line used. However, in some insect cells (KC cells, derived from Culicoides sonorensis) and adult vector insects, core particles are only slightly less infectious than intact virions (particle infectivity ratio of 1900:1). Treatment of virus with chymotrypsin or trypsin results in production of ISVPs, in which VP2 is cleaved. BTV ISVPs lack hemaglutinating activity, as well as the tendency to aggregate, but have a significantly elevated infectivity for adults of insect vectors and some insect cell lines (a particle infectivity ratio of approximately 13:1 for KC cells).
Different orbiviruses infect a wide range of vertebrate hosts including ruminants (domesticated and wild), equids (domesticated and wild), rodents, bats, marsupials, birds, sloths and primates, including humans. Orbiviruses can also replicate in, and are primarily transmitted by, arthropod vectors (gnats, mosquitoes, phlebotomines or ticks, depending on the virus). Trans-stadial transmission in ticks has been demonstrated for some viruses. Infection of vertebrates in utero may also occur. Orbiviruses, particularly those transmitted by short-lived vectors (gnats, mosquitoes and phlebotomines), are only enzootic in areas where adults of the competent vector species persist and are present all, or most, of the year. Orbiviral RNA has been detected in Culicoides larvae recovered from outbreak areas, although trans-ovarial transmission has not been confirmed by the recovery of infectious virus. Orbiviruses have also been detected in cell lines derived from tick eggs. BTV and EHDV are distributed worldwide between about 50° north and 30° south in the Americas and between 40° north and 35° south in the rest of the world. These limits have recently expanded to 53° north (e.g. in Europe), possibly as the result of climate change.
There is also evidence for persistence of these viruses over winter in the absence of overt disease. Mechanisms for persistence in the vertebrate host species even at low levels (including vertical transmission) may be of particular importance. Virus distribution also depends on the initial introduction into areas containing susceptible vertebrate hosts and competent vector species. For this reason, not all serotypes of each virus (e.g. BTV) are present at locations where some serotypes are endemic.
Orbivirus infection of arthropods has little or no evident effect. Infection in vertebrates can vary from inapparent to fatal, depending on both the virus and the host. Some BTV strains cause death in sheep, others cause a variety of pathologies, including hemorrhagic conditions, lameness, oedema, a transitory cyanotic appearance of the tongue (giving rise to the species name), nasal and mouth lesions, etc.; still others cause no overt pathology. BTV infection of cattle may show no signs of disease but can involve long-lived viraemias. AHSV, EHDV and EEV can cause severe pathology in their respective vertebrate hosts. Mortality rates in serologically naive populations can be over 98% (AHSV).
Species demarcation criteria in the genus
In addition to the other general criteria used throughout the family, members of a species in the genus Orbivirus may be identified by:
- High levels of serological cross reaction using either polyclonal sera or monoclonal antibodies against conserved antigens such as VP7. For example, in competition ELISA at a test serum dilution of 1/5, a positive serum will show >50% inhibition of color formation, while a negative control serum, or a serum that is specific for a different species, will normally produce <25% inhibition of color compared to a no antibody control. Distinct but related species may show low level serological cross-reaction, which may be only one-way.
- Sequence analysis: In the conserved Seg3 (encoding the major subcore structural protein, VP3), viruses within the same species will normally have >76% nucleotide identity while those in different species usally have <74% identity. These differences are also reflected in the amino acid sequences of the viral proteins.
- Relatively efficient cross-hybridization of conserved genome segments (those not encoding outer capsid components or other variable proteins) under high stringency conditions (>85% identity).
- Identification of common vector or host species and the clinical signs produced. For example BTV is transmitted only by certain Culicoides species and will infect cattle and sheep, producing clinical signs of varying severity, but is not thought to infect horses. The reverse is true of AHSV.
List of species in the genus Orbivirus
| African horse sickness virus (9 serotypes) {Culicoides: Equids, dogs, elephants, camels, cattle, sheep, goats, predatory carnivores and (in special circumstances) humans} |
|
|
| African horse sickness virus 1 | [Seg1:AM883164, Seg2:AM883165, Seg3: AM883166, Seg4: AM883167, Seg5: AM883168, Seg6: AM883169, Seg7: AM883170, Seg8: AM883171, Seg9: AM883172, Seg10: AM883173] | (AHSV1) |
| African horse sickness virus 2 | [Seg2: AY163332, Seg10: U59279] | (AHSV2) |
| African horse sickness virus 3 | [Seg2: U01832, Seg4: AF246225, Seg7: 545433, Seg8: AF545434, Seg9: U19881, Seg10: AJ007303] | (AHSV3) |
| African horse sickness virus 4 | [Seg2: U21956, Seg3: D26572, Seg4: D14402, Seg5: 11390, Seg6: M94731, Seg7: A27209, Seg10: U02712 ] | (AHSV4) |
| African horse sickness virus 5 | [Seg2: AY163331, Seg10: U60188] | (AHSV5) |
| African horse sickness virus 6 | [Seg2: AF021235, Seg3: AF021236, Seg5: U73658, Seg6: AF021237, Seg7: AF021238, Seg9: U33000, Seg10: U26171] | (AHSV6) |
| African horse sickness virus 7 | [Seg2: AY16330, Seg10: U60190] | (AHSV7) |
| African horse sickness virus 8 | [Seg2: AY163333, Seg10: U02713] | (AHSV8) |
| African horse sickness virus 9 | [Seg1: U94887, Seg2: AF043926, Seg5: U01069, Seg6: U74489, Seg7: S69829, Seg8: M69090, Seg10: D12480] | (AHSV9) |
| Bluetongue virus (26 serotypes) |
|
|
| Bluetongue virus 1 | [Seg2: AJ585122, Seg3: DQ186792, Seg5: M36713, Seg6 AJ586695, Seg7: X53740, Seg8: X58064, Seg9: D10905, Seg10: D00253] | (BTV1) |
| Bluetongue virus 2 | [Seg1: AJ585123, Seg2: M21946, Seg3: DQ186793, Seg4: L08637, Seg5: AM773705, Seg6: AJ586696, Seg7: M64997, Seg8: AY138896, Seg9: L08668, Seg10: AF1235224] | (BTV2) |
| Bluetongue virus 3 | [Seg2: AJ585124, Seg6: AJ586697 Seg7: AF172827, Seg10:AF135225] | (BTV3) |
| Bluetongue virus 4 | [Seg2: AJ585125, Seg3: DQ186794, Seg4: DQ186794, Seg6: AJ586699, Seg7: AF172828, Seg9: 403423, Seg10: AF135226] | (BTV4) |
| Bluetongue virus 5 | [Seg2:AJ585126, Seg6:AJ586700] | (BTV5) |
| Bluetongue virus 6 | [Seg1: GQ506498, Seg2:AJ585127, Seg3: GQ506499, Seg4: GQ506500, Seg5: GQ506501, Seg6: AJ586703, Seg7: GQ506502, Seg8: GQ506503, Seg9: GQ506504, Seg10: GQ506505] | (BTV6) |
| Bluetongue virus 7 | [Seg2:AJ585128, Seg6:AJ586704] | (BTV7) |
| Bluetongue virus 8 | [Seg1:AM498051, Seg2:AM498052, Seg3:AM498053, Seg4:AM498054, Seg5:AM498055, Seg6:AM498056, Seg7:AM498057, Seg8: AM498058, Seg9: AM498059, Seg10 AM498060] | (BTV8) |
| Bluetongue virus 9 | [Seg2:AJ585130, Seg3: DQ186795, Seg6:AJ586708] | (BTV9) |
| Bluetongue virus 10 | [Seg1:X12819, Seg2:M11787, Seg3:M22096, Seg4:Y00421, Seg5:Y00422, Seg6:D12532, Seg7:X06463, Seg8: D00500, Seg9: D00509, Seg10: AF044372] | (BTV10) |
| Bluetongue virus 11 | [Seg1:L20445, Seg2:AJ585132, Seg3:L19968, Seg4: L08638, Seg6:AJ586710, Seg7: M32102, Seg9:L08670, Seg10:L08631] | (BTV11) |
| Bluetongue virus 12 | [Seg2:AJ585133, Seg6:AJ586711, Seg7: AY263377, Seg10: AF135227] | (BTV12) |
| Bluetongue virus 13 | [Seg1:L20446, Seg2:AJ585134, Seg3: L19969, Seg4:L08640, Seg5:M97762, Seg6:AJ586713, Seg7:J04365, Seg9:L08671, Seg10:L08629] | (BTV13) |
| Bluetongue virus 14 | [Seg2:AJ585135, Seg6:AJ586714] | (BTV14) |
| Bluetongue virus 15 | [Seg2:AJ585136, Seg6:AJ586716, Seg7: L11723, Seg10: AF135228] | (BTV15) |
| Bluetongue virus 16 | [Seg2: AJ585137, Seg3: DQ186796, Seg5: AM773707, Seg6:AJ586719 Seg7: AF172831, Seg10: AF135229] | (BTV16) |
| Bluetongue virus 17 | [Seg1:L20447, Seg2:M17438, Seg3:AF017280, Seg4:L08639, Seg5:X17041, Seg6:X55359, Seg7: 53693, Seg8:P33473, Seg9:L08672, Seg10:L08630] | (BTV17) |
| Bluetongue virus 18 | [Seg2:AJ585139, Seg6:AJ586721, Seg10: AF512915] | (BTV18) |
| Bluetongue virus 19 | [Seg2: AJ585240, Seg6: AJ586722] | (BTV19) |
| Bluetongue virus 20 | [Seg2:AJ585141, Seg5: X56735, Seg6:AJ586723, Seg10: AF529055] | (BTV20) |
| Bluetongue virus 21 | [Seg2:AJ585142, Seg3:AF529047, Seg6:AJ586724, Seg8: AM900390 Seg10:AF529053] | (BTV21) |
| Bluetongue virus 22 | [Seg2:AJ585143, Seg6:AJ586725] | (BTV22) |
| Bluetongue virus 23 | [Seg2:AJ585144, Seg6:AJ586727, Seg7: AJ277802, Seg10: AF529051] | (BTV23) |
| Bluetongue virus 24 | [Seg2:AJ585145, Seg6:AJ586730] | (BTV-24) |
| Bluetongue virus 25 (Toggenburg orbivirus) | [Seg1: GQ982522, Seg2: EU839840, Seg3: GQ982523, Seg4: GQ982524, Seg5: EU839841, Seg6: EU839842, Seg7: EU839843, Seg8: EU839844, Seg9: EU839845, Seg10: EU839846] | (BTV-25) |
| Bluetongue virus 26 |
| (BTV-26) |
|
|
|
|
| Changuinola virus (12 serotypes) {phlebotomines, culicine mosquitoes: humans, rodents, sloths} |
|
|
| Almeirim virus |
| (ALMV) |
| Altamira virus |
| (ALTV) |
| Caninde virus |
| (CANV) |
| Changuinola virus |
| (CGLV) |
| Gurupi virus |
| (GURV) |
| Irituia virus |
| (IRIV) |
| Jamanxi virus |
| (JAMV) |
| Jari virus |
| (JARIV) |
| Monte Dourado virus |
| (MDOV) |
| Ourem virus |
| (OURV) |
| Purus virus |
| (PURV) |
| Saraca virus |
| (SRAV) |
| Chenuda virus (7 serotypes) {ticks: seabirds} |
|
|
| Baku virus |
| (BAKUV) |
| Chenuda virus |
| (CNUV) |
| Essaouira virus |
| (ESSV) |
| Huncho virus |
| (HUAV) |
| Kala Iris virus |
| (KIRV) |
| Mono Lake virus |
| (MLV) |
| Sixgun city virus |
| (SCV) |
|
|
|
|
| Chobar Gorge virus (2 serotypes) {ticks: bats} |
|
|
| Chobar Gorge virus |
| (CGV) |
| Fomede virus |
| (FV) |
| Corriparta virus (6 serotypes/strains*) {culicine mosquitoes: humans, rodents} |
|
|
| Acado virus |
| (ACDV) |
| Corriparta virus (CS109) |
| (CORV-CS109) |
| Corriparta virus (V654) |
| (CORV-V654) |
| Corriparta virus (V370) |
| (CORV-V370) |
| Corriparta virus MRM1 | [Seg3: AF530086] | (CORV-MRM1) |
| Jacareacanga virus |
| (JACV) |
| Epizootic hemorrhagic disease virus (7 serotypes/strains*) {Culicoides: cattle, sheep, deer, camels, llamas, wild ruminants, marsupials} |
|
|
| Epizootic hemorrhagic disease virus 1 | [Seg1:AM744977, Seg2: AM744978, Seg3:AM744979, Seg4:AM744980, Seg5:AM744981, Seg6: AM744982, Seg7:AM744983, Seg8: AM744984, Seg9:AM744985,Seg10: AM744986] | (EHDV1) |
| Epizootic hemorrhagic disease virus 2 | [Seg1:AM744997, Seg2: AM744998, Seg3: AM744999, Seg4: M745000, Seg5: AM745001, Seg6: AM745002, Seg7: AM745003, Seg8: AM745004, Seg9: AM745005, Seg10: AM745006] | (EHDV2) |
| Ibaraki virus (EHDV-2) | [Seg1: AM745077, Seg2: AM745078, Seg3: AM745079, Seg4: AM745080, Seg5: AM745081, Seg6: AM745082, Seg7: AM745083, Seg8: AM745084, Seg9: AM745085, Seg10, AM745086] | (IBAV) |
| Epizootic hemorrhagic disease virus 3 (Regrouped as EHDV-1) | [Seg1: AM745007,Seg2: AM745008, Seg3: AM745009, Seg4: AM745010, Seg5: AM745011, Seg6:AM745012, Seg7: AM745013, Seg8: AM745014, Seg9: AM745015, Seg10: AM745016] | (EHDV3) |
| Epizootic hemorrhagic disease virus 4 | [Seg1: AM745017, Seg2: AM745018, Seg3: AM745019, Seg4: AM745020, Seg5: AM745021, Seg6: AM745022, Seg7: AM745023, Seg8: AM745024, Seg9: AM745025, Seg10, AM745026] | (EHDV4) |
| Epizootic hemorrhagic disease virus 5 | [Seg1: AM745027, Seg2: AM745028, Seg3: AM745029, Seg4: AM745030, Seg5: AM745031, Seg6: AM745032, Seg7: AM745033, Seg8: AM745034, Seg9: AM745035, Seg10: AM745036] | (EHDV5) |
| Epizootic hemorrhagic disease virus 6 | [Seg1: AM745037, Seg2: AM745038, Seg3:AM745039, Seg4: AM745040, Seg5 AM745041, Seg6: AM745042, Seg7: AM745043, Seg8: AM745044, Seg9: AM745045, Seg10: AM745046]
| (EHDV6) |
| Epizootic hemorrhagic disease virus 318 (EHDV-6) | [Seg1:AM745067, Seg2: AM745068, Seg3: AM745069, Seg4: AM745070, Seg5: AM745071, Seg6: AM745072, Seg7: AM745073, Seg8: AM745074, Seg9: AM745075, Seg10, AM745076]
| (EHDV-318) |
| Epizootic hemorrhagic disease virus 7 | [Seg1: AM745047, Seg2: AM745048, Seg3: AM745049, Seg4: AM745050, Seg5: AM745051, Seg6: AM745052, Seg7: AM745053, Seg8:AM745054, Seg9: AM745055, Seg10: AM745056] | (EHDV7) |
| Epizootic hemorrhagic disease virus 8 | [Seg1: AM745057, Seg2:AM745058, Seg3: AM745059, Seg4: AM745060, Seg5: AM745061, Seg6: AM745062, Seg7: AM745063, Seg8: AM745064, Seg9: AM745065, Seg10: AM745066] | (EHDV8) |
|
|
|
|
| Equine encephalosis virus (7 serotypes) {Culicoides: equids} |
|
|
| Equine encephalosis virus 1 | [Seg1: FJ183384, Seg2: FJ183385, Seg3: FJ183386, Seg4: FJ183387, Seg5: FJ183388, Seg6: FJ183389, Seg7: FJ183391, Seg8: FJ183390, Seg9: FJ183392, Seg10: FJ183393] | (EEV-1) |
| Equine encephalosis virus 2 | [Seg10: AY115871] | (EEV-2) |
| Equine encephalosis virus 3 | [Seg10: AY115874] | (EEV-3) |
| Equine encephalosis virus 4 | [Seg10: AY115868] | (EEV-4) |
| Equine encephalosis virus 5 | [Seg10: AY115869] | (EEV-5) |
| Equine encephalosis virus 6 | [Seg10: AY115873] | (EEV-6) |
| Equine encephalosis virus 7 | [Seg10: AY115870] | (EEV-7) |
| Eubenangee virus (4 serotypes) {Culicoides, anopheline and Culicine mosquitoes: unknown hosts} |
|
|
| Eubenangee virus | [Seg3: AF530087] | (EUBV) |
| Ngoupe virus |
| (NGOV) |
| Pata virus |
| (PATAV) |
| Tilligerry virus |
| (TILV) |
| Great Island virus (36 serotypes / strains*) {Argas, Ornithodoros, Ixodes ticks: seabirds, rodents, humans} |
| (GIV) |
| Above Maiden virus |
| (ABMV) |
| Arbroath virus |
| (ABRV) |
| Bauline virus |
| (BAUV) |
| Broadhaven virus | [Seg2: M87875, Seg5: M58030, Seg6: X82599, Seg7: M87876, Seg10: M83197] | (BRDV) |
| Cape Wrath virus |
| (CWV) |
| Colony virus |
| (COYV) |
| Colony B North virus |
| (CBNV) |
| Ellidaey virus |
| (ELLV) |
| Foula virus |
| (FOUV) |
| Great Island virus | [Seg1: HM543465, Seg2: HM543466, Seg3: HM543467, Seg4: HM543468, Seg5: HM543469, Seg6: HM543470, Seg7: HM543471, Seg8: HM543472, Seg9: HM543473, Seg10: HM543474] | (GIV) |
| Great Saltee Island virus |
| (GSIV) |
| Grimsey virus |
| (GSYV) |
| Inner Farne virus |
| (INFV) |
| Kemerovo virus | [Seg1: HM543481, Seg2: HM543482, Seg6: HM543483] | (KEMV) |
| Kenai virus |
| (KENV) |
| Kharagysh virus |
| (KHAV) |
| Lipovnik virus | [Seg1: HM543475, Seg2: HM543476, Seg6: HM543477] | (LIPV) |
| Lundy virus |
| (LUNV) |
| Maiden virus |
| (MDNV) |
| Mill Door virus |
| (MDRV) |
| Mykines virus |
| (MYKV) |
| North Clett virus |
| (NCLV) |
| North End virus |
| (NEDV) |
| Nugget virus |
| (NUGV) |
| Okhotskiy virus |
| (OKHV) |
| Poovoot virus |
| (POOV) |
| Rost Island virus |
| (RSTV) |
| St Abb's Head virus |
| (SAHV) |
| Shiant Islands virus |
| (SHIV) |
| Thormodseyjarlettur virus |
| (THRV) |
| Tillamook virus |
| (TLMV) |
| Tindholmur virus |
| (TDMV) |
| Tribec virus | [Seg1: HM543478, Seg2: HM543479, Seg6: HM543480] | (TRBV) |
| Vearoy virus |
| (VAEV) |
| Wexford virus |
| (WEXV) |
| Yaquina Head virus |
| (YHV) |
| Ieri virus (3 serotypes) {mosquitoes: birds} |
|
|
| Ieri virus |
| (IERIV) |
| Gomoka virus |
| GMKV) |
| Arkonam virus |
| (ARKV) |
| Lebombo virus (1 serotype) {culicine mosquitoes: humans, rodents} |
|
|
| Lebombo virus 1 |
| (LEBV-1) |
| Orungo virus (4 serotypes) {culicine mosquitoes: humans, camels, cattle, goats, sheep, monkeys} |
|
|
| Orungo virus 1 |
| (ORUV1) |
| Orungo virus 2 |
| (ORUV2) |
| Orungo virus 3 |
| (ORUV3) |
| Orungo virus 4 |
| (ORUV4) |
| Palyam virus (13 serotypes / strains*) {Culicoides, culicine mosquitoes: Cattle, sheep} |
|
|
| Abadina virus |
| (ABAV) |
| Bunyip creek virus | [Seg5: AB034595, Seg7: AB034671, Seg9: AB034681] | (BCV) |
| CSIRO village virus | [Seg5: AB034594, Seg7: AB034670, Seg9: AB034680] | (CVGV) |
| D'Aguilar virus | [Seg3: AF530085, Seg5: AB034593, Seg7: AB034666, Seg9: AB034676] | (DAGV) |
| Gweru virus | [Seg5: AB034598, Seg7: AB034674, Seg9: AB034683] | (GWV) |
| Kasba virus (Chuzan virus) | [Seg1: AB018086, Seg2: AB014725, Seg3: AB014728, Seg4: AB018087, Seg5: AB018089, Seg6: AB014726, Seg7: AB014727, Seg8: AB018090, Seg9: AB018088, Seg10: AB018091] | (KASV) |
| Kindia virus |
| (KINV) |
| Marrakai virus | [Seg5: AB034592, Seg7: AB034668, Seg9: AB034678] | (MARV) |
| Marondera virus | [Seg5: AB034597, Seg7: AB034673] | (MRDV) |
| Nyabira virus | [Seg5: AB034596, Seg7: AB034672, Seg9: AB034682] | (NYAV) |
| Palyam virus |
| (PALV) |
| Petevo virus |
| (PETV) |
| Vellore virus |
| (VELV) |
| Peruvian horse sickness virus (1 serotype) { mosquitoes: horses } |
|
|
| Peruvian horse sickness virus - 1 | [Seg1: DQ248057, Seg2: DQ248058, Seg3: DQ248059, Seg4: DQ248060, Seg5: DQ248064, Seg6: DQ248061, Seg7: DQ248065, Seg8: DQ248063, Seg9: DQ248062, Seg10: DQ248066] | (PHSV-1) |
| St Croix River virus {ticks: unknown hosts} |
|
|
| St Croix River virus -1 | [Seg1: AF133431, Seg2: AF133432, Seg3: AF145400, Seg4: AF145401, Seg5: AF145402, Seg6: AF145403, Seg7: AF145404, Seg8: AF145405, Seg9: AF145406, Seg10: AF145407] | (SCRV-1) |
| Umatilla virus (4 serotypes) {culicine mosquitoes: birds} |
|
|
| Llano Seco virus |
| (LLSV) |
| Minnal virus |
| (MINV) |
| Netivot virus |
| (NETV) |
| Umatilla virus |
| (UMAV) |
| Wad Medani virus (2 serotypes) {Boophilus, Rhipicephalus, Hyalomma, Argas ticks: domesticated animals} |
|
|
| Seletar virus |
| (SELV) |
| Wad Medani virus |
| (WMV) |
| Wallal virus (3 serotypes/strains*) {Culicoides: marsupials} |
|
|
| Mudjinbarry virus |
| (MUDV) |
| Wallal virus | [Seg3: AF530084] | (WALV) |
| Wallal K virus |
| (WALKV) |
| Warrego virus (3 serotypes/strains*) {Culicoides, anopheline and culicine mosquitoes: marsupials} |
|
|
| Mitchell river virus |
| (MRV) |
| Warrego virus | [Seg3: AF530083] | (WARV) |
| Warrego K virus |
| (WARKV) |
| Wongorr virus (8 serotypes/strains*) {Culicoides, mosquitoes: Cattle, macropods} |
| (WGRV) |
| Paroo river virus | [Seg3: U56993] | (PRV) |
| Picola virus | [Seg3: U56994] | (PIAV) |
| Wongorr virus: MRM13443 | [Seg3: U56992] | (WGRV-MRM13443) |
| Wongorr virus CS131 |
| (WGRV-CS131) |
| Wongorr virus V195 | [Seg3: U56990] | (WGRV-V195) |
| Wongorr virus V199 | [Seg3: U56991] | (WGRV-V199) |
| Wongorr virus V595 |
| (WGRV-V595) |
| Wongorr virus V1447 | [Seg3: U56989] | (WGRV-V1447) |
| Yunnan orbivirus (2 serotypes) {Mosquitoes: Cattle, sheep, donkeys} |
|
|
| Yunnan orbivirus 1 | [Seg1: AY701509, Seg2: AY701510, Seg3: AY701511, Seg4: AY701512, Seg5: AY701513, Seg6: AY701514, Seg7: AY701515, Seg8: AY701516, Seg9: AY701517, Seg10: AY701518]
| (YUOV-1) |
| Yunnan orbivirus 2 (Middle point orbivirus) | Seg2: EF591620, Seg3: EF591621]
| (YUOV-2)
|
Species names are in italic script; names of isolates are in roman script. Sequence accessions [ ], arthropod vector and host names { } and assigned abbreviations ( ) are also listed.*In some species the serological relationship between strains has not been fully determined.
List of other related viruses which may be members of the genus Orbivirus but have not been approved as species
| Andasibe virus {mosquitoes: unknown hosts} |
| (ANDV) |
| Codajas virus {mosquitoes: rodents} |
| (COV) |
| Ife virus {mosquitoes: rodents, birds, ruminants} |
| (IFEV) |
| Itupiranga virus {mosquitoes: unknown hosts} |
| (ITUV) |
| Japanaut virus {mosquitoes: unknown hosts} |
| (JAPV) |
| Kammavanpettai virus {unknown vectors: birds} |
| (KMPV) |
| Lake Clarendon virus {ticks: birds} |
| (LCV) |
| Matucare virus {ticks: unknown hosts} |
| (MATV) |
| Tembe virus {mosquitoes: unknown hosts} |
| (TMEV) |
| Tracambe virus {mosquitoes: unknown hosts} |
| (TRV) |
Phylogenetic relationships within the genus
The phylogenetic relationships within the genus are illustrated in Figures 22 and 23 (pp. 604 and 605).
Genus Rotavirus
Type species Rotavirus A
Distinguishing features
Rotaviruses infect only vertebrates and are transmitted by a fecal–oral route. When viewed by negative contrast electron microscopy (Figure 24), virus particles have a wheel-like appearance from which the genus derives its name (Latin rota, “wheel”). The triple-layered capsid encloses a genome of 11 linear dsRNA segments and is formed in a unique morphogenic pathway, which involves acquisition of a transient lipid envelope during budding of immature particles into the endoplasmic reticulum (ER).
Virion properties
Morphology
The data for simian rotavirus A/SA11 (SiRV-A/SA11) represent a paradigm for other viruses within the genus. The mature infectious virion has an overall diameter of about 100 nm, is made up of three concentric protein layers and lacks a lipid-containing envelope (Figure 24). The detailed topology of these layers and their protein components has been revealed using cryoEM, followed by image processing of viral and subviral particles, as well as virus-like particles formed using recombinant baculoviruses that express specific rotavirus structural proteins (Figure 25). The innermost layer of the virion, composed of VP2, is about 3.5 nm thick. This layer is comparable to the internal capsid layer of members of other genera within the family Reoviridae (e.g. the VP3 (T2) layer of the orbiviruses). The VP2 layer (T=1; 60 asymmetric dimers of VP2) surrounds the genomic dsRNAs and two structural proteins, the RdRp VP1 and the capping enzyme VP3, which are organized as a series of up to 12 enzymatic complexes, tethered to the inner surface of VP2 near the five-fold axes of symmetry (Figures 24 and 25).
The assembled genomic dsRNAs, along with the enzymatic complexes and VP2 layer, comprise the rotavirus core, which has a diameter of about 51 nm. Each of the two outer layers of the rotavirus virion is organized with T=13 (laevo) icosahedral symmetry. Spanning these layers is a uniquely characteristic set of 132 large channels that link the outer surface with the inner VP2 protein layer. The intermediate capsid layer is composed of 780 copies of VP6, arranged as 260 trimeric morphological units positioned at the local and strict three-fold axes of the icosahedral lattice (Figure 25). The VP6 layer forms the outer surface of the double-layered particle (DLP; ca. 70.5 nm in diameter) and is directly comparable to one of the capsid layers of viruses of some other genera within the Reoviridae (e.g. the VP7 (T13) layer of the orbiviruses). Two proteins (VP4 and VP7) form the outermost layer of the rotavirus virion (ca. 75 nm in diameter, not including spikes) and are required for infectivity (Figure 25). The glycoprotein VP7 makes up the surface of the outermost shell, which is arranged as 260 trimers stabilized by Ca2+ bound in the inter-subunit interfaces. VP7 trimers cap the trimeric pillars of VP6 and grip them with amino-terminal arms. Projecting from the VP7 layer are 60 trimeric spikes formed by VP4. Trypsin cleavage of VP4 generates amino-terminal (VP8*) and carboxyl-terminal (VP5*) fragments of VP4 and primes virions for infectivity. Primed VP4 spikes are about 20 nm long and extend about 12 nm from the surface of the outer VP7 layer, giving a final maximum particle diameter of about 100 nm. The foot of the VP4 spike has three-fold symmetry and interacts extensively with VP6. Distal to the foot, the spike lacks three-fold symmetry. A stalk formed primarily by VP5* connects the foot to a paired body, atop which two heads formed by the VP8* receptor-binding domain sit. Each VP5* monomer contains a β-barrel domain, tipped by a hydrophobic apex, which is buried in the cleaved spike. Similar to enveloped virus fusion proteins, during entry VP4 undergoes a rearrangement that is linked to membrane penetration. The hydrophobic apices of VP5* are exposed as the β-barrel domains fold back and a trimeric coiled-coil zips up.
Physicochemical and physical properties
The rotavirus virion has a density of 1.36 g cm−3 in CsCl and sediments at 520–530S in sucrose. Virus infectivity is dependent upon the presence of the VP4-VP7 outermost protein layer, the integrity of which requires Ca2+. Treatment of virions with Ca2+-chelating agents, such as EGTA or EDTA, destabilizes VP7 trimers, leading to loss of the outer capsid. Infectivity is not affected by exposure of infectious virions to pH ranges from 3 to 9 and, in the presence of 1.5 mM CaCl2, by storage for months at 4 °C or even 20 °C. Infectivity is also relatively thermostable at 50 °C but can be lost by repeated cycles of freezing and thawing. Infectivity is generally resistant to fluorocarbon extraction, treatment with solvents such as ether and chloroform, or non-ionic detergents such as deoxycholate, all of which reflect the absence of a lipid-containing envelope on the mature particle. However, infectivity is lost by treatment with sodium dodecyl sulfate (0.1%), or a number of disinfectants such as betapropiolactone, chlorine, formalin and phenols. In addition, 95% ethanol, which disrupts the outer protein layer of the virion, represents an effective disinfectant. The VP4-mediated hemagglutinin activity of the infectious virion is lost rapidly at 45 °C or as a result of freezing and thawing. Some variation has been observed in the physicochemical properties and stability of intact virions of different rotavirus strains. For example, not all human rotaviruses exhibit hemagglutinin activity, and they tend to lose the proteins of their outer layer more easily than some animal strains. In re-assortment studies, some of this variation has been attributed to the parental origin of the VP4 component in the virus particle.
Double-layered particles (DLPs) are non-infectious, have a density of 1.38 g cm−3 in CsCl and sediment at 380–400S in sucrose. These are equivalent to cores of the orbiviruses. Single-layered rotavirus particles (cores) can be produced by treatment of DLPs with either chaotropic agents such as sodium thiocyanate or high concentrations of CaCl2. Cores have a density of 1.44 g cm−3 in CsCl, sediment at 280S in sucrose, and are readily disrupted by incubation in hypotonic solutions, and are structurally equivalent to sub-cores of the orbiviruses.
Nucleic acid
The rotavirus genome consists of 11 discrete segments of linear dsRNA, which are organized with dodecahedral symmetry in the core. For SiRV-A/SA11, the segments range in size from 3302 to 667 bp and have a combined size of 18,555 bp. When resolved by gel electrophoresis, the segments of Rotavirus A strains (not including isolates from avian species) typically display a 4:2:3:2 distribution pattern. The genome segments are numbered segment 1 (Seg1) to segment-11 (Seg11) in order of increasing mobility during electrophoresis, although the migration order of cognate segments, particularly in the Seg7 to Seg9 triplet, does vary. In some Rotavirus A isolates, deviation from the 4:2:3:2 RNA migration pattern is indicative of a concatemerization event, in which up to an additional 1800 bp is packaged into viable virions, due to the partial duplication of a genome segment. Rotavirus genomic RNA sequences are A+U rich (58–67%). The segments are completely base-paired, and the plus-strand contains a 5′-terminal cap structure (m7GpppG(m)GC), but lacks a polyadenylation signal near its 3′ end. In contrast to members of the genus Orthoreovirus, there is no evidence for the presence of single-stranded oligonucleotides within the virion. Two levels of terminal sequence conservation are evident in Rotavirus A isolates. Firstly, all genome segments share short, conserved 5′ and 3′ termini. The 5′-terminal conserved region has a consensus sequence of 5′-(GGCUUUUAAA…)-3′, and the 3′ terminus has the consensus sequence 5′-(…AUGUGACC)-3′ (Table 23). Immediately internal to these terminal regions at each end of the different segments, there is a second region of conservation of at least 30–40 nt, which is segment-specific. These two levels of sequence conservation may be indicative of cis-acting signals that are important for controlling transcription, replication and segment selection for packaging. The 5′ nontranslated regions (NTRs) vary in length, but are typically less than 50 nt and are followed by at least one long ORF beginning with the first AUG. Some segments contain additional in-frame (Seg7, Seg9 and Seg10) or out-of-frame (Seg11) ORFs. However, only in the case of Seg9 and Seg11 are alternate start codons used to initiate synthesis of more than a single primary translational product from each segment. The 3′ NTRs vary in length from 17 nt (Seg1) to 182 nt (Seg10).
Detailed genomic information for Rotavirus B, Rotavirus C, Rotavirus D and novel adult diarrhea rotaviruses (NADRVs) is sparse. However, complete sequence information is available for all genome segments of at least one isolate for each species. In all cases, the genome is made up of 11 segments, which have broadly similar properties to SiRV-A/SA11 in terms of length, presence of ORFs and conserved terminal sequences.
Table 23 Conserved terminal sequences (positive strand) of rotavirus genome segments
| Virus species | Strain | 5′ end | 3′ end |
| Rotavirus A | SA11 | 5′-GGC(U/A)2U(A/U)4 | (A/U)U(G/A)UG(A/G)CC-3′* |
| Rotavirus B | WH-1 | 5′-GG(U/C)(A/U)N(A/U)5** | (A/U)3(A/G)2A(C/A)CC-3′ |
| Rotavirus C | Bristol | 5′-GCC(A/U)7 | UGUGGCU-3′ |
| Rotavirus D | 05V0049 | 5′-GG(U/C)( A/U)4AA(A/U) | (U/A/C)U(G/A/U)(U/C)GACC-3′ |
| (not classified) | NADRV-J19 | 5′-GG(CAU)A(CAU) | (A/G)UA(U/C)ACCC-3′ |
* For segment 5, the 3′-terminal sequence is CUGUGAACC.
** Rarely, a G or C is found at position 6, 8, or 10.
Proteins
Thirteen primary gene products have been defined for SiRV-A/SA11. The majority of genome segments encode a single protein, but two segments (Seg9 and Seg11) each encode two primary translation products. In the case of Seg9, two initiation codons in the same reading frame may be used, giving largely overlapping forms of the protein product VP7. Gene 11 contains two out-of-frame ORFs, translation of which results in two unrelated non-structural proteins, NSP5 and NSP6 (Table 24).
The nomenclature system employed for rotavirus proteins numbers them according to their migration rates upon SDS-PAGE, starting with the slowest (i.e. highest molecular weight). Structural proteins are given the prefix “VP”, whereas non-structural proteins are given the prefix “NSP”. Six structural proteins have been identified, and their approximate locations within the virion have been defined. The viral core, which encapsidates the dsRNA genome, is composed of three proteins. Two of the core proteins (VP1 and VP3) are directly associated with the genome, while the third (VP2) makes up the core shell, the innermost protein layer of the capsid. VP1, the largest viral protein (125 kDa), is a four-tunneled RdRp, responsible for both transcription and replication. Transcriptional activity can be detected in preparations of purified DLPs. Genome replication (negative strand synthesis) can be achieved in vitro using disrupted viral core preparations or recombinant VP1 protein. However, VP1 is active only in the presence of VP2. Viral core preparations also exhibit guanylyltransferase and methyltransferase activities. The VP3 component (98 kDa; the capping enzyme) of DLPs, binds ssRNA and forms adducts with GTP and S-adenosyl-l-methionine. VP1 and VP3 form a complex that is tethered near the inward protruding hubs at the five-fold vertices of the VP2 capsid layer (Figure 25). The amount of VP1 and VP3 in the virion is known to be low (12 molecules/particle) but has not been measured precisely. VP2 (102 kDa) is the most abundant protein of the viral core, with 120 molecules per virion. VP2 is required for encapsidation of VP1 and VP3 and has nonspecific ssRNA- and dsRNA-binding activity. By analogy with other reoviruses, it is likely that VP2 determines both the size of the particle as well as the structural organization of the outer capsid components, the internal enzymatic components and the RNAs. This important functional load is reflected in the highly conserved nature of VP2.
The intermediate protein layer of the virion is made up of 260 trimers of VP6 (45 kDa). The two remaining structural proteins of the virion, VP4 (87 kDa) and VP7 (36 kDa), of which there are 60 trimers and 260 trimers per virion, respectively, make up the outermost protein layer (Figure 25). The spike protein VP4 contains a trypsin cleavage site approximately one-third of the way along its length. Cleavage of the protein in vitro by treatment with trypsin produces two products, VP5* (60 kDa) and VP8* (28 kDa), enhances virus infectivity, and induces conformational changes that stabilize the spike structure. The VP8* cleavage product has hemagglutinin activity and contains a carbohydrate binding site (galectin-like fold). VP7 is the primary component of the outer shell of the virion. The VP7 glycoprotein is synthesized on the rough ER (RER) and co-translationally inserted in the ER membrane. As indicated earlier, Seg9 contains two in-frame initiation codons, both of which may be used to generate protein products. However, post-translational cleavage results in removal of the amino terminus of both forms of VP7, yielding identical protein products that are incorporated into the virion.
Six non-structural proteins are encoded by the viral genome. NSP1 (59 kDa), encoded by Seg5, is the most variable of all the rotavirus proteins within a single species, with as much as 65% sequence diversity observed between strains of Rotavirus A. The total length of this genome segment, as well as the size of the ORF, can vary among strains. Although highly variable overall, NSP1 has a conserved cysteine-rich motif near the amino terminus, which is organized in a manner characteristic of zinc-binding RING-domain proteins. NSP1 has been shown to bind both the 5′ end of viral RNA and zinc. As indicated by studies of rotavirus reassortants in a mouse model of infection, NSP1 has a role in viral pathogenesis and is a virulence determinant. NSP1 antagonizes the host innate immune response by inducing the degradation of interferon regulatory factors (IRF3, IRF5 and IRF7) required for expression of type I interferon, possibly by acting as an E3 ubiquitin ligase. NSP1 may also play a role in host range restriction, suggesting that its activity may vary depending on the host species.
NSP2 (35 kDa) contains a HIT-like fold and self-assembles into a stable octamer, the functional form of the protein. It binds nonspecifically to ssRNA, has NTPase, RTPase and helix destabilizing activities, and interacts with NSP5 to form viroplasms (equivalent to VIBs generated by members of other genera within the family Reoviridae). Viruses that contain temperature-sensitive mutations in Seg8, which encodes NSP2, have an RNA-negative phenotype at the non-permissive temperature, indicating that NSP2 has a direct role in the mechanism of virus replication. It has been hypothesized that NSP2 functions as a molecular motor and plays an important role in viral RNA packaging.
NSP3 (34 kDa) is a multifunctional protein that forms dimers and has several binding partners. NSP3 binds specifically to the 3′-terminal conserved sequence of viral mRNAs via its amino terminus. The carboxyl-terminal region of NSP3 interacts with the eukaryotic translation initiation factor eIF4G, and its middle region binds RoXaN (rotavirus X protein associated with NSP3). In uninfected cells, eIF4G interacts with polyA-binding protein (PABP), which binds to the 3′-polyA tail of mRNAs and stimulates translation by circularizing host mRNAs. NSP3 is proposed to inhibit host cellular protein synthesis during infection by evicting PABP from its binding site on eIF4G. NSP3 may facilitate circularization of viral mRNA through its specific interactions with eIF4G and the 3′-conserved sequence of viral mRNAs, thereby promoting viral translation.
NSP4 (20 and 28 kDa) is synthesized on the RER as a transmembrane protein and may be post-translationally glycosylated. Both glycosylated and nonglycosylated forms of NSP4 are detected in infected cells. During later stages of virion maturation, membrane-associated NSP4 functions as a receptor for DLPs, aids in acquisition of VP4 and VP7 and budding through the ER membrane. NSP4 also functions as a viral enterotoxin that leads to Ca2+ release from internal stores in the ER and induction of age-dependent diarrhea in mice. An NSP4 cleavage product is secreted from infected cells and binds to integrins α1β1 or α2β1, triggering a signalling pathway that activates phospholipase C and elevates inositol 1,4,5-triphosphate, leading to Ca2+ release. This pathway is distinct from that of intracellularly expressed NSP4.
The two remaining non-structural proteins, NSP5 (26 kDa) and NSP6 (12 kDa), are encoded by two ORFs in the same viral gene. NSP5, which is serine-threonine rich, is post-translationally modified by both phosphorylation and glycosylation. The protein has ssRNA- and dsRNA-binding activities, forms dimers, and is essential for viroplasm formation and genome replication. Multiple phosphorylated isomers of NSP5 exist in the infected cell. Phosphorylation is stimulated by NSP2 in vivo, but mediated by cellular kinases and possibly an NSP5 autokinase activity. Highly phosphorylated forms of NSP5 localize to viroplasms; the significance of phosphorylation to NSP5 function is unknown. Interactions of NSP2 and NSP5 are responsible for viroplasm formation and localization of core proteins. Interactions of NSP2 and NSP5 with core proteins regulate progeny core formation. A carboxyl-terminal domain of NSP5 is required for multimerization, hyperphosphorylation and interactions with NSP6. A potential ORF encoding NSP6 is conserved among many, but not all, virus isolates. However, NSP6 protein expression has been demonstrated for only a few strains, and its function is not clearly defined. For SiRV-A/SA11, NSP6 (11 kDa) is phosphorylated and localizes to viroplasms, where it may play a regulatory role in the self-association of NSP5.
Information on proteins encoded by viruses of species other than Rotavirus A is less abundant and primarily drawn from sequence analysis of viral genes. The majority of rotaviruses are predicted to encode a single protein product per segment. It is clear that other rotavirus species have homologs of the proteins characterized for Rotavirus A viruses. However, high levels of variation are observed between protein homologs (or putative protein products) of different species (e.g. >84% for VP3 and >87% for VP4), in comparison to the variation observed among Rotavirus A proteins (e.g. <25% for VP2 and <45% for VP4) (Figure 26 and Table 25). In a few cases, proteins of species other than Rotavirus A have been analyzed. For example, the structure of Rotavirus C NSP2 has been determined. Although structurally quite similar, this protein was unable to complement the function of Rotavirus A NSP2 in SiRV-A/SA11-infected cells in which expression of the cognate NSP2 had been knocked down. Additionally, it has been shown that recombinant Rotavirus C VP1 will replicate Rotavirus A RNA, or its own RNA, but only in the presence of its cognate VP2. Rotavirus C VP1 is also predicted to have a structure similar to SiRV-A/SA11 VP1. Further insights into the structure and function of viral proteins from species other than Rotavirus A await the establishment of suitable highly permissive cell culture systems and the expression and characterization of additional non-Rotavirus A recombinant proteins.
Table 24 Genome segments and protein products of Simian rotavirus A/SA11
| Genome segment | Size (bp) | Protein* | Protein size (kDa) | Protein copy number/particle | Location | Function |
| Seg1 | 3302 | VP1 (Pol) | 125.0 | ≤ 12 | core | RNA-dependent RNA polymerase, minor core component, activated by VP2 |
| Seg2 | 2690 | VP2 (T1) | 102.4 | 120 | innermost capsid | Core shell protein, RNA binding, sub-group specificity antigen |
| Seg3 | 2591 | VP3 (Cap) | 98.1 | ≤ 12 | core | Guanylyltransferase, methyltransferase, ssRNA binding, minor core component |
| Seg4 | 2362 | VP4 | 86.8 | 180 | outer capsid | P-type neutralization antigen, viral attachment protein, homotrimer, spike, hemagglutinin, activated by trypsin cleavage (VP5*, membrane penetration; VP8*, carbohydrate binding) |
| VP5* | 60.0 |
|
|
| ||
| VP8* | 28.0 |
|
|
| ||
| Seg5 | 1611 | NSP1 | 58.7 | 0 | cytoplasm | Antagonist of interferon expression, putative viral E3 ubiquitin ligase, RNA binding, RING domain |
| Seg6 | 1356 | VP6 (T13) | 44.8 | 780 | intermediate capsid | Group and sub-group specificity antigen, trimeric, major virion protein |
| Seg7 | 1104 | NSP3 | 34. 6 | 0 | cytoplasm | Binds 3’ end of viral mRNA and cellular eIF4G, promotes circularization of viral mRNAs, surrogate of PABP, inhibits host translation |
| Seg8 | 1059 | NSP2 (ViP) | 36.7 | 0 | viroplasm | Octamer with NTPase, RTPase, and helix destabilizing activities, ssRNA binding, essential viroplasm component, interacts with NSP5 |
| Seg9 | 1062 | VP7 (1) | 37.4 | 780 | outer capsid | G-type neutralization antigen, virion surface glycoprotein, forms Ca2+-stabilized trimer |
| VP7 (2) cleaved form | 33.9 |
|
|
| ||
| Seg10 | 751 | NSP4 | 20.3 | 0 | RER membrane | RER transmembrane glycoprotein, binds DLPs, essential for budding into ER and addition of outer capsid, age-dependent diarrhea-inducing enterotoxin, disrupts Ca2+ homeostasis |
| Seg11 | 667 | NSP5 | 21.7 | 0 | viroplasm | Phosphorylated, O-linked glycosylation, serine-threonine rich, RNA binding, essential viroplasm component, interacts with NSP2 |
| NSP6 | 11.0 | 0 | viroplasm | Product of second ORF, RNA binding, interacts with NSP5, non-essential viroplasm component |
* Protein structure/function: Pol, RNA polymerase ; Cap, capping enzyme (guanylyltransferase and transmethylase); T1, inner virus structural protein with T=1 symmetry with two molecules in the icosahedral asymmetric unit (interpreted in the orbiviruses as T=2 symmetry); T13, inner virus structural protein with T=13 symmetry; ViP, viral inclusion body or viroplasm matrix protein. Viruses from other species within the genus may have proteins with significant differences in sizes.
Table 25 Percentage amino acid differences between the inner capsid protein (VP2) of isolates of Rotavirus A, B, C and D, and novel adult diarrhea rotavirus (NADRV)
|
| Rotavirus A | Rotavirus B | Rotavirus C | Rotavirus D | NADRV | ||||
| Hu/Wa | Mu/ETD_822 | Av/PO-13 | Hu/WH-1 | Hu/Bristol | Av/05V0049 | Hu/J19 |
| ||
| Accession no. | CAA34733 | X14942 | ACY95261 | BAA24147 | AAT09117 | CAC44890 | ADN06424 | YP_392491 | |
| Rotavirus A | Si/SA11 | 0 | 7.5 | 11.6 | 23.6 | 88.2 | 53.0 | 53.4 | 87.5 |
| Hu/Wa | 0 | 12.2 | 23.8 | 88.2 | 52.8 | 53.2 | 88.1 | ||
| Mu/ETD_822 |
| 0 | 24.6 | 88.2 | 52.8 | 54.0 | 87.6 |
| |
| Av/PO-13 |
|
| 0 | 89.3 | 51.2 | 52.9 | 88.3 |
| |
| Rotavirus B | Hu/WH-1 |
|
|
|
| 0 | 89.8 | 89.0 | 52.9 |
| Rotavirus C | Hu/Bristol |
|
|
|
|
| 0 | 55.0 | 89.6 |
| Rotavirus D | Av/05V0049 |
|
|
|
|
|
| 0 | 88.6 |
| NADRV | Hu/J19 |
|
|
|
|
|
|
| 0 |
The abbreviations used to indicate host species are: Av, avian; Hu, human; Mu, murine; Si, simian. MEGA 4 software was used to calculate p-distances on the amino acid level.
Lipids
None reported. Immature particles acquire a transient membrane during budding into the ER.
Carbohydrates
Three viral proteins have been shown to be glycosylated. In two cases (VP7 and NSP4), the sugar is N-linked to asparagine, and in the third (NSP5) it is O-linked to serine and/or threonine.
Genome organization and replication
The complete RNA-protein coding assignments have been determined for several Rotavirus A isolates. The coding assignments for the SiRV-A/SA11 strain are given in Table 24. The replication cycle for many animal rotavirus strains, which is typically completed in 12–15 h at 37 °C, has been studied primarily in continuous cell cultures derived from monkey kidneys. There is little definitive information about the early steps in the replication cycle. VP4 is the viral attachment-protein but the cellular receptor has not been conclusively identified. Some rotavirus strains attach to the N-acetyl-neuraminic (sialic) acid residues on the cell surface, and there is evidence to suggest that several integrins and heat shock protein 70 serve as co-receptors. Current data suggest that rotaviruses may enter cells either by receptor-mediated endocytosis or by direct membrane penetration. In both cases, virus entry leads to loss of the outer VP4 and VP7 protein layer and release of a transcriptionally-active DLP into the cytoplasm. DLP-associated enzymes produce 5′-capped, non-polyadenylated mRNAs, which are synthesized using the full-length minus strand of each genome segment as a template. In part, gene expression is regulated by differences in the level of transcription occurring from each genome segment. The viral mRNAs serve two functions. Firstly, they act as templates for translation, producing viral proteins. Efficiency of mRNA translation regulates expression levels of the individual viral proteins. Secondly, viral mRNAs act as templates for minus-strand synthesis, producing dsRNA genome segments. The use of short interfering RNA technology has provided evidence that the pools of mRNAs that serve as templates for translation and replication are distinct. Virus assembly begins in cytoplasmic inclusions termed viroplasms, formation of which is mediated by NSP2 and NSP5. During early assembly steps, the 11 different viral mRNAs interact with one another and with VP1 and VP3. These interactions are followed by association with VP2, which triggers minus-strand synthesis and results in the formation of cores containing a complete set of 11 dsRNA segments. Cis-acting replication elements for minus-strand synthesis have been identified using an in vitro replication system, the most critical of which is the seven 3′-terminal nucleotides of the mRNAs. Following their assembly, VP6 is added to cores, forming DLPs. The next steps in the morphogenesis of progeny virions are unique to rotaviruses and involve recruitment of DLPs to the ER by the transmembrane glycoprotein NSP4. Budding of DLPs through the ER results in the transient acquisition of a lipid envelope and addition of VP4 and VP7 to form the outer virion shell.
Antigenic properties
Three viral proteins (VP4, VP6 and VP7) of Rotavirus A isolates have been subjected to detailed antigenic characterization. VP6, which forms the intermediate capsid layer, is a highly conserved and highly immunogenic protein, carrying both virus group and sub-group (SG)-specific determinants. It does not elicit the production of neutralizing antibodies, but it may play a role in the induction of protective immunity. VP6 has been the major target of diagnostic assays for rotaviruses and specifies the group (i.e. the species or candidate species listed for this genus).
Within Rotavirus A isolates, VP6 bears the epitopes of the SG specificities that allow antigenic classification of rotavirus into SG I, SG II, both SG I and II, or neither SG, according to reactivity with two monoclonal antibodies. More than a decade ago, a binary classification system reminiscent of the one used for the classification of influenza A viruses was established. This system was based on antigenic reactivities of the two outer capsid proteins, VP4 and VP7, which independently elicit neutralizing antibodies. Thus, rotavirus strains were classified into P serotypes (VP4 is protease sensitive) and G serotypes (VP7 is a glycoprotein). Classification of rotaviruses into P (VP4) or G (VP7) serotypes is performed by cross-neutralization assays using hyperimmune sera raised to prototype viruses or laboratory-engineered mono-reassortants. So far, 14 G serotypes and 14 P serotypes have been identified. As the ease of nucleic acid sequencing has increased, antigenic classification has slowly been replaced by a classification system of rotaviruses into P (VP4) and G (VP7) genotypes that is based on nucleotide sequence similarities of VP4 and VP7 genes. G serotype designations largely coincide with G genotype designations. There is greater discrepancy among P serotype and genotype designations. Thus, a dual nomenclature has been adopted for VP4 antigenic and genetic classification. The P serotype, when known, is denoted by a number. The P genotype is denoted by a number within squared brackets, which immediately follows the P serotype. When the P serotype is not known, only the P genotype is used.
Recently, a uniform classification and nomenclature system, based on nucleotide identities of the 11 rotavirus genome segments and phylogenetic dendrograms, has been established by the Rotavirus Classification Working Group for Rotavirus A. This classification scheme currently comprises 23 G (VP7), 32 P (VP4), 14 I (VP6; inner capsid), 6 R (VP1; RNA-dependent RNA polymerase), 6 C (VP2; core), 7 M (VP3; methyltransferase), 16 A (NSP1; interferon antagonist), 6 N (NSP2; NTPase), 8 T (NSP3; translation enhancer), 12 E (NSP4; enterotoxin), and 8 H (NSP5/6; phosphoprotein) genotypes.
In the case of species other than Rotavirus A, little information is available at present on the extent of antigenic diversity.
Biological properties
Rotaviruses can prove difficult to cultivate in vitro, with highly permissive growth generally restricted to epithelial cell lines derived from monkey kidneys. Infection can be enhanced by pre-treatment of virus with trypsin. The restriction of virus growth in vitro parallels the in vivo situation, in which virus replication is typically restricted to the terminally differentiated enterocytes lining the tips of the microvilli in the small intestine. However, recent evidence suggests that some rotavirus infections may cause antigenemia and viremia due to replication at secondary sites in the host.
There are several mechanisms of pathogenesis, including the destruction of enterocytes, leading to malabsorption and an osmotic diarrhea. Prior to the appearance of histologic changes, a watery diarrhea is often seen, which is thought to be secretory, possibly induced by the action of the rotavirus enterotoxin NSP4. It has been proposed that the destruction of enterocytes causes a loss of the permeability barrier between the gut lumen and the vasculature, resulting in the osmotic pull of fluid from the circulation into the gut and the watery characteristic of rotavirus-induced diarrhea. Rotaviruses infect a wide range of avian and mammalian species, with disease being restricted in the great majority of cases to the young. Isolates of Rotavirus A, Rotavirus B and Rotavirus C can infect humans, with Rotavirus A being responsible for the majority of seasonal endemic diarrheal disease in young children. Isolates of Rotavirus B have caused sporadic epidemic outbreaks of gastroenteritis in adults. Rotavirus C has been associated with self-limiting outbreaks of gastroenteritis in humans, primarily in the young.
Species demarcation criteria in the genus
The rotaviruses are currently divided into five species (Rotavirus A to Rotavirus E), and three additional groups that have not yet been classified (Rotavirus F, Rotavirus G and NADRV). Viruses within different species are thought to be unable to reassort their genome segments under normal circumstances, and each species may therefore represent a separate gene pool.
In addition to the other general criteria used throughout the family, members of a species in the genus Rotavirus may be identified by:
- Serological cross-reactivity by ELISA, using either polyclonal sera or monoclonal antibodies against VP6 (or its homolog in isolates of other species).
- Sequence analysis of conserved genome segments (e.g. Seg1 and Seg6). Viruses within the same species will normally have >75% nucleotide in Seg6. However, this is not a consistent method for differentiating all species, as avian and mammalian isolates of Rotavirus A may have only about 60% identity.
- Host range. For example, Rotavirus E isolates have to date only been found in pigs. Rotavirus D isolates and the Rotavirus F and Rotavirus G groups have only been isolated from avian species.
List of species in the genus Rotavirus
| Rotavirus A |
|
|
| Simian rotavirus A/SA11 | [Seg1: X16830, Seg2: X16831, Seg3: X16062, Seg4: X14204, Seg5: X14914, Seg6: X00421, Seg7: X00355, Seg8: J02353, Seg9: K02028, Seg10: KO1138, Seg11: X07831] | (SiRV-A/SA11) |
| Rotavirus B |
|
|
| Rotavirus B | [Seg1: EU490413, Seg2: AY539859, Seg3: EU490416, Seg4: AY539857, Seg5: AY539862, Seg6 AY539858, Seg7: AY539860, Seg8: AY539861, Seg9: AY539856, Seg10: AY539864, Seg11: AY539863] | (RV-B/WH-1) |
| Rotavirus C |
|
|
| Human rotavirus C/Bristol | [Seg1: AJ304859, Seg2: AJ303139,Seg3: X79442, Seg4: X96697, Seg5: X59843, Seg6: AJ132203, Seg7: AJ132204, Seg8: X77257, Seg9: AJ132205, Seg10: M81488, Seg11: X83967] | (HRV-C/Bristol) |
| Rotavirus D |
|
|
| Chicken rotavirus 05V0059 | [Seg1: GU733443, Seg2: GU733444, Seg3: GU733445, Seg4: GU733446, Seg5: GU733447, Seg6: GU733448, Seg7: GU733449, Seg8: GU733450, Seg9: GU733451, Seg10: GU733452, Seg11: GU733453] | (AvRV-D/05V0059) |
| Rotavirus E |
|
|
| Porcine rotavirus E/DC-9 |
| (PoRV-E/DC-9) |
List of other related viruses which may be members of the genus Rotavirus but have not been approved as species
| Rotavirus F |
| (RV-F) |
| Chicken rotavirus F/A4 |
| (AvRV-F/A4) |
| Rotavirus G |
| (RV-G) |
| Chicken rotavirus G/555 |
| (AvRV-G/555) |
| NADRV |
| (NADRV) |
| Human NADRV/ J19 | [Seg1: DQ113897, Seg2: DQ113898, Seg3: DQ113900, Seg4: DQ113899, Seg5: DQ113901, Seg6: DQ113902, Seg7: DQ113903, Seg8: DQ113904, Seg9: DQ113905, Seg10: DQ113906, Seg11: DQ113907] | (Hu NADRV/J19) |
Genus Seadornavirus
Type species Banna virus
Distinguishing features
The seadornavirus genome consists of 12 segments of dsRNA. During replication, viruses are found in the cell cytoplasm. Viruses are transmitted to vertebrate hosts by mosquito vectors.
Virion properties
Morphology
Particles are non-enveloped with a diameter ranging between 60 and 70 nm having two concentric capsid shells with a core that is about 40–50 nm in diameter. Electron microscopic studies, using negative staining have shown that particles have a well-defined surface capsomeric structure and icosahedral symmetry (Figure 27).
Physicochemical and physical properties
The buoyant density of the virus in CsCl is 1.36 g cm−3. Viruses are stable around neutral pH, but lose infectivity at pH 3.0. At 4 °C, the virus is stable for long periods, even non-purified in cell culture lysate, which is a convenient way for medium-term storage. Heating to 55 °C considerably decreases the viral infectivity. Seadornaviruses are stable upon treatment with freon, which could be used for purification of viral particles from cell lysate. The viral infectivity is abolished by treatment with sodium dodecyl sulfate. Viruses can be stored for long periods at −80 °C, and infectivity is further protected by addition of 50% fetal calf serum.
Nucleic acid
The genome consists of 12 dsRNA segments that are numbered in order of reducing Mr, or increasing electrophoretic mobility during agarose gel electrophoresis. The genome comprises approximately 21,000 bp, with segment lengths that range between 3747 and 862 bp. The RNA genome segments of Banna virus (BAV), Kadipiro virus (KDV) and Liao ning virus (LNV) migrate as groups of 6-6, 6-5-1 and 6-2-3-1, respectively during 1% agarose gel electrophoresis (AGE) (Figure 28). These patterns are thought to be characteristic of each virus species.
From the full-genome sequence analyses of different Banna virus isolates, two distinct genotypes have been identified (based on Seg7 and Seg9) that correlate with virus serotype. Genotype A includes isolates BAV-Ch (China) and BAV-In6423 (Indonesia), while genotype B includes BAV-In6969 and BAV-In7043 (Indonesia). The proteins translated from Seg7 and Seg9 show 72% and 54% aa identity between genotypes A and B respectively, while all of the other segments appear to be more conserved, showing 83% to 98% identity.
Two genotypes of LNV have also been identified that correlate with virus serotype. Amino acid identity in the cell attachment outer capsid protein VP10 between these two genotypes was found to be 81%.
Much lower levels of aa sequence identity were detected between homologous proteins of different species: between BAV and KDV (24% to 42%), between BAV and LNV (18% to 41%) or between LNV and KDV (21% to 42%). In each case highest levels of aa identity were detected in the viral polymerase (VP1[Pol]).
The genome segments of BAV and KDV have a G+C content that varies between 37% and 39%, with 5′- and 3′-NTRs that vary between 76 and 200 nt respectively. The NTRs of all seaodornaviruses so far studied (Table 26) contain conserved base pairs at both termini (+ve 5′-GU…GAC-3′).
Seadornavirus mRNAs are usually regarded as non-infectious. However, fully functional and infectious viruses have been recovered by the introduction of all twelve mRNAs into BSR cells.
Table 26 Conserved terminal sequences (positive strand) of seadornavirus genome segments
| Virus species | Strain | 5′ end | 3′ end |
| Banna virus | BAV-In6423 | 5′-GUAUA/UA/UAAA/UA/UU | A/GCC/UGAC-3′ |
| Kadipiro virus | KDV-Ja7075 | 5′-GUAGAAA/UA/UA/UU | AA/CC/UGAC-3′ |
| Liao ning virus | LNSV-NE97-31 | 5′-GUUAUA/UA/UA/U | A/CU/CCGAC-3′ |
Proteins
Native proteins of BAV particles were characterized by mass spectrometry analysis and by radiolabeling of BAV infected cells. Their putative functions are indicated in Table 27.
Host–cell protein synthesis shut-off starts at 2 h post BAV infection of C6/36 cells (a mosquito cell line), and the shut-off is complete by 6 hpi. [35S]-methionine added to C6/36 cell cultures at 6 hpi was incorporated almost exclusively into 12 protein bands (resolved by SDS PAGE, which are thought to represent the different viral proteins (one protein per genome segment). Most of these proteins have apparent molecular masses that agree with the theoretical sizes predicted by sequence analysis of the viral genome. The only exception is VP7, which migrates more slowly than expected.
Purified BAV-Ch virus particles contain seven structural proteins, each of which co-migrated with one of the radio-labeled proteins from infected cells (VP1, VP2, VP3, VP4, VP8, VP9 and VP10). Only five of these proteins were also detected in cores, indicating that the outer coat (like those of the non-turreted orbiviruses and rotaviruses) is composed of two proteins (VP4 and VP9). Analyses of BAV-Ch structural protein sequences by mass spectrometry confirmed the identity of the core (VP1, VP2, VP3, VP8, VP10) and outer capsid components (made of VP9 and VP4), demonstrating that VP5, VP6, VP7, VP11 and VP12 are non-structural proteins.
[35S] methionine labeled BAV-Ch particles were purified and analyzed by SDS-PAGE and the ratios of the different structural proteins were calculated. VP2 and VP8 are the two most abundant proteins of the BAV core. The lower relative abundance and higher molecular weight of VP2 identifies it as the subcore-shell T2 protein [equivalent to VP3 of bluetongue virus (BTV) and VP2 of rotavirus]. In contrast, VP8 is smaller and more abundant, identifying it as the core-surface T13 protein. VP8 and VP2 have a molar ratio of 6.5 in purified BAV-Ch particles, identical to the ratio of 780/120 previously detected between the subcore and core-surface proteins of both BTV and rotaviruses. On this basis, the numbers of the VP8 and VP2 molecules in the BAV core are assumed to be 780 and 120 per particle, respectively, allowing the average copy number of the other protein components of virus particles or cores to be calculated: 24 copies for VP1(Pol), 12 copies for the VP3(Cap), 333 copies for the VP4, 310 copies for VP9 and VP10.
The structure of the BAV outer-capsid protein VP9 was determined by X-ray crystallography at 2.6 Å resolution, revealing a trimeric molecule, held together by an N-terminal helical bundle, reminiscent of coiled-coil structures found in fusion-active proteins such as HIV gp41. The major domain of VP9 contains stacked β sheets with marked structural similarities to the rotavirus receptor binding protein VP8. Anti-VP9 antibodies neutralize viral infectivity, and, remarkably, pretreatment of cells with trimeric VP9 increased viral infectivity, indicating that VP9 is involved in virus attachment to cell surface and subsequent internalization. Sequence similarities were also detected between BAV VP10 and the VP5 portion of rotavirus VP4, suggesting that the receptor binding and internalization apparatus, which is a single gene product activated by rotavirus proteoloysis, is the product of two separate genome segments in BAV.
Table 27 Genome segments and protein products of Banna virus-Indonesia-6423
| Genome segment | Size (bp) | Protein (copy number per particle) | Protein size (kDa)* | Structure/function |
| Seg1 | 3747 | VP1(Pol) [24] | 137 | RdRp |
| Seg2 | 3048 | VP2(T2) [120] | 108 | Inner layer of core, T2 protein |
| Seg3 | 2400 | VP3(Cap) [12] | 82 | Capping enzyme |
| Seg4 | 2038 | VP4 [333] | 64 | Outer coat protein |
| Seg5 | 1716 | VP5-NS [0] | 56 | Non-structural |
| Seg6 | 1671 | VP6-NS [0] | 48 | Non-structural |
| Seg7 | 1136 | VP7-NS [0] | 35 | Protein kinase |
| Seg8 | 1119 | VP8 [780] | 32 | T13, outer layer of core |
| Seg9 | 1101 | VP9 [310] | 31 | Outer coat cell attachment protein |
| Seg10 | 977 | VP10 [310] | 29 | Stalk base for VP10 |
| Seg11 | 867 | VP11-NS [0] | 21 | Non-structural |
| Seg12 | 862 | VP12-NS [0] | 24 | dsRNA-binding |
* Calculated from the sequenced segments.
Lipids
None reported.
Carbohydrates
None reported.
Genome organization and replication
The 12 genome segments are all monocistronic (Table 27). Seadornavirus isolates replicate in mosquito cell lines, and considerable amounts of virus (over 40% of progeny) are liberated into the culture medium prior to cell death and gross CPE, which usually occurs by 40 h post infection with BAV, and 72 h post infection with KDV. BAV was also found to replicate in BSR cells (a clone of BHK-21). Liao ning virus is the only seadornavirus known to replicate in a wide variety of mammalian cells, including primary cell cultures.
Large electron-dense structures occur in the cytoplasm of BAV-Ch infected cells, which correspond to the viral inclusion bodies (VIB) thought to be the main site of replication and particle assembly of other reoviruses. Particles (ca. 50 nm in diameter) with a smooth surface were detected mainly at the periphery of the VIB, although some particles were observed within the VIB matrix. Virus particles were also detected within large vacuoles that were dispersed throughout the infected cell cytoplasm. These vacuoles contained multiple double-layered vesicles, lined with viral particles (ca. 50 nm in diameter) at their inner surface and it is possible that this reflects some involvement of cellular membrane structures or organelles in virus morphogenesis, transport or replication (as previously reported for the rotaviruses). Virus entry into cells by endocytosis was suggested by the detection of virus particles in pits at the cell surface. Virions were also observed near the cell membrane, which appeared to be budding from the cell surface.
Antigenic properties
BAV from China, Vietnam and Indonesia, KDV from China and Indonesia and Liao ning virus from China are classified as three distinct species and show little cross-reaction in neutralization tests.
Biological properties
Seadornaviruses have been isolated from humans and mosquitoes, which serve as vectors. The mosquito species that have been implicated include Culex vishnui, C. fuscocephalus, Anopeles vagus, Anopheles aconitus, Anopheles subpictus and Aedes dorsalis. Experimentally, the viruses were also found to replicate in adult mice and were detected in infected mouse blood 3 days post infection. LNV kills adult mice causing a hemorrhagic syndrome. BAV was isolated from the serum and cerebrospinal fluids of human patients showing neurological manifestations. The pathology provoked by this virus is only poorly described. KDV has only been isolated from mosquitoes.
BAV and KDV occur in tropical and subtropical regions, where other mosquito-borne viral disease especially Japanese encephalitis and dengue have been reported as endemic. Despite the isolation of BAV from infected human patients, no surveys have been reported concerning the detection and prevalence of antibodies to these viruses in human sera. Liao ning virus has been isolated from mosquitoes.
Species demarcation criteria in the genus
In addition to the other general criteria used throughout the family, members of a species in the genus Seadornavirus may be identified by:
- RNA cross-hybridization assays: within a single species: RNA sequence that exhibit more than 74% similarity will hybridize at 36 °C below the Tm of fully base-paired duplex.
- Serological comparisons by neutralization assays. Hyperimmune ascitic fluids against genotype A viruses of BAV, do not efficiently cross-neutralize those of genotype B. However, isolates within a single genotype show high levels of cross-neutralization. There is no cross-neutralization or cross-reactivity between members of the species Banna virus and Kadipiro virus.
- Sequence analysis: In the conserved Seg12, viruses within the same species will normally have >89% nucleotide identity. Within the polymerase (the most conserved protein) isolates of different species have <50% aa identity.
List of species in the genus Seadornavirus
| Banna virus {Culex and Anopheles mosquitoes : Humans} |
|
|
| Banna virus - China | [Seg1: AF134525, Seg2: AF13526, Seg6: AF13527, Seg7: AF052035, Seg8: AF052034, Seg9: AF0520333, Seg10: AF052032, Seg11: AF052031, Seg12: AF052030] | (BAV-Ch) |
| Banna virus - Indonesia-6423 | [Seg1: AF133430, Seg2: AF134514,Seg3: AF134515, Seg4: AF134516, Seg5: AF134517, Seg6: AF134518,Seg7: AF052018, Seg8: AF052017, Seg9: AF052016, Seg10: AF052015, Seg11: AF052014, Seg12: AF019908] | (BAV-In6423) |
| Banna virus - Indonesia-6969 | [Seg1: AF134522, Seg2: AF134523,Seg6: AF134524, Seg7: AF052013, Seg8: AF052012, Seg9: AF052011, Seg10: AF052010, Seg11: AF052009, Seg12: AF052008] | (BAV-In6969) |
| Banna virus - Indonesia-7043 | [Seg1: AF134519, Seg2: AF134520, Seg6: AF134521, Seg7: AF052029, Seg8: AF052028, Seg9: AF052027, Seg10: AF052026, Seg11: AF052025, Seg12: AF052024] | (BAV-In7043) |
| Kadipiro virus {Culex mosquitoes} |
|
|
| Kadipiro virus - Java-7075 | [Seg1: AF133429, Seg2: AF134509,Seg3: AF134510, Seg4: AF134511, Seg5: AF134512, Seg6: AF134513, Seg7: AF052023, Seg8: AF052022, Seg9: AF052021, Seg10: AF052020, Seg11: AF052019, Seg12: AF019909] | (KDV-Ja7075) |
| Liao ning virus {Aedes dorsalis : mosquito} |
|
|
| Liao ning virus (NE97-12) | [Seg1: AY701339, Seg2: AY701340,Seg3: AY701341, Seg4: AY701342, Seg5: AY701343, Seg6: AY701344, Seg7: AY701345, Seg8: AY701346, Seg9: AY701347, Seg10: AY701348, Seg11: AY701349, Seg12: AY701350] | (LNV-NE97-12) |
| Liao ning virus (NE97-31) | [Seg1: AY317099, Seg2: AY317100,Seg3: AY317101, Seg4: AY317102, Seg5: AY317103, Seg6: AY317104, Seg7: AY317105, Seg8: AY317106, Seg9: AY317107, Seg10: AY317108, Seg11: AY317109, Seg12: AY317110] | (LNV-NE97-31) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ], arthropod vector and host names { } and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Seadornavirus but have not been approved as species
| Banna virus ACH |
| (BAV-ACH) |
| Banna virus HN59 |
| (BAV-HN59) |
| Banna virus HN131 |
| (BAV-HN131) |
| Banna virus HN191 |
| (BAV-HN191) |
| Banna virus HN295 |
| (BAV-HN295) |
| Banna virus LY1 |
| (BAV-LY1) |
| Banna virus LY2 |
| (BAV-LY2) |
| Banna virus LY3 |
| (BAV-LY3) |
| Banna virus M14 |
| (BAV-M14) |
| Banna virus TRT2 |
| (BAV-TRT2) |
| Banna virus TRT5 |
| (BAV-TRT5) |
| Banna virus WX1 |
| (BAV-WX1) |
| Banna virus WX2 |
| (BAV-WX2) |
| Banna virus WX3 |
| (BAV-WX3) |
| Banna virus WX8 |
| (BAV-WX8) |
These isolates of seadornaviruses have been isolated in many provinces in China including Beijing, Gansu, Yuannan, Hainan, Henan, Shanshi, Xinjiang and recently from Liao ning. Viruses other than Banna, Kadipiro and Liao-ning viruses are still uncharacterized and have been temporary designated Banna virus isolates. Their serological relationship to Banna virus was not fully explored. These isolates are probably distinct from Banna virus, and at least some should represent new species within genus Seadornavirus.
Phylogenetic relationships within the genus
Phylogenetic tree for homologous genome segments from members of the species Banna virus, Kadipiro virus and Liao ning virus. The tree is based on outer-capsid and cell attachment protein sequences (encoded by Seg9 (VP9) of BAV, its homologous Seg11 (VP11) of KDV and Seg10 (VP10) of LNV). This tree also shows the two genotypes (serotypes) of BAV (A and B). The amino acid identity between VP9 of BAV and VP11 of KDV ranges between 14% and 16%. Between VP9 of BAV and VP10 of LNV amino acid identity ranges from 17% to 19%. Between VP11 of KDV and VP10 of LNV amino acid identities ranged from 17% to 18%. Between the two genotypes of BAV, it ranges from 41% to 43%. Within a given genotype of BAV it ranges between 90 and 96%. Between the two genotypes (serotypes) of LNV amino acid identity is 81%. Sequences were aligned using ClustalX and the tree constructed in MEGA4 using the neighbor-joining method and the P-distance algorithm. Branching is supported by bootstrap values >85%.
Genus Phytoreovirus
Type species Wound tumor virus
Distinguishing features
Phytoreovirus particles have icosahedral symmetry with a distinctive angular appearance and possess 12 dsRNA species. They are transmitted by cicadellid leafhoppers to susceptible plant species, replicating in both hosts and vectors.
Virion properties
Morphology
Virions of rice dwarf virus (RDV) are icosahedral, appear to be double-shelled and about 70 nm in diameter (Figure 30). The outer layer of RDV contains 260 trimers of P8 (46 kDa): a total of 780 molecules, arranged with T=13 l symmetry (Figure 30). The relative location of the neighboring capsomers on the icosahedral particle is such that they form pentameric or hexameric rings. The inner capsid layer is reported to be a complete protein shell, composed of 60 dimers of P3 (114 kDa), a total of 120 molecules, arranged with a suggested T=1 icosahedral symmetry (Figure 30). The outer capsid P8 trimers bind more tightly at the threefold positions of the single-layered core. The RDV particle structure appears to be comparable to that of core or double layered particles of some other genera (Orbivirus and Rotavirus respectively). Ordered structures are visible in the periphery of the RNA region.
Wound tumor virus (WTV) is reported to possess three protein shells, including an outer amorphous layer, an internal layer of distinct capsomers and a smooth core that is about 50 nm in diameter but lacking spikes.
Physicochemical and physical properties
The Mr of phytoreoviruses is about 75×106. The virion S20,w is about 510. The optimal stability of particles is at pH 6.6. The buoyant density of RDV is 1.39–1.42 g cm−3 and the virion is unstable losing P8 in CsCl. CCl4 removes P2 from the RDV virion.
Nucleic acid
Phytoreoviruses have 12 genome segments of linear dsRNA, numbered according to their migration during PAGE. However, their relative sizes based on RNA sequence data indicate that Seg4 and Seg5, or Seg9 and Seg10, may migrate in the reverse order during agarose gel electrophoresis (Table 28). The RNA constitutes about 22% of the virion dry weight. The dsRNA Mr is in the range 0.3 to 3.0×106, with characteristic sizes for each virus. For WTV Seg4=2565bp; Seg5=2613bp; Seg6=1700bp; Seg7=1726bp; Seg8=1472bp; Seg9=1382bp; Seg10=1172bp; Seg11=1128bp; Seg12=851bp. G+C content is 38–44% and 41–48% for the genomic segments of WTV and RDV respectively. The positive strand of each genome segment, of all viruses in the genus, contains the conserved sequence 5′-GG(U/C)A---UGAU-3′ except for RDV Seg9 which has 5′-GGUA---CGAU-3′ (Table 29). These genus-specific terminal sequences are situated adjacent to inverted repeats, which are 6–14 bases long. These sequences differ for each RNA segment. Individual isolates of RDV can frequently be distinguished by electrophoretic profiles of at least one of the 12 genomic segments in PAGE. RDV particles encapsidate the genomic dsRNA in supercoiled form.
Table 28 Genome segments and protein products of rice dwarf virus, Akita isolate
| Genome segment | Size (bp) | Non-coding regions (bp) 5′ – 3′ | Protein* | Protein size (kDa)** | Function and location (Number per particle) |
| Seg1 | 4423 | 35–53 | P1 (Pol) | 164.1 (170) | Core, RNA polymerase |
| Seg2 | 3512 | 14–147 | P2 | 123.0 (130) | Outer capsid, essential for vector transmission |
| Seg3 | 3195 | 38–97 | P3 | 114.3 (110) | Major core (120) |
| Seg4 | 2468 | 63–221 | Pns4 | 79.8 (83) | Non-structural (Phosphorylated) |
| Seg5 | 2570 | 26–138 | P5 (Cap) | 90.5 (89) | Core, guanylyltransferase |
| Seg6 | 1699 | 48–121 | Pns6 | 57.4 (56) | Non-structural |
| Seg7 | 1696 | 25–150 | P7 | 55.3 (58) | Core, nucleic acid binding protein |
| Seg8 | 1427 | 23–138 | P8 (T13) | 46.5 (43) | Major outer capsid (780) (trimer) |
| Seg9 | 1305 | 24–225 | Pns9 | 38.9 (49) | Non-structural |
| Seg10 | 1321 | 26–233 | Pns10 | 39.2 (35) | Non-structural (silencing suppressor) |
| Seg11 | 1067 | 29–492 | Pns11a | 20.0 (23) | Non-structural (nucleic acid binding protein) |
| 5–492 | Pns11b | 20.8 (24) |
| ||
| Seg12 | 1066 | 41–86 | Pns12 | 33.9 (34) | Non-structural |
| 312–475 |
| Pns12OPa |
| ||
| 336–475 | Pns12OPb | 9.6 (7) |
|
* Protein structure/function: Pol, RNA polymerase ; Cap, capping enzyme (guanylyltransferase and transmethylase); T13, inner virus structural protein with T=13 symmetry.
** Calculated from nt sequences (size determined by SDS PAGE in brackets).
Table 29 Conserved terminal sequences (positive strand) of phytoreovirus genome segments
| Virus species | Strain | 5′ end | 3′ end |
| Rice dwarf virus | RDV | 5′-GGU/CAAA | U/CGAU-3′ |
| Rice gall dwarf virus | RGDV | 5′-GGU/CAA/UUUU | UGAU-3′ |
| Wound tumor virus | WTV | 5′-GGUAUU | UGAU-3′ |
| (not classified) | Homalodisca vitripennis reovirus | 5′-GGCG/A | U/CGAU-3′ |
Proteins
Phytoreoviruses have six or seven structural proteins in the range 45 to 160 kDa. RDV has six structural proteins (P1(Pol), P2, P3, P5(Cap), P7, and P8). For WTV the seven CPs are organized in three shells consisting of an amorphous outer layer of two CPs, an inner shell of two CPs and a core of three CPs. Protein constitutes about 78% of the particle dry weight. Removal of the outer shell is not required for activation of the virus transcriptase and associated enzymes. Removal of RDV P2 abolishes the ability to infect vector cell monolayers but virus particles without P2 retain viral transcriptase activity and can infect vector insects by an injection method. P1 is the transcriptase/polymerase and binds to genomic dsRNA. P7 has non-specific nucleic acid binding activity. P3 binds to P3, P7 and P8. P7 binds to P1 and P8. P5 is probably a guanylyl transferase and has GTP, ATP and UTP binding activities. P3 and P8 form virus-like particles in transgenic rice plants. P8 interacts with rice glycolate oxidase, a typical enzyme of peroxisomes. P7 was found to contain dsRNA-binding domains. Of the non-structural proteins, Pns4 is phosphorylated and is associated with large cytoplasmic fibrils and formed novel minitubules in infected cultured cells of its leafhopper insect vector, as revealed by immunofluorescence and immunoelectron microscopy. Early in infection, Pns4 was detected at the periphery of the viroplasm, and it was then observed on amorphous or fibrillar inclusions, which were identified as bundles of minitubules, at later stages of infection. Pns10 protein was found to possess an antisilencing activity, which suppresses the host RNA silencing machinery. Pns11 has nonspecific nucleic acid binding activity and Pns 12 can be phosphorylated and is one of the early proteins expressed in cultured insect cells.
Lipids
None known.
Carbohydrates
None known.
Genome organization and replication
The coding strand of each dsRNA has a single ORF, except for Seg11 and Seg12 of RDV (Table 28), Seg9 of rice gall dwarf virus (RGDV) and Seg9 of WTV. RDV Seg11 has two in-frame initiation codons, thus resulting in two ORFs. RDV Seg12, RGDV Seg9 and WTV Seg9 possess a second, small out-of-frame and over-lapping ORF, downstream within the major ORF. No evidence has yet been obtained for the expression of this second ORF. Five structural and five NS WTV proteins have been assigned to their respective genome segments. RDV Seg1 encodes the putative transcriptase. Genus-specific and segment-specific sequence motifs appear to be necessary for successful replication, translation and encapsidation. Laboratory strains having internal deletions in some segments, but intact termini, replicate and compete favorably with wild-type virus, although the proteins expressed are aberrant, and the ability of the viruses to be transmitted by vectors may be lost. Virus replication occurs in the cytoplasm of infected cells in association with viroplasms. WTV and RGDV are confined to phloem tissues of the plant host, whereas RDV can also multiply elsewhere.
Antigenic properties
The three recognized phytoreoviruses are antigenically distinct. Epitopes representing the outer surface are unrelated to each other, while the inner surface epitopes of the capsid of RDV and RGDV will cross-react.
Biological properties
Plant hosts are either dicotyledonous (WTV), or graminaceous (RDV and RGDV). WTV was originally identified in northeastern USA in the leafhopper Agalliopsis novella. The virus was recently found in New Jersey, USA, in a single periwinkle (Catharanthus) plant set out as bait for mycoplasmas in a blueberry (Vaccinium) field. The experimental plant host range of WTV is wide and encompasses many dicotyledonous plants. The name of this virus derives from the fact that infected plants develop phloem-derived galls (tumors) at wound sites, notably at the emergence of side roots.
RDV and RGDV have narrow and overlapping host ranges. RDV causes severe disease in rice crops in South-East Asia, China, Japan and Korea, Nepal and the Philippines. RGDV has been reported in Thailand, Malaysia and China. RDV induces white flecks and streaks on leaves, with stunting and excessive production of side shoots. RDV is the only plant reovirus that is not limited to the phloem. Plants infected with RDV are stunted and fail to bear seeds. Since the virus is widespread among rice plants in southern China and other Asian countries, it is considered likely to be the cause of a significant overall reduction in rice production. RDV does not provoke enlargement or division of infected cells and does not induce galls, enations, or tumors. RGDV was found in a rice field in Thailand and induces stunting, shoot proliferation, a dark green color and enations in rice.
Phytoreoviruses induce no marked disease in the insect vectors. Virus replication occurs in the cytoplasm of infected cells in association with viroplasm. In the vector, there are no particular tissue tropisms. However, RDV induces abnormalities in fat body cells and mycetocytes. They are all transmitted propagatively by cicadellid leafhoppers (Hemiptera, Cicadellidae, e.g., Agallia, Agalliopsis, Nephotettix and Recilia). Virus is acquired from plants shortly after feeding. The latent period in leafhoppers is about 10–20 days. Thereafter, infected insects have a lifelong ability to transmit virus to plants. Phytoreoviruses are also transmitted transovarially in their insect vectors. Experimental data suggest that phytoreoviruses are not mechanically transmissible from plant to plant. No seed transmission has been reported.
Species demarcation criteria in the genus
In addition to the other general criteria used throughout the family, members of a species in the genus Phytoreovirus may be identified by:
- Sequence analysis: Nucleotide sequence identities amongst RDV isolates from different countries are more than 90% (Table 30). Amino acid identities are >80% within species and <56% between species.
- Cross-hybridization using conditions designed to detect >80% similarity.
- Host plant species; dicotyledons (WTV), or the family Graminae (RDV and RGDV).
Table 30 Percentage nucleotide differences in genome Seg8 amongst different phytoreoviruses
| Species (virus) | RDV | RGDV | WTV | |||
| Rice dwarf virus (RDV-B) | 0.0 | 1.8 | 2.4 | 5.2 | 44.8 | 47.4 |
| Rice dwarf virus (RDV-S) |
| 0.0 | 2.7 | 5.2 | 45.0 | 47.1 |
| Rice dwarf virus (RDV-A) |
|
| 0.0 | 5.5 | 44.4 | 47.4 |
| Rice dwarf virus (RDV-China) |
|
|
| 0.0 | 45.4 | 46.5 |
| Rice gall dwarf virus (RGDV) |
|
|
|
| 0.0 | 44.9 |
| Wound tumor virus (WTV) |
|
|
|
|
| 0.0 |
List of species in the genus Phytoreovirus
| Rice dwarf virus |
|
|
| Rice dwarf virus-Akita | [Seg1: D90198, Seg2: AB263418, Seg3: X54620, Seg4: X54622, Seg5: D90033, Seg6: M91653, Seg7: D10218, Seg8: D10219, Seg9: D10220, Seg10: D10221, Seg11: D10249, Seg12: D90200] | (RDV-A) |
| {Nephotettix cincticeps, N. nigropictus, Recilia dorsalis: Gramineae} | ||
| Rice gall dwarf virus |
|
|
| Rice gall dwarf virus-Thailand | [Seg1: AB254451, Seg2: D86439, Seg3: D13774, Seg4: AB254452, Seg5: D76429, Seg6, AB254454, Seg7: AB254453, Seg8: D13410,Seg9: D01047, Seg10: D13411, Seg11: AB030009, Seg12: AB254455] | (RGDV-TH) |
| {Nephotettix cincticeps, N. nigropictus, N. virescens, N. malayanus, Recilia dorsalis: Gramineae} | ||
| Wound tumor virus |
|
|
| Wound tumor virus | [Seg4: M24117, Seg5: J03020, Seg6: M24116, Seg7: X14218, Seg8: J04344, Seg9: M24115, Seg10: M24114, Seg11: X14219,Seg12: M11133] | (WTV) |
| {Agallia constricta, A. quadripunctata, Agalliopsis novella: many dicotyledons} |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ], insect vector and host names { } and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Phytoreovirus but have not been approved as species
| Homalodisca vitripennis reovirus | [Seg1: FJ497789, Seg2: FJ497790, Seg3: FJ497791, Seg4: FJ497792, Seg5: FJ497793, Seg6: FJ497794, Seg7: FJ497795, Seg8: FJ497796, Seg9: FJ497797, Seg10: FJ497798, Seg11: FJ497799, Seg12: FJ497800] | (HoVRV) |
| {Hemiptera: Homalodisca vitripennis} | ||
| Tobacco leaf enation phytoreovirus* | [Seg5: AY587757, Seg7: AY587758, Seg8: AY587759, Seg10: AY587760, Seg11: AY587761, Seg12: AY587762] | (TLEV) |
| {tobacco: dicotyledon} |
* First phytoreovirus isolated from Africa.
Phylogenetic relationships within the genus
See Table 30 and Figure 31.
Genus Cardoreovirus
Type species Eriocheir sinensis reovirus
Distinguishing features
The cardoreovirus genome consists of 12 segments of linear dsRNA. All three members or possible members of the genus (Eirocheir sinensis reovirus (ESRV), Macropipus depurator reovirus and Carcinus mediterraneus reovirus (CMRV)) have been isolated from crabs. Intact virus particles show capsomeric structures by negative staining and transmission electron microscopy (TEM) (Figure 32). Replication is accompanied by production of filaments, tubules and viral inclusion bodies (VIB).
Virion properties
Morphology
Purification of ESRV or CMRV particles on CsCl, and analysis by TEM and negative staining, revealed intact icosahedral particles about 70 nm in diameter with a morphology typical of non-turreted reovirus cores (Figure 32). In particular, the morphology of ESRV was similar to that observed for the seadornaviruses and orbiviruses. Negative staining showed subunits 8 to 9 nm in diameter on the surface of the particles. Empty particles have a hexagonal electron-dense (stained) centre, 50 nm in diameter, surrounded by an 8 to 10 nm thick capsid. Some of the empty capsids appear as two concentric layers of similar thickness. The double layered core is surrounded by a further outer-capsid layer. Cores of particles containing RNA are about 55 nm in diameter.
Physicochemical and physical properties
The buoyant density of ESRV viral particles in CsCl is 1.39 g cm−3 for full particles and 1.29 g cm−3 for empty particles.
Nucleic acid
The genome consists of 12 segments of linear dsRNA that are numbered in order of reducing Mr, or increasing electrophoretic mobility during agarose gel electrophoresis (AGE). The genome comprises approximately 23,000 bp. The AGE electropherotype follows a 3-4-2-3 pattern. The sizes of the 12 segments are estimated as: 3.7, 3.2, 2.8, 1.85, 1.6, 1.6, 1.4, 1.2, 1.1, 0.95, 0.9 and 0.75 kb. The terminal sequences of ESRV Seg1 are 5′-GGAUUUAAAA ….AUAACAGAC-3′.
Proteins
Genome segment 1 of ESRV is the only segment so far sequenced. It encodes an RdRp of 1217 amino acid with an estimated molecular weight of 138 kDa. The structural proteins of CMRV can be separated by SDS-PAGE and six proteins have been identified by silver staining, with molecular weights of 120, 94, 76, 44, 32 and 24 kDa.
Lipids
None reported.
Carbohydrates
None reported.
Genome organization and replication
Replication of cardoreoviruses occurs in the cytoplasm of crab cells (there is no available cell culture system which support growth of cardoreoviruses), where normal virions can associate in rosettes. During the early stages of infection, virions appear within viral inclusion bodies (VIB), which are dispersed in several regions of the cytoplasm. Viruses accumulate in rosettes around VIBs. Filaments and tubular structures have also been observed in the cytoplasm of infected cells. Infected tissues appear to be progressively destroyed and their normal structures are replaced by disorganized cell debris and cells derived from blood or connective tissues.
Antigenic properties
None reported.
Biological properties
Cardoreoviruses have only been isolated from diseased crabs. Experimental inoculation of the viruses into healthy crabs caused symptoms of infection which included absence of aggressiveness, increasing weakness and lack of appetite. Infected crabs survived from 7 to 20 days. Connective tissue of many organs including the hepatopancreas, digestive tract, gills and hematopoietic organs showed severe damage. The most obvious lesions were observed in connective tissue surrounding tubules of the hepatopancreas, although all cellular types were progressively destroyed. Connective tissue was replaced by isolated cells, degenerative cells and debris which did not exhibit any organization. Inside these necrotic areas, nodules appeared which consisted of debris surrounded by aggregated haemocytes. Epithelial cells of the digestive tract, gills and hepatopancreas did not seem to be affected by the virus.
Species demarcation criteria in the genus
Not applicable.
List of species in the genus Cardoreovirus
| Eriocheir sinensis reovirus |
|
|
| {Chinese mitten crab} |
|
|
| Eriocheir sinensis reovirus | [Seg1: AY542965] | (ESRV) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ], host names { } and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Cardoreovirus but have not been approved as species
| Carcinus mediterraneus reovirus (isolate W2) |
| (CMRV) |
| Macropipus depurator reovirus (isolate P) |
| (MDRV) |
Phylogenetic relationships within the genus and with other members of subfamily Sedoreovirinae
Since the first crab virus was reported in 1966, numerous viruses from crabs have been described. Most have been associated with marine crabs and only a few have been reported from freshwater crustaceans. The three crab reovirus isolates described (ESRV, Macropipus depurator P virus and CMRV) have been isolated from three different crab species (Eriocheir sinensis, Macropipus depurator and Carcinus mediterraneus respectively). All three viruses have multi-segmented dsRNA genomes made of 12 segments of linear dsRNA but so far only one segment of ESRV has been sequenced and the relationship between the three viruses is uncertain. Their genome electrophoretic profiles are distinct from the other members of family Reoviridae with 12 segmented genomes, namely the phytoreoviruses (plant viruses) and seadornaviruses (insect-borne arboviruses). In comparisons of the ESRV RdRp with other reoviruses only a distant relationship with the seadornaviruses was identified (aa identity as low as 27%), suggesting that they have a distant but common origin.
Genus Mimoreovirus
Type species Micromomonas pusilla reovirus
Distinguishing features
The mimoreovirus genome consists of 11 segments of linear dsRNA. The only known representative of this genus is Micromonas pusilla reovirus (MpRV) which has been isolated from the marine protist Micromonas pusilla. MpRV does not grow in mammalian or fish cells lines but grows in Micromonas pusilla LAC38 strain.
Virion properties
Morphology
The virus particles, isolated on Percoll® gradients, from supernatants of infected Micromonas pusilla protest, have an average diameter of 90–95 nm (Figure 33), which is larger than any previously described member of family Reoviridae. Some damaged particles showed an outer layer of protein (ca. 15 nm thick) surrounding a more compact internal structure (ca. 75 nm diameter).
MpRV particles purified on CsCl gradients have an average diameter of 75 nm, suggesting that they have lost the outer capsid proteins, although this size is similar to that of whole particles of other reoviruses. Twin bands of viruses were recovered from CsCl gradients. The denser layer has traces of a protein of an identical size as the VP1. The protein content of these particles is similar to those of non-turreted intact particles such as rotaviruses, orbiviruses and seadornaviruses.
MpRV particles that were treated with CaCl2 then purified on Percoll® gradients have a diameter of 50 nm, showing that they have lost outer capsid proteins and may have lost other components from the underlying capsid layer. These particles have a smooth outline (no turrets; Figure 33), similar that observed for orbivirus (sub-core particles), rotaviruses and seadornaviruses.
The outer layer of the MpRV particle appears to represent a pseudo-envelope (or an additional coat) formed by the VP1 protein. Transient envelope structures have been described for orbiviruses, coltiviruses, rotaviruses and seadornaviruses as a consequence of budding of virus particles from the cell membrane or budding into the endoplasmic reticulum during morphogenesis. MpRV may be the first member of family Reoviridae to possess a constitutive pseudo-envelope structure or an additional protein coat.
Physicochemical and physical properties
Thermostability of the virus was tested at temperatures ranging from −196 °C (liquid nitrogen) to 95 °C. The virus was inactivated at temperatures above 35 °C. Freezing at temperatures below −20 °C preserved virus infectivity. Treatment with sodium dodecyl sulfate (0.1–0.5%) abolished infectivity, while treatment with non-ionic detergents such as Tween 80, NP40 and triton X-100 (0.1–1%) did not affect infectivity. Exposure to acetone or alcohol inactivated the virus while treatment with diethyl ether, chloroform or Vertrel XF preserved infectivity. These organic solvents might therefore be useful during purification. Exposure to acidic conditions (pH<5) inactivated the virus, while pH between 7 and 10 did not affect virus infectivity.
Incubation of infected Micromonas pusilla in the absence of light inhibited viral replication.
Nucleic acid
The genome consists of 11 dsRNA segments that are numbered in order of reducing Mr, or increasing electrophoretic mobility during agarose gel electrophoresis (AGE). The total genome length of MpRV is 25,563 bp, with segment lengths that range between 5792 bp and 741 bp. Electrophoretic analysis (1% AGE) of genomic RNA shows a 1-1-2-5-2 migration pattern (Figure 34). AGE profiles are thought to be characteristic of each virus species.
Sequence analysis of the MpRV genome has shown that each genome segment contains a single large ORF. The only exception is Seg5, which contains an ORF spanning nucleotides 44 to 2005 that is interrupted by an in-frame “leaky” TGA stop codon, at position 1571–1573. The G+C content of the MpRV genome segments is between 41 and 50% (the highest value found in Seg5). The conserved terminal sequences of the 11 segments are: 5′-GAAGAA/U …. A/GAAAGUC-3′.
Proteins
The protein content of virions purified using CsCl gradient centrifugation has been determined. Eight structural proteins were detected, with Mr of 200, 150, 120, 107, 67, 53, 35 and 32 kDa. The relative position of these proteins is not yet known and the role of the various structural and non-structural proteins is yet to be characterized although some putative assignments can be made based on sequence comparisons (see Genome organization and replication, below).
Lipids
None reported.
Carbohydrates
VP1 might be glycosylated based on it relatedness to mucin.
Genome organization and replication
Each of the 11 genome segments encodes a single protein except Seg5, where a “readthrough” inferred from sequence analysis may result in the production of two related proteins. The shorter 53.1 kDa protein, (VP5ter) is the early-termination translation product, while the readthrough protein (VP5rdt) is 68.9 kDa. The other proteins encoded by the different genome segments are identified as VP1-VP4 and VP6-VP11 respectively. There are thus 12 translation products (Table 31).
Sequence analysis showed that VP1 amino acids 88 to 255 showed 24% identity with the minor capsid protein sigma-1 (a hemagglutinin and responsible for cell attachment) of MRV and its equivalent sigma-c (22% identity, aa 172-321 of the VP1) of the Pulau reovirus.
VP1 shows significant aa identity with hemagglutinins from viral and non-viral origins, including those of the bacterial pathogens Burkholderia species (aa identity 20%, similarity 40%) and Staphylococcus species (aa identity 19%, similarity 38%) and the yeasts Candida albicans (identity 20%, similarity 39%) and Saccharomyces cerevisiae (identity 20%, similarity 37%). It also matched large-DNA-virus proteins, such as: (i) those of the family Phycodnaviridae including the Paramecium bursaria Chlorella virus (PBCV) Vp260 (24% identity, 39% similarity) which is a surface glycoprotein; (ii) those of family Herpesviridae including the equine herpesvirus glycoprotein 2 (gp2: identity 20%, similarity 32%) which is an envelope protein; (iii) those of bacteriophages, including the envelope protein of Acholeplasma phage L2 (identity 26%, similarity 46%). All of these glycoproteins are found in envelopes or cell wall structures.
MpRV VP1 has a high serine and threonine content (≥11% each) compared to 1 to 7.5 % for other amino acids. This is characteristic of glycoproteins and in particular for mucin and mucin-like proteins and cell wall adhesins. Such serine and threonine rich proteins are usually heavily O-glycosylated. Amino acid sequence repeats were identified within VP1. Interestingly each repeat was found to align best with a protein sequence immediately N-terminal to it in the VP1. The repeated sequences were not fully identical to the matching sequences. This suggests what has previously been described as sequence duplication in viral genes, followed by distinct and diverging evolution of both the parental and the daughter repeat sequences.
In summary, VP1 is considered likely to form the outermost surface of the virus, possibly representing an extra coat in comparison to other reoviruses.
Seg2 of MpRV encodes VP2, which is thought to be the viral RdRp (or Pol). RdRp core motifs identified in VP2, include the motif SG (position 801-802) and the motif GDD (position 835-837). A partial match (aa 647–962, identity 21%) was also found within the enzyme “core” region, with the RdRp of human rotavirus C (accession number CAC44891), another 11 segmented dsRNA virus belonging to family Reoviridae.
Seg3 of MpRV encodes VP3, which by analogy with other reoviruses, appears likely to represent the structural protein which forms the inner capsid shell (sub-core). In other reoviruses this layer has been shown to have pseudo T=2 icosahedral symmetry (also described as a modified T=1 symmetry) and the sub-core shell proteins is also identified as the T2 protein. VP3 was found to partially (aa 229–311, identity 26%) match the P3(T2) of rice dwarf virus (phytoreovirus), lambda-1(T2) of MRV-3 (aa 50-145, identity 20%). Lambda-1 of MRV-3 also possesses NTPase and helicase activities).
Seg5 of MpRV encodes VP5, which was found to partially match (aa 214–318, 21% identity) the outer capsid spike protein VP4 of rotavirus A. It also showed 24% identity with the killer toxin protein (accession number S51548) of the dsRNA M28 satellite of Saccharomyces cerevisiae L-A virus.
Seg7 of MpRV encodes VP7, which partially matches (aa 130–209, 32% identity) the non-structural protein NS1 of cypovirus type 1, while VP8 of MpRV (encoded by Seg8) partially matches (aa 42–66, 28% identity) NSP2 of human rotavirus A (NSP2 has a dsRNA helix destabilisation activity, binds RNA and is an NTPase).
Seg9 of MpRV encodes VP9, which partially matches (aa 269–338, 28% identity) the protein encoded by Seg7 of Nilaparvata luguens reovirus (genus Fijivirus), which is a core protein with nucleotide-binding activity.
No significant matches were found for VP4, VP6, VP10 and VP11.
Table 31 Genome segments and protein products of Micromonas pusilla reovirus
| Genome segment | Size (bp) | Protein | Protein size (kDa), calculated from RNA sequences | Structure/putative function |
| Seg1 | 5792 | VP1 | 201.35 | Outer layer/pseudo-envelope |
| Seg2 | 4175 | VP2 | 154.69 | RNA-dependent RNA polymerase (RdRp or Pol) |
| Seg3 | 3129 | VP3 | 116.27 | Sub-core “2” layer |
| Seg4 | 2833 | VP4 | 102.64 | Unknown |
| Seg5 | 2027 | VP5(tr) / VP5(rdt) | 53.17 / 68.93 | Similar to rotavirus VP4 outer capsid protein |
| Seg6 | 1687 | VP6 | 59.0 | Unknown |
| Seg7 | 1556 | VP7 | 55.41 | Show similarity to cypovirus NS1 |
| Seg8 | 1449 | VP8 | 51.69 | Show similarity to rotavirus NSP2 |
| Seg9 | 1296 | VP9 | 44.31 | Show similarity to nucleotide binding core-protein of fijiviruses |
| Seg10 | 878 | VP10 | 24.69 | Unknown |
| Seg11 | 741 | VP11 | 22.25 | Unknown |
Antigenic properties
None reported.
Biological properties
MpRV was found in a sample of the marine protist Micromonas pusilla, along with a larger dsDNA virus particle (size 100–140 nm). However, the larger virus type was removed by passage through a 0.1 Am pore-size filter and end-point dilution. Ten virus clones were obtained, all showing comparable particle size and host specificity as well as similar behavior on infection. It did not infect insect or mammalian cells and, out of six strains of M. pusilla, only strain LAC38 supported replication of the virus, suggesting a high degree of strain specificity. LAC38 originates from Norwegian coastal waters, while the other five strains were isolated from different locations in the English Channel, English coastal waters, or the Gulf of Maine in the United States.
The growth of M. pusilla was inhibited by addition of MpRV within 24 h. The number of free virus particles started to increase at 36 h post infection. A decline in algal cell numbers was also observed by 40 h post infection, with the percentage of dead algal cells steadily increasing, to match an increase in virus released from the host cells (>60% dead cells). The estimated burst size was 460–520 virus particles per lysed algal cell.
Species demarcation criteria in the genus
Not applicable.
List of species in the genus Mimoreovirus
| Micromonas pusilla reovirus |
|
|
| {Marine protist} |
|
|
| Micromonas pusilla reovirus | [Seg1: DQ126101, Seg2: DQ126102, Seg3: DQ126103, Seg4: DQ126104, Seg5: DQ126105, Seg6: DQ126106, Seg7: DQ126107, Seg8: DQ126108, Seg9: DQ126109, Seg10: DQ126110, Seg11: DQ126111] | (MpRV) |
Species names are in italic script; names of isolates are in roman script. Sequence accession numbers [ ], host { } and assigned abbreviations ( ) are also listed.
List of other related viruses which may be members of the genus Mimoreovirus but have not been approved as species
None reported.
List of unassigned species in the family Reoviridae
None.
List of other related viruses which may be members of the family Reoviridae but have not been approved as species
| Viruses of Arthropoda |
|
|
|
| Cimex lactularius reovirus | {Cimex lactularius (Hemiptera: bed bug)} | (ClRV) | 11 segments, icosahedral double-shelled capsid about 50 nm diameter |
| Viruses of Crustacea |
|
|
|
| Porcelio dilatatus reovirus | {Porcelio dilatatus (Isopoda: terrestrial crustacean)} | (PdRV) | Uncharacterized |
| Viruses of Arachnida |
|
|
|
| Buthus occitanus reovirus | {Buthus occitanus (Scorpionidae: scorpion)} | (BoRV) | Uncharacterized |
Source or host species { }, assigned abbreviation ( ) and characteristics are also given.
Phylogenetic relationships in the family
The Reoviridae represents the largest family of dsRNA viruses. It contains 15 genera of viruses having genomes composed of 9, 10, 11 or 12 segments of linear dsRNA. Member viruses have been isolated from a wide range of mammals, birds, reptiles, fish, crustaceans, marine protists, insects, ticks, arachnids, plants and fungi and include a total of 75 virus species.
Phylogenetic analyses, using amino acid sequences of the RNA-dependent RNA polymerase, have shown that the different genera of reoviruses exhibit amino acid sequence identities of less than 30%. There are two exceptions: (i) Rotavirus B, shows only 22% identity with other rotaviruses, (ii) Aquareovirus and Orthoreovirus, show an amino acid sequence identity of up to 42%. In most cases the phylogenetic tree of the polymerase (shown in Figure 35), clearly identifies the turreted viruses and non-turreted viruses as members of separate clades. However, it is noteworthy that the functional core of the RdRp contains similar motifs across the entire family (Table 32).
Overall phylogenetic analyses support the co-speciation hypothesis that reoviruses have co-evolved with their respective hosts and/or arthropod vectors. Although reoviruses form a single monophyletic group, they have been evolving for over about 550 million years, resulting in sequence divergence to near randomness and clear structural differences between the more distantly related genera. However, the replication mechanisms used and certain structural parameters of the internal proteins (of the subcore shell and polymerase complexes) remain fundamentally and remarkably similar. Evolutionary analyses suggest that the non-turreted viruses may represent an ancestral lineage from which the turreted viruses have evolved.
Closer relations between certain genera have been identified by structural analyses of the viral proteins, and may be indicated by the presence of signature sequences. For example, comparisons of polymerase, capping enzyme and capsid protein sequences, as well as structural analyses of outer capsid proteins, suggest that an evolutionary jump has occurred between the seadornaviruses and rotaviruses, within the Sedoreovirinae. This is thought to have involved gene duplication and rearrangement, changing the number of genome segments (from 11 for the rotavirus to 12 for the seadornaviruses).
Closer relationships have also been identified between the aquareoviruses and orthoreoviruses within the Spinareovirinae, with homologous proteins exhibiting up to 42% amino acid identity and particle morphology that is super-imposable (revealed by cryoEM). Gene duplication followed by rearrangement, also appears to have occurred between the aquareoviruses and coltiviruses, increasing the number of segments from 11 (aquareoviruses) to 12 (coltiviruses).
Table 32 Comparison of sequences surrounding the conserved RdRp motifs of reoviruses
| Motifs | I | IV,1,A | V,2,B | VI,3,C | D |
| Consensus | grrtRiI | D.s.wd. | SGe.aTs.a..nla | qvqGDDtlm.ikdg | he.n.sK.s |
| BTV (515) | PIKATRTI 72 | DYSEYDTH 119 | SGENSTLIANSMHNMA 21 | EQYVGDDTLFYTKLD 22 | HEASPSKTM (804) |
| Rotavirus (455) | PGRRTRII 57 | DVSQWDSS 63 | SGEKQTKAANSIANLA 19 | IRVDGDDNYAVLQFN 20 | RMNAKVKAL (669) |
| RDV (643) | AWRPVRPI 73 | DCTSWDQT 76 | SGRLDTFFMNSVQNLI 20 | FQVAGDDAIM.VYDG 24 | HIINPQKTV (890) |
| Reovirus S3 (521) | VQRRPRSI 56 | DISACDAS 89 | SGSTATSTEHTANNST 31 | YVCQGDDGLM.IIDG 21 | GEEFGWKYD (772) |
| NLRV (646) | IDRRGRII 60 | DMSGMDAH 90 | SGLFATSGQHT.MFLV 20 | NYVMGDDIFQNIKNG 24 | IDGNYSKYS (894) |
| RRSV (500) | IGRRQRAI 62 | DASVQASV 83 | SGQPFTTVHHTFTLSN 1 | LTVQGDDTRT.INYG 15 | VSDWGFKVS (735) |
| BAV (557) | LVRGTRAK 74 | DTSQWGQI 62 | SGELTTQTRNTTTNIS 25 | DNKVGDDSVEVLRVV 24 | HLEISAKRTI (415) |
The regions covering the putative polymerase module in RRSV P4 (aa 500 to 735) and other reoviruses were analyzed using the GCG program PILEUP and further aligned manually taking into account the polymerase motifs presented and aligned by Poch et al. (1989) (A–D), Bruenn (1991) (1–3) and Koonin (1992) (I, IV–VI).References:Bruenn, J. (1991). Relationships among the positive strand and double-strand RNA viruses as viewed through their RNA-dependent RNA polymerases. Nucleic Acids Res., 19, 217–226.Koonin, E.V. (1992). Evolution of double-stranded RNA viruses: a case for polyphyletic origin from different groups of positive-stranded RN A viruses. Semin. Virol., 3, 327–339.Poch, O., Sauvageut, I., Delarue, M. and Tordo, N. (1989). Identification of four conserved motifs among the RNA-dependent polymerase encoding elements. EMBO J., 8, 3867–3874.
Similarities with other taxa
Although there is little evidence for nucleotide or amino acid sequence similarities with other families of dsRNA viruses, it may be significant that some (e.g. the families Cystoviridae and Totiviridae) also have particles in which the inner shell is characteristically composed of 120 copies of a triangular protein, arranged in a manner similar to that of the members of the family Reoviridae. This protein provides an apparently simple yet elegant mechanism of assembling the inner icosahedral capsid shell, which has been described alternatively as having T=1 or T=2 symmetry, although it is important to note these are different academic interpretations of a similar particle architecture. These similarities may also indicate a common, if distant, ancestry and again suggest that these viruses may even have diversified and evolved along with their host species.
Derivation of names
Cardo: from Carcinus (crab) and dodeca, from Ancient Greek dodeka, “12”.
Cypo: from cytoplasmic polyhedrosis.
Dinoverna: from double stranded insect novem (“nine” in Latin) segmented RNA viruses.
Fiji: from the country where the virus was first isolated.
Idno: from insect-derived non-occluded (in contrast to the cypoviruses).
Mimo: from Micromonas, the genus name of the type member.
Myco: from myco, Latin for “fungus”.
Orbi: from orbis, Latin for “ring” or “circle”, in recognition of the ring-like structures observed in micrographs of the surface of core particles.
Ortho: from orthos, Greek for “straight”.
Oryza: from the genus name of rice, the host of the type member.
Phyto: from phyton, Greek for “plant”.
Reo: from respiratory enteric orphan, due to the early recognition that the viruses caused respiratory and enteric infections, and the (incorrect) belief that they were not associated with disease and so were considered orphan viruses.
Rota: from rota, Latin for “wheel”.
Seadorna: from South-Eastern Asia dodeca RNA viruses.
Sedo: from sedo, Latin for “smooth”.
Spina: from spina, Latin for “spike”.
Further reading
A supplementary list of references is available online on Science Direct®, www.sciencedirect.com.
Day, 2009 J.M. Day, The diversity of the orthoreoviruses: molecular taxonomy and phylogentic divides. Infect. Genet. Evol. 9 (2009) 390–400.
Guglielmi et al., 2006 K.M. Guglielmi, E.M. Johnson, T. Stehle, T.S. Dermody, Attachment and cell entry of mammalian orthoreovirus. Curr. Top. Microbiol. Immunol. 309 (2006) 1–38.
Kim et al., 2004 J. Kim, Y. Tao, K.M. Reinisch, S.C. Harrison, ML. Nibert, Orthoreovirus and Aquareovirus core proteins: conserved enzymatic surfaces, but not protein-protein interfaces. Virus Res. 101 (2004) 15–28.
Matthijnssens et al., 2008 J. Matthijnssens, M. Ciarlet, M. Rahmanm, H. Attouim, K. Bányaim, M.K. Estesm, J.R. Gentschm, M. Iturriza-Gómara, C.D. Kirkwood, V. Martella, P.P. Mertens, O. Nakagomi, J.T. Patton, F.M. Rugger,i, L.J. Saif, N. Santos, A. Steyer, K. Taniguchi, U. Desselberger, M. Van Ranst, Recommendations for the classification of group A rotaviruses using all 11 genomic RNA segments. Arch. Virol. 153 (2008) 1621–1629.
Mertens and Diprose, 2004 P.P. Mertens, J. Diprose, The bluetongue virus core: a nano-scale transcription machine. Virus Res. 101 (2004) 29–43.
Nibert et al., 1991 M.L. Nibert, D.B. Furlong, B.N. Fields, Mechanisms of viral pathogenesis. Distinct forms of reoviruses and their roles during replication in cells and host. J. Clin. Invest. 88 (3) (1991) 727–734.
Roy, 2008 P. Roy, Bluetongue virus: dissection of the polymerase complex. J. Gen. Virol. 89 (2008) 1789–1804.
Roy, 2008 P. Roy, Functional mapping of bluetongue virus proteins and their interactions with host proteins during virus replication. Cell Biochem. Biophys. 50 (2008) 143–157.
Stuart and Grimes, 2006 D.I. Stuart, J.M. Grimes, Structural studies on orbivirus proteins and particles. Curr. Top. Microbiol. Immunol. 309 (2006) 221–244.
Contributed by
Attoui, H., Mertens, P.P.C., Becnel, J., Belaganahalli, S., Bergoin, M., Brussaard, C.P., Chappell, J.D., Ciarlet, M., del Vas, M., Dermody, T.S., Dormitzer, P.R., Duncan, R., Fang, Q., Graham, R., Guglielmi, K.M., Harding, R.M., Hillman, B., Makkay, A., Marzachì, C., Matthijnssens, J., Milne, R.G., Mohd Jaafar, F., Mori, H., Noordeloos, A.A., Omura, T., Patton, J.T., Rao, S., Maan, M., Stoltz, D., Suzuki, N., Upadhyaya, N.M., Wei, C. and Zhou, H.
Figures
Figure 1 (Top and center) A comparison of two distinct core particle morphologies (spiked and unspiked) present amongst members of different genera of the family Reoviridae. Orbivirus: a 3D model from x-ray crystallography of the core particle of an isolate of bluetongue-1 virus. Orthoreovirus: a 3D model from x-ray crystallography studies of a core particle of an isolate of mammalian orthoreovirus 3. Cypovirus: a 3D cryoEM reconstruction of a particle of an isolate of Cypovirus 5, at 25 resolution. Rotavirus: a 3D cryoEM reconstruction of a double shelled particle of an isolate of rotavirus A (SiRV-A/SA11), at 25 resolution. Fijivirus: an electron micrograph of a core particle of an isolate of maize rough dwarf virus. Phytoreovirus: a 3D cryoEM reconstruction of the double shelled particle of an isolate of rice dwarf virus, at 25 resolution (highlighted in colour are a contiguous group of 5 trimers found in each asymmetric unit). Coltivirus: an electron micrograph of a negatively stained double shelled particle of an isolate of Colorado tick fever virus. Oryzavirus: an electron micrograph of a negatively stained core particle of an isolate of rice ragged stunt virus. Mycoreovirus: an electron micrograph of a negatively stained core particle of mycoreovirus 1 (Rosallinia necatrix mycoreovirus-1). Seadornavirus: an electron micrograph of a negatively stained core particle of an isolate of Banna virus. The reconstructions and electron micrographs are not shown to exactly the same scale. The outer capsid morphologies of members of the different genera of the family Reoviridae are more variable and may appear smooth, or with surface projects, or may even be absent. (Bottom) A diagrammatic representation of the core particles (on the left) of an orbivirus (BTV), or rotavirus (RV), which have a well defined capsomeric structure but lack large surface projections at the five-fold icosahedral axes, as compared to the turreted (spiked) core particle (on the right) of an orthoreovirus (Reo).
(Courtesy of J. Diprose.)

Figure 2 Typical virus replication cycle of a reovirus (presented for an orbivirus).
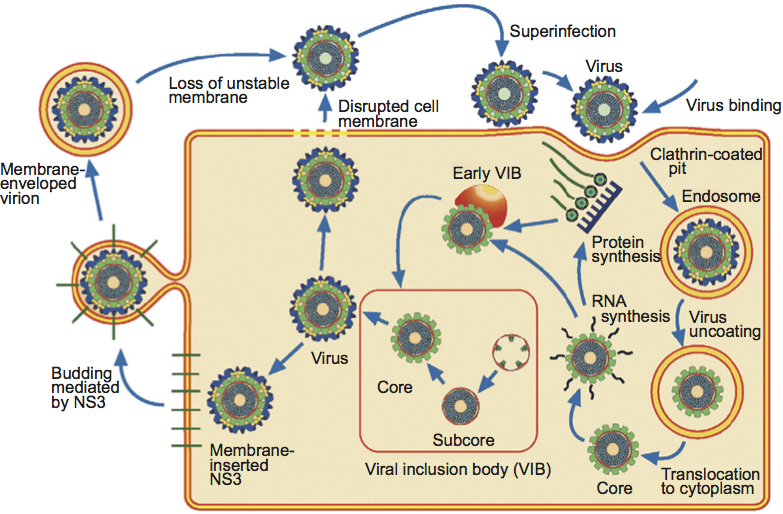
Figure 3 (Top left) Diagrammatic representation of an orthoreovirus particle in cross-section. The locations and identities of the virus structural proteins are indicated using the nomenclature scheme for both mammalian orthoreovirus (MRV) and avian orthoreovirus (ARV). The protein components of the inner and outer capsids are indicated (Duncan, R. (1999). Virology, 260, 316-328; Palmer, E.L. and Martin, M.L. (1977). Virology, 76, 109-113). (Top right) Computer-generated image of the inner capsid of mammalian orthoreovirus 1 (MRV-1), based on X-ray crystallography data. (Bottom) Electron micrograph of a negatively stained MRV-1 particle (panel A). Image reconstructions from cryoEM of MRV-1 virions (panel B), infectious subviral particles (ISVPs) (panel C) and cores (panel D). All particles are viewed from the three-fold axis of rotational symmetry.
(Courtesy of M. Nibert and T. Baker.)
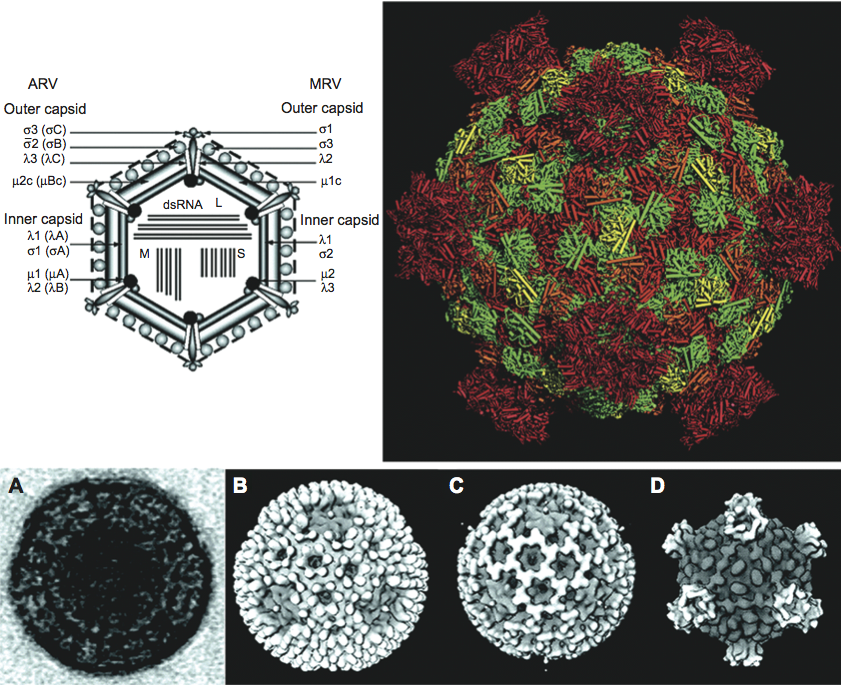
Figure 4 Gene organization of the polycistronic genome segments of the five species of orthoreoviruses. The solid line indicates the dsRNA, and the numbers refer to the first and last nucleotides of the genome segment, along with the nt positions of the various ORFs (excluding the termination codons) indicated by the rectangles. The identities of the gene products encoded by the various ORFs are indicated within the rectangles. The virus species and the genome segment are indicated on the left. The code for the abbreviations can be found in the list of species.
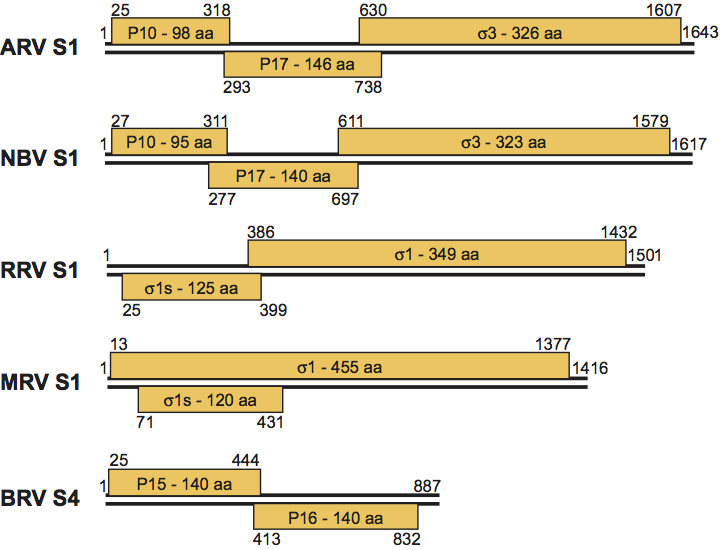
Figure 5 Phylogenetic relationships between the five orthoreovirus species using the aa sequences of the sigma-class major outer CPs of various isolates. Sequences were aligned using ClustalX and the unrooted neighbor-joining tree constructed using MEGA4 (Dayhoff distances and 10,000 bootstrap replicates). Accession numbers used were ARV-13 (AF059271), ARV-11 (U20642), ARV-Tu (AF4645799), ARV-Md (AJ006476), ARV-Go (AY114138), BRV (AF059723), MRV-1 (X61586), MRV-2 (X60066), MRV-3 (HM159622), MRV-4 (AF368037), NBV (AF059722) and RRV (AY238886).
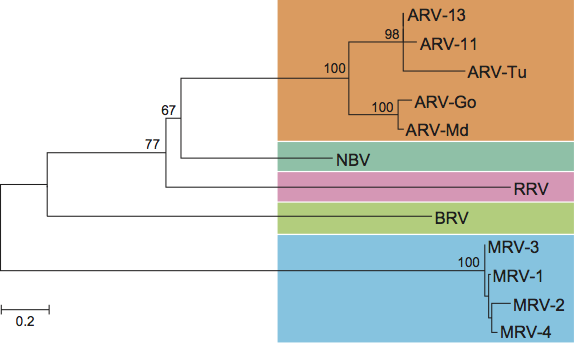
Figure 6 (Top panel) Structural representation of grass carp reovirus virion and core by cryoEM. Triangles represent the VP5VP7 complex on the virion (from Cheng, L., Fang, Q., Shah, S., Atanasov, I. C. and Zhou Z. H. (2008). J. Mol. Biol., 382, 213222). (Middle panel) Transmission electronmicroscopy (TEM) of negatively stained grass carp reovirus (GCRV) particles. From left to right: intact virion, core and top components (empty particles). The scale bar represents 100 nm (from Fang, Q., Seng, E. K., Ding, Q. Q. and Zhang, L. L. (2008). Arch. Virol., 153, 675682). (Bottom panel) Complete atomic model of grass carp infectious subviral particle. In the right hand side of the CryoEM reconstruction the removal of the VP5 coat reveals the core proteins. Ribbon models of the atomic structures of the six conformers from four structural proteins are shown in the periphery. The black triangle encloses a VP5 trimer
(from Zhang et al. (2010). Cell, 141, 472482).
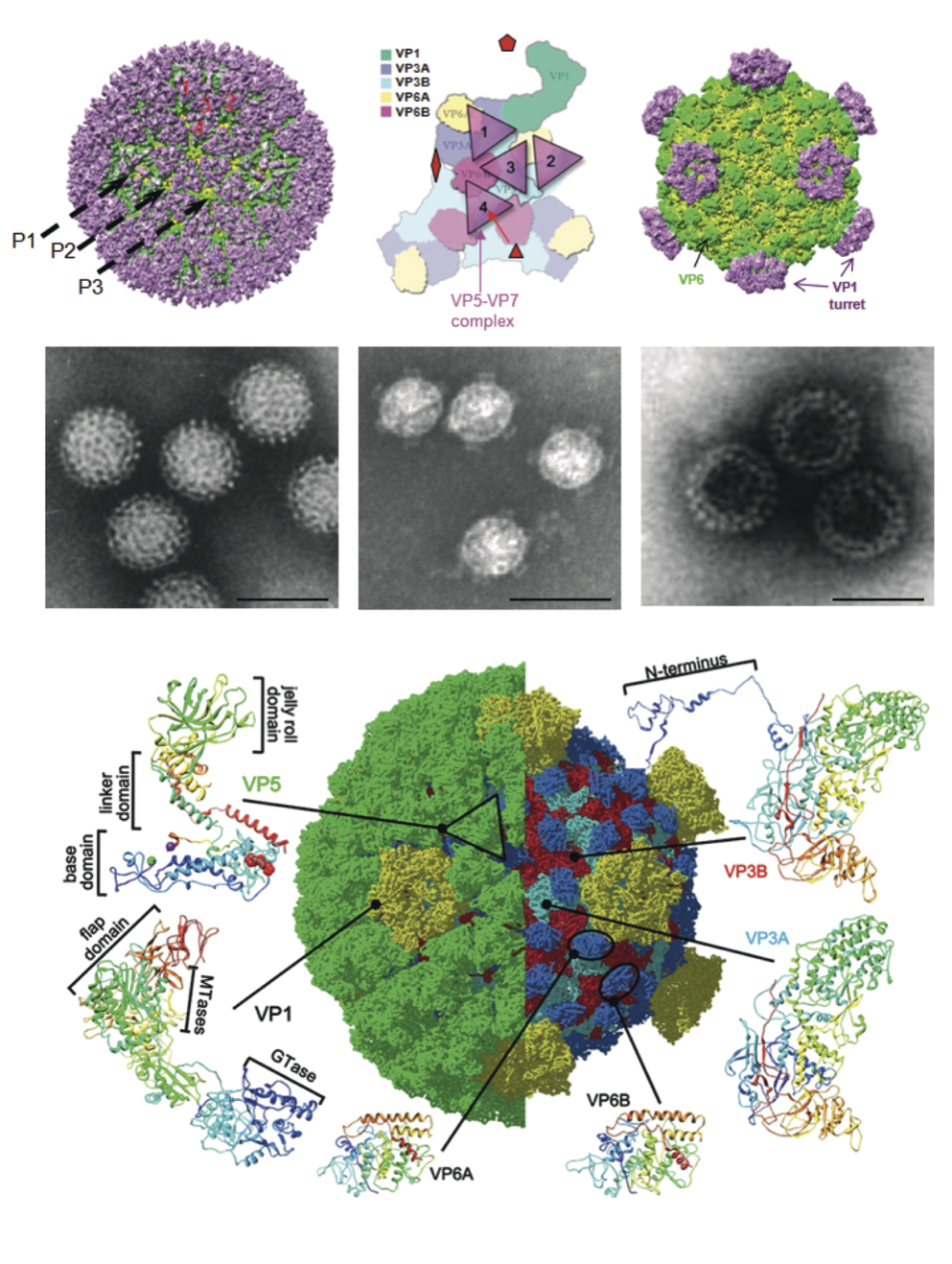
Figure 7 Genome organization of the 11 dsRNA segments of golden shiner virus (species Aquareovirus C). Each segment has a single ORF, except Seg7 which contains two ORFs. The green arrows indicate the upstream conserved terminal sequence (+ve 5-GUUAUUU/G.) while the red arrows indicate the downstream conserved terminal sequence (+ve .A/UUCAUC-3).
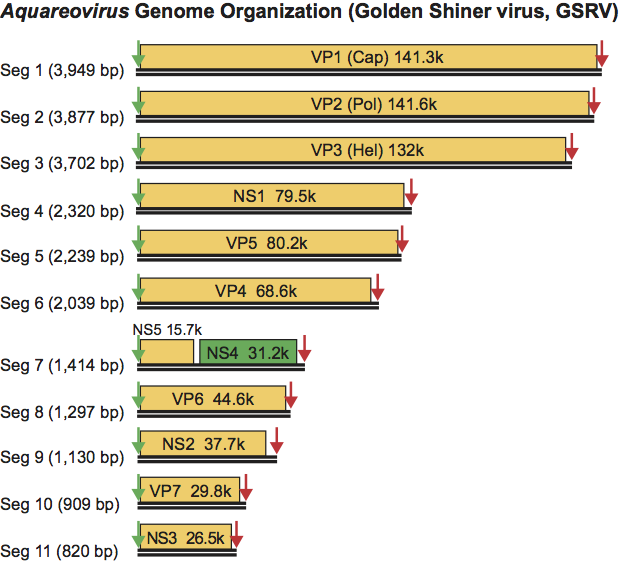
Figure 8 (Top left) Electron micrograph of rice ragged stunt virus (RRSV) particles (courtesy of R.G. Milne). (Bottom left) schematic of RRSV particle; (right panel) micrographs of the virus showing 2-, 3- and 5-fold symmetries (A1, B1 and C1, respectively) images of the same rotated by increments of 180 (A2), or 120 (B2), or 72 (C2) and proposed models of the 2-, 3- and 5-fold symmetries (A3, B3 and C3 respectively) (courtesy of E. Shikata). The bar represents 50 nm.
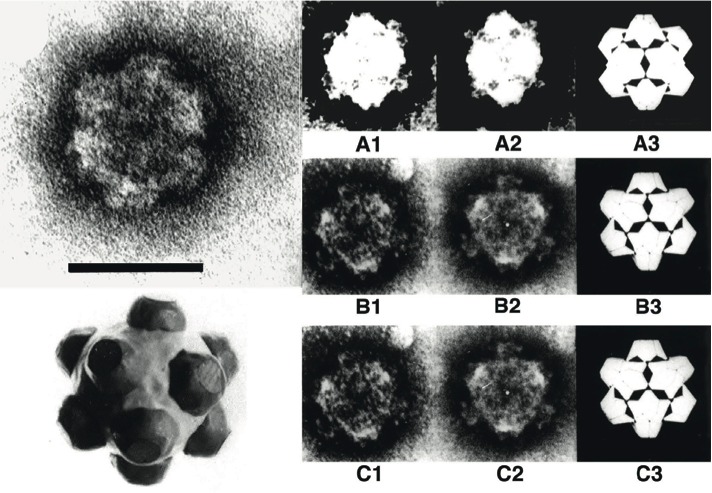
Figure 9 (Left) Negative contrast electron micrograph of maize rough dwarf virus (MRDV) virions stained with uranyl acetate showing A spikes; (center) smooth subcores derived from MRDV on staining with neutral phosphotungstate; (right) B spikes on virus-derived MRDV cores stained with uranyl acetate (courtesy of R. G. Milne). The bar represents 100 nm.
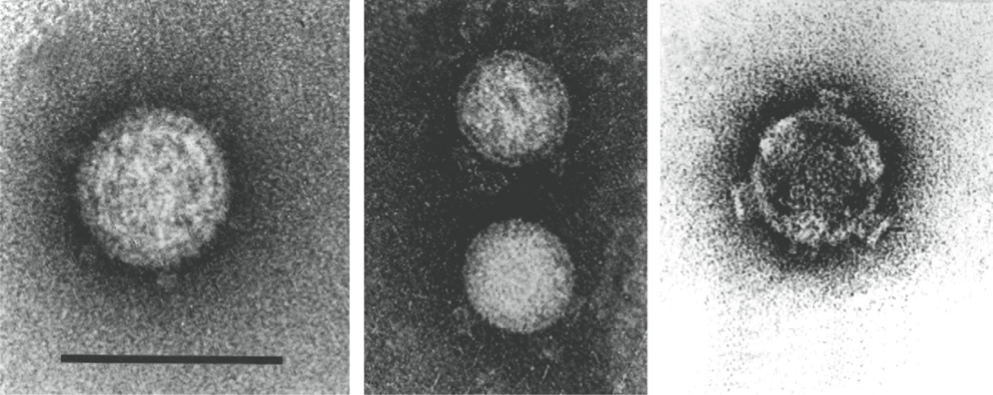
Figure 10 Neighbor-joining phylogenetic tree constructed using the amino acid sequences of outer capsid proteins of fijiviruses. (Fiji disease virus, FDV (Seg10) [AY297694]; maize rough dwarf virus, MRDV (Seg10) [L76560]; rice black streaked dwarf virus, RBSDV (Seg10) [D00606]; oat sterile dwarf virus, OSDV (Seg8) [AB011025]; Nilaparvata lugens reovirus, NLRV (Seg8) [D26127]; mal de Rio Cuarto virus, MRCV (Seg10) [AY607586]; southern rice black streaked dwarf virus, SRBSDV (Seg10) [EU784840]). Sequences were aligned using ClustalX and the tree constructed using MEGA4 (Dayhoff distances and 10,000 bootstrap replicates).
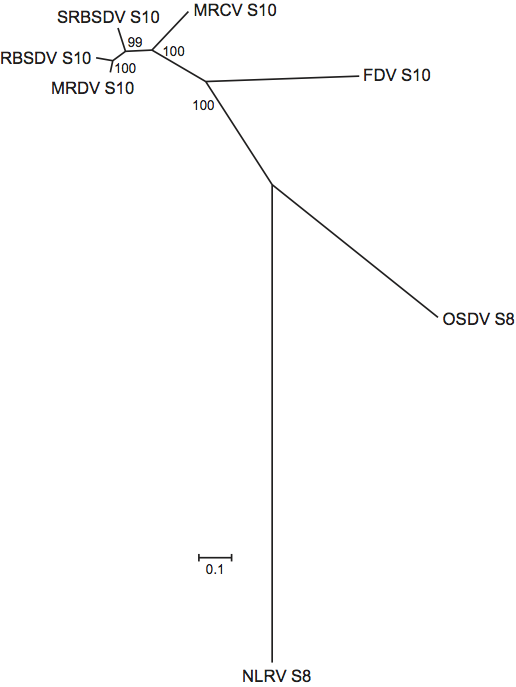
Figure 11 (Left) Electron micrograph of virus particles of Cryphonectria parasitica mycoreovirus-1 (CpMYRV-1) after purification by sucrose gradient centrifugation, stained with 1% uranyl acetate (courtesy of B. Hillman). (Right) Core particle of Rosellinia necatrix mycoreovirus-3 (RnMYRV-3) showing icosahedral arrangement surface projections (turrets or spikes), stained with 1% uranyl acetate (courtesy of C. Wei). The bar (right panel) represents 50 nm.
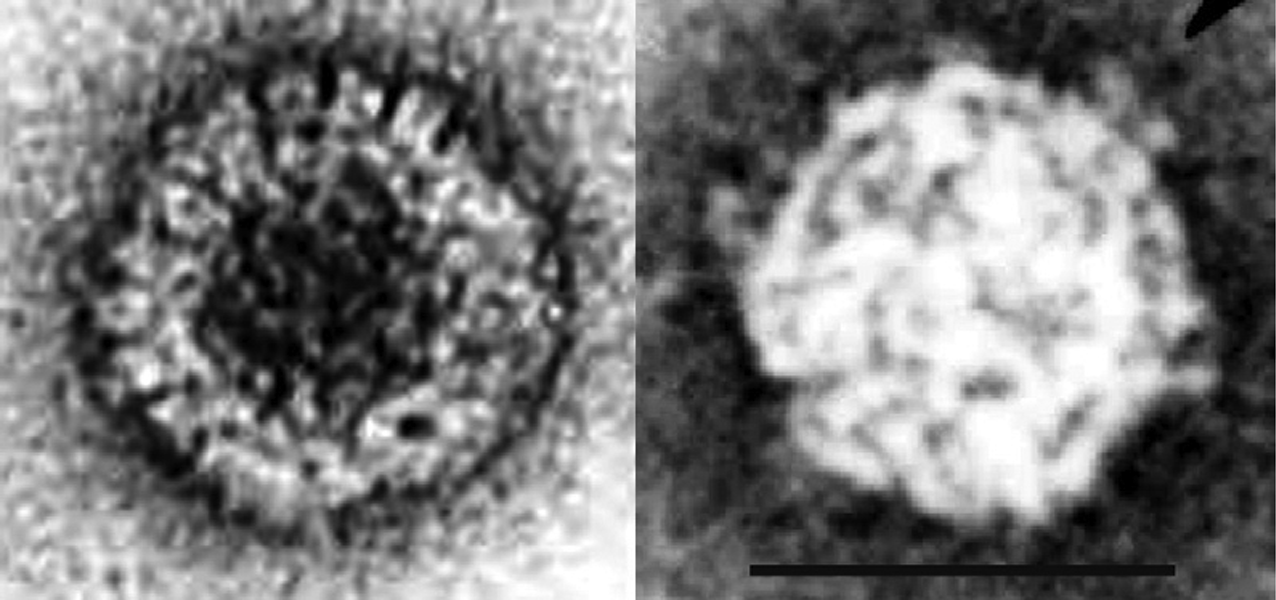
Figure 12 (Left) Negative contrast electron micrograph of a non-occluded virion of Orgyia pseudosugata cypovirus 5 (OpCPV-5). (Right) Negative contrast electron micrograph of empty and full occluded virions (purified from polyhedra) of OpCPV-5, stained with uranyl acetate (courtesy of C. L. Hill). The bars represent 20 nm.

Figure 13 CryoEM reconstructions of Orgyia pseudosugata cypovirus 5 (OpCPV-5) virions, to 26 resolution: (top left) non-occluded virion; (top right) occluded virion; (bottom left) cross-section of a full occluded virion; (bottom center) cross-section of a full non-occluded virion; (bottom right) cross-section of an empty virion. The cross-sections show evidence of dsRNA packaged as distinct layers and suggest localization of the transcriptase complexes at the five-fold axes of symmetry.
(Courtesy of C. L. Hill.)
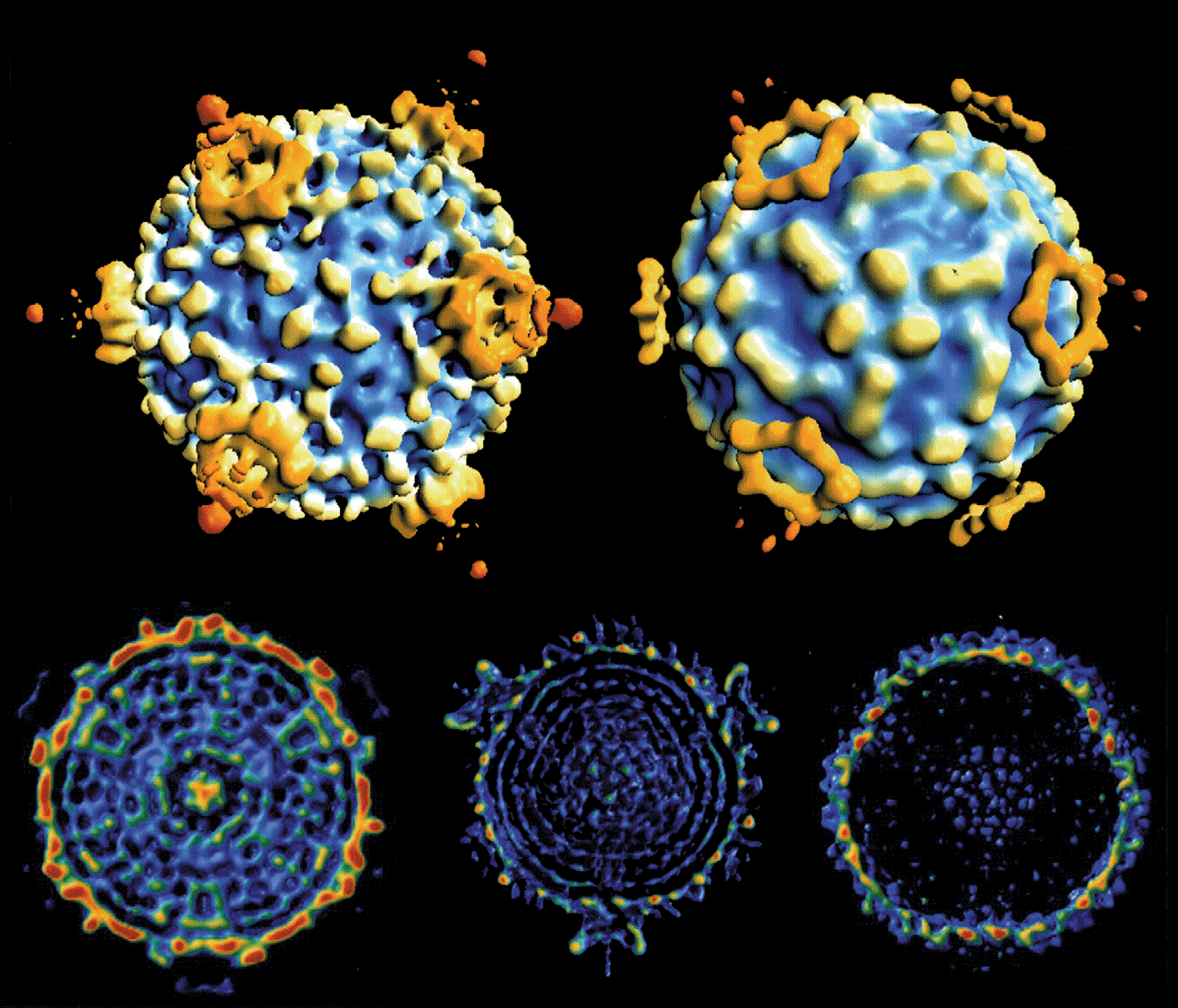
Figure 14 Phylogenetic tree for polyhedrin proteins from 11 cypovirus isolates. Sequences were aligned using ClustalX and the tree constructed in MEGA4 using the neighbour-joining method and the P-distance algorithm. Branching is supported by bootstrap values >85%.
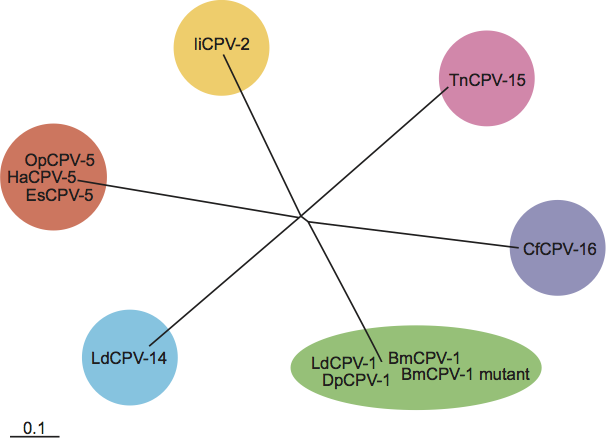
Figure 15 Electron micrographs of purified virus particles (far left) and core particles (second left) of Hyposoter exiguae idnoreovirus-2 (HelRV-2), stained with uranyl acetate (courtesy of A. Makkay and D. Stoltz). Electron micrographs of a virus particle (second right) and core particle (far right) from purified preparations of Dacus oleae idnoreovirus-4 (DoIRV-4), stained with sodium phosphotungstate (courtesy of M. Bergoin). DoIRV-4 virions have small icosahedrally arranged surface projections (estimated 12 in number). The DoIRV-4 cores have twelve large icosahedrally arranged spikes or turrets, which (like those of the cypoviruses) may lose a portion near to the tip.
Figure 16 Negative contrast electron micrograph of particles of Aedes pseudoscutellaris reovirus purified using Iodixanol (Optiprep) gradient
(courtesy of H. Attoui).

Figure 17 Agarose gel electrophoretic profiles of genome segments of Aedes pseudoscutellaris (propagated in C6/36 cells) in 1% agarose gel. These migration patterns (electropherotype) are thought to be characteristic of each virus species.
Figure 18 Negative contrast electron micrograph of particles of Colorado tick fever virus (CTFV) (courtesy of F. A. Murphy). The bar represents 50 nm.

Figure 19 Electrophoretic profiles of the genome segments of Colorado tick fever virus isolate Florio (CTFV-Fl) and California hare virus (CTFV-Ca) in 1% agarose gel. Genome migration patterns (electropherotype) are thought to be characteristic of each virus species.

Figure 20 (Top) Diagram of the bluetongue virus (BTV) particle structure, constructed using data from biochemical analyses, electron microscopy, cryoEM and X-ray crystallography (courtesy of P.P.C. Mertens and S. Archibald). (Bottom) Electron micrographs of African horse sickness virus (AHSV) serotype 9 particles stained with 2% aqueous uranyl acetate (left) virus particles, showing the relatively featureless surface structure. (Center) Infectious subviral particles (ISVP), containing chymotrypsin cleaved outer capsid protein VP2 and showing some discontinuities in the outer capsid layer. (Right) core particles, from which the entire outer capsid has been removed, to reveal the structure of the VP7(T13) core surface layer and showing the ring shaped capsomeres
(courtesy of P. P. C. Mertens).
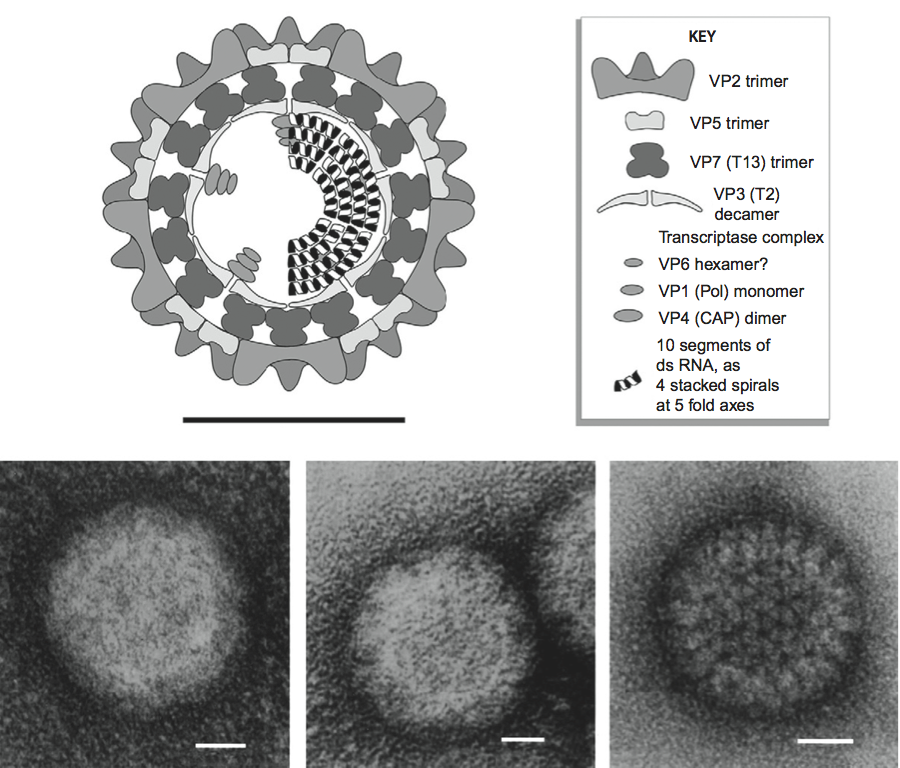
Figure 21 (Top left) The outer capsid layer of bluetongue virus (BTV) (from cryoEM) showing trimers of VP2 in red and trimers of VP5 in yellow, superimposed on the underlying X-ray crystallography structure for the BTV core. (Top right) The structure of the BTV core as determined by X-ray crystallography of the native core particle. The outer core surface, composed of 260 trimers of VP7 arranged with T=13 l symmetry. The chemically identical but structurally different trimers are named and colored in order of increasing distance from the five-fold axes of symmetry (P [red], Q [orange], R [green], S [yellow] and T [blue] situated at the three-fold axes). (Bottom left) The BTV 1 subcore shell (from X-ray crystallography) is composed of 120 copies of VP3, arranged with T=2 symmetry. The chemically identical but structurally different molecules are shown: A (green: surrounding the five-fold axis) and B (red: surrounding the three-fold axis). (Bottom right) Model cross-section of the BTV core showing packaging of the dsRNA as four concentric shells
(courtesy of D. I. Stuart, J. Grimes, P. Gouet, J. Diprose, R. Malby, P. Roy, B. P. V. Prasad and P. P. C. Mertens).

Figure 22 Phylogenetic tree of the T2 subcore shell proteins of members of the genus Orbivirus. The tree shows two groups: a mosquito-borne/tick-borne group where the second largest viral protein (VP2) forms the T2 sub-core capsid layer; and a Culicoides-borne group where the third largest viral protein (VP3) forms the T2-layer. Sequences were aligned using ClustalX and the tree constructed in MEGA4 using the neighbor-joining method and the P-distance algorithm. Branching is supported by bootstrap values >85%.
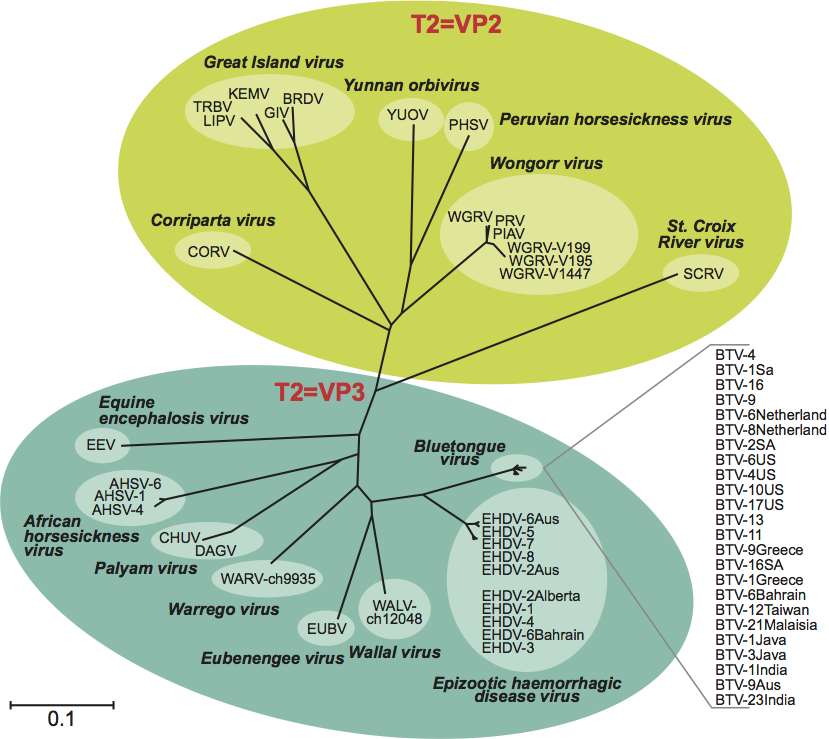
Figure 23 Phylogenetic tree, constructed using complete amino acid sequences of the cell attachment protein (VP2 of the Culicoides-borne viruses, VP3 of the mosquito-borne viruses and VP4 of Great Island virus, which is tick-borne). In bluetongue virus (the Orbivirus type species) VP2 is the larger of the two outer CPs and is the most variable and primary neutralization antigen. Amino acid sequences were aligned using Clustal X and the trees were constructed using the neighbor-joining method and P-distance in MEGA4 (courtesy of H. Attoui). Names and abbreviations correspond to those used in the list of species. Sequences were aligned using ClustalX and the tree constructed in MEGA4 using the neighbor-joining method and the P-distance algorithm. Branching is supported by bootstrap values >85%.
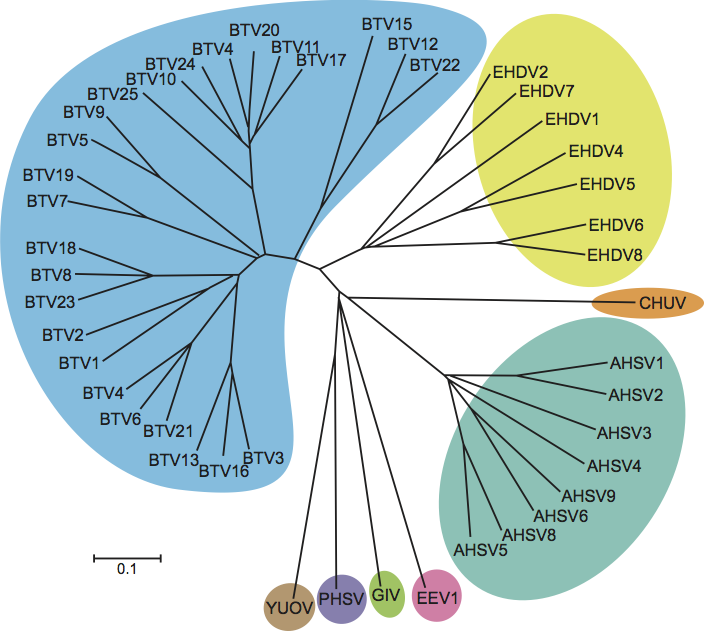
Figure 24 Rotavirus morphology. (Left panel) Cartoon representation of a rotavirus triple-layered particle, with proteins labeled. Black corkscrews represent segments of genomic dsRNA. The precise locations of VP1 and VP3 have not been determined. (Right panel) Electron micrograph of rotavirus particles viewed by negative staining. Bar represents 100 nm.
(Provided by B. V. V. Prasad.)
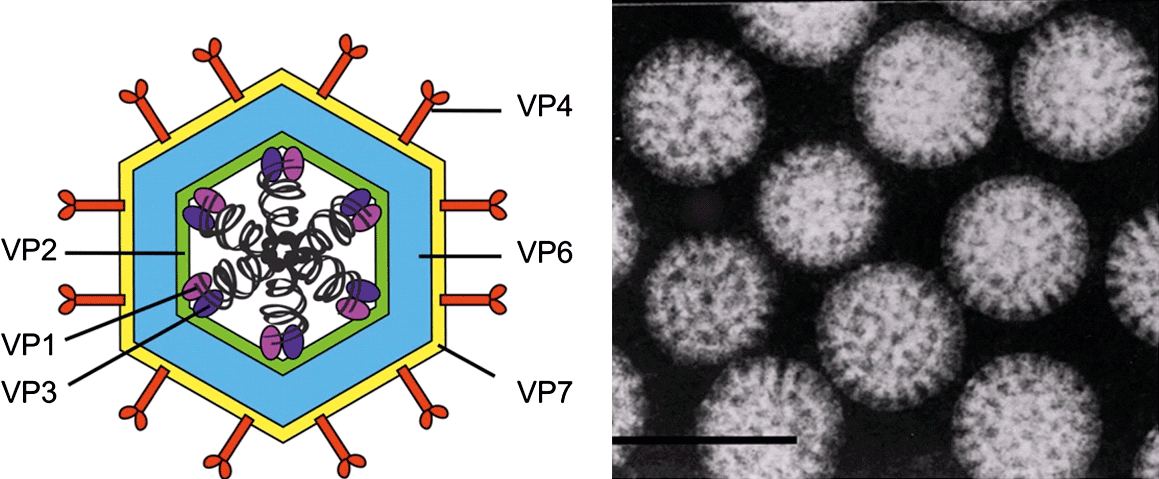
Figure 25 Structure and location of protein components of the rotavirus virion. A cutaway view of a cryoEM image reconstruction of a rotavirus virion at 9.5 resolution (center) is a reference for enlarged, high-resolution images of specific virus components. The particle and components are colored as follows: VP4 spikes (red), VP7 layer (yellow), VP6 layer (blue) and VP2 layer (green). A portion of VP2 that extends into the interior of the core (the hub) and transcriptional enzymes VP1 and VP3 are colored gold in the enlarged image of a five-fold vertex (bottom, center). The VP8* and VP5* cleavage products of the VP4 spike are indicated. The trimeric foot and dimeric stalk and head of VP4 can be seen. PDB files 3KZ4 (VP6 and VP2) and 3GZT (VP7) were used to make images.
(Courtesy of B. V. V. Prasad.)
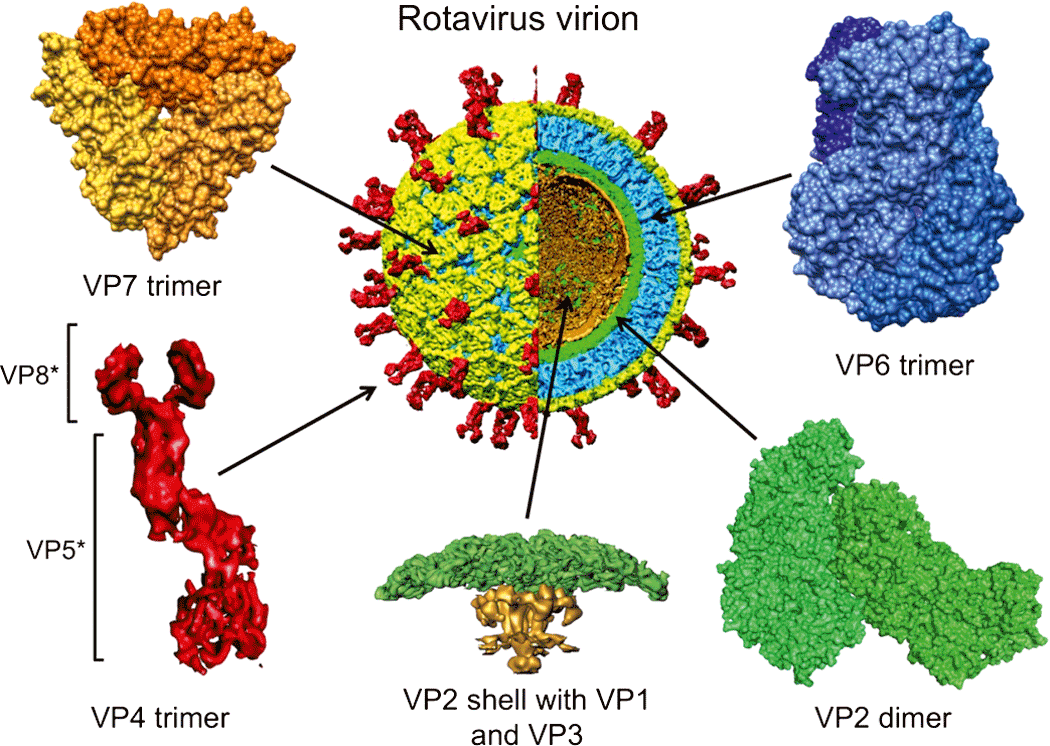
Figure 26 Phylogenetic tree comparing amino acid sequences of the rotavirus inner core shell protein VP2 (left) and intermediate capsid protein VP6 (right). Isolates and accession numbers used are: Bo/UKtc (P17462,P18610), Si/SA11 (CAA34733, AAO32085), Hu/Wa (X14942, P03530), Po/Gottfried (ADE44250, P16593), Mu/ETD_822 (ACY95261), Mu/EDIM (AAC57838), Av/PO-13 (BAA24147, BAA03836), Hu/WH-1 (AAT09117, AAT09116), Hu/Bang117 (ADF57895, ADF57898), Bo/DB176 (ADC53105, ADC53099), Mu/IDIR (AAA17401, QO1754), Hu/Bristol (CAC44890, CAA42504), Po/Cowden (P26191, AAA47097), Av/05V0049 (ADN06424, ADN06428), Hu/J19 (YP_392491, AAZ03490), and Hu/B219 (ABR32123, ABA60393). Sequences were aligned and trees calculated (neighbor-joining method) using MEGA 4 (courtesy of J. Matthijnssens). Abbreviations used to indicate host species are: Av, avian; Bo, bovine; Hu, human; Mu, murine; Po, porcine; Si, simian. Sequences were aligned using ClustalX and the tree constructed in MEGA4 using the neighbor-joining method and the P-distance algorithm. Branching is supported by bootstrap values >85%.
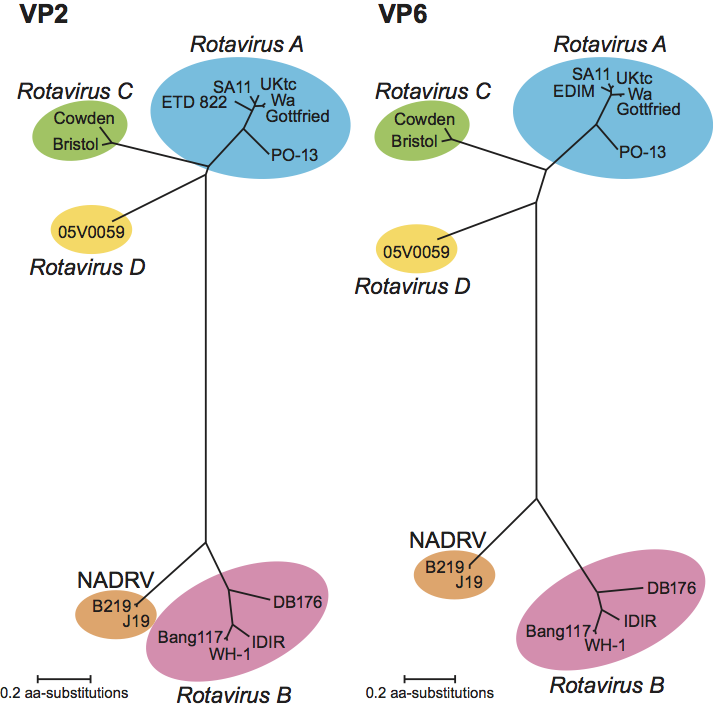
Figure 27 (Top) Diagram of the Banna virus (BAV) particle structure, constructed using data from biochemical analyses, electron microscopy and X-ray crystallography. (Bottom) Negative contrast electron micrograph of Banna virus particles: (left hand side) full BAV particles showing multiple of protein spikes; (right hand side) double layered cores of BAV
(courtesy of H. Attoui).
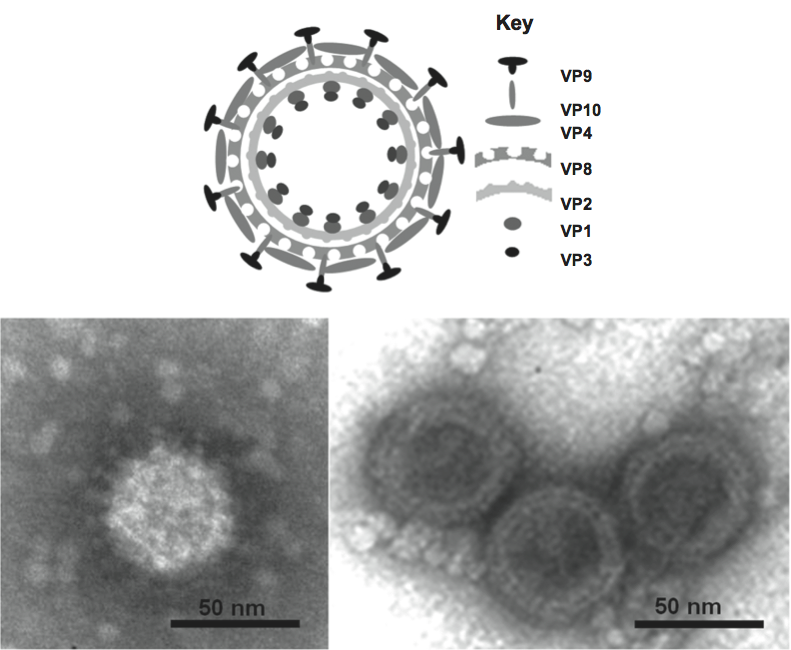
Figure 28 Migration patterns (electropherotypes) of genome segments from isolates of Banna virus (BAV), Kadipiro virus (KDV) and Liao ning virus (LNV) in 1% agarose gel. Electropherotype is thought to be characteristic of each virus species.

Figure 29 Phylogenetic tree for homologous genome segments from members of the species Banna virus, Kadipiro virus and Liao ning virus. The tree is based on outer-capsid and cell attachment protein sequences (encoded by Seg9 (VP9) of BAV, its homologous Seg11 (VP11) of KDV and Seg10 (VP10) of LNV). This tree also shows the two genotypes (serotypes) of BAV (A and B). The amino acid identity between VP9 of BAV and VP11 of KDV ranges between 14% and 16%. Between VP9 of BAV and VP10 of LNV amino acid identity ranges from 17% to 19%. Between VP11 of KDV and VP10 of LNV amino acid identities ranged from 17% to 18%. Between the two genotypes of BAV, it ranges from 41% to 43%. Within a given genotype of BAV it ranges between 90 and 96%. Between the two genotypes (serotypes) of LNV amino acid identity is 81%. Sequences were aligned using ClustalX and the tree constructed in MEGA4 using the neighbor-joining method and the P-distance algorithm. Branching is supported by bootstrap values >85%.
Figure 30 (Top left) Schematic diagram representing a T=13 capsid structure. (Top right) Negative contrast electron micrograph of rice gall dwarf virus particles, negatively stained with phosphotungstic acid. The bar represents 50 nm. (Bottom left) Electron cryo-microscopic image and 25 resolution 3D structure of the double shelled rice dwarf virus (RDV). (Bottom center) Inner shell computationally extracted with 59 nm diameter. It exhibits T=1 lattice. Dashed triangle designates one triangular face of the icosahedron. (Bottom right) Schematic diagram of fish-shaped density distribution within a triangle in a T=1 lattice.
(Courtesy of Hong Zhou and Wah Chiu, from Lu et al. (1998). J. Virol., 72, 85418549.)
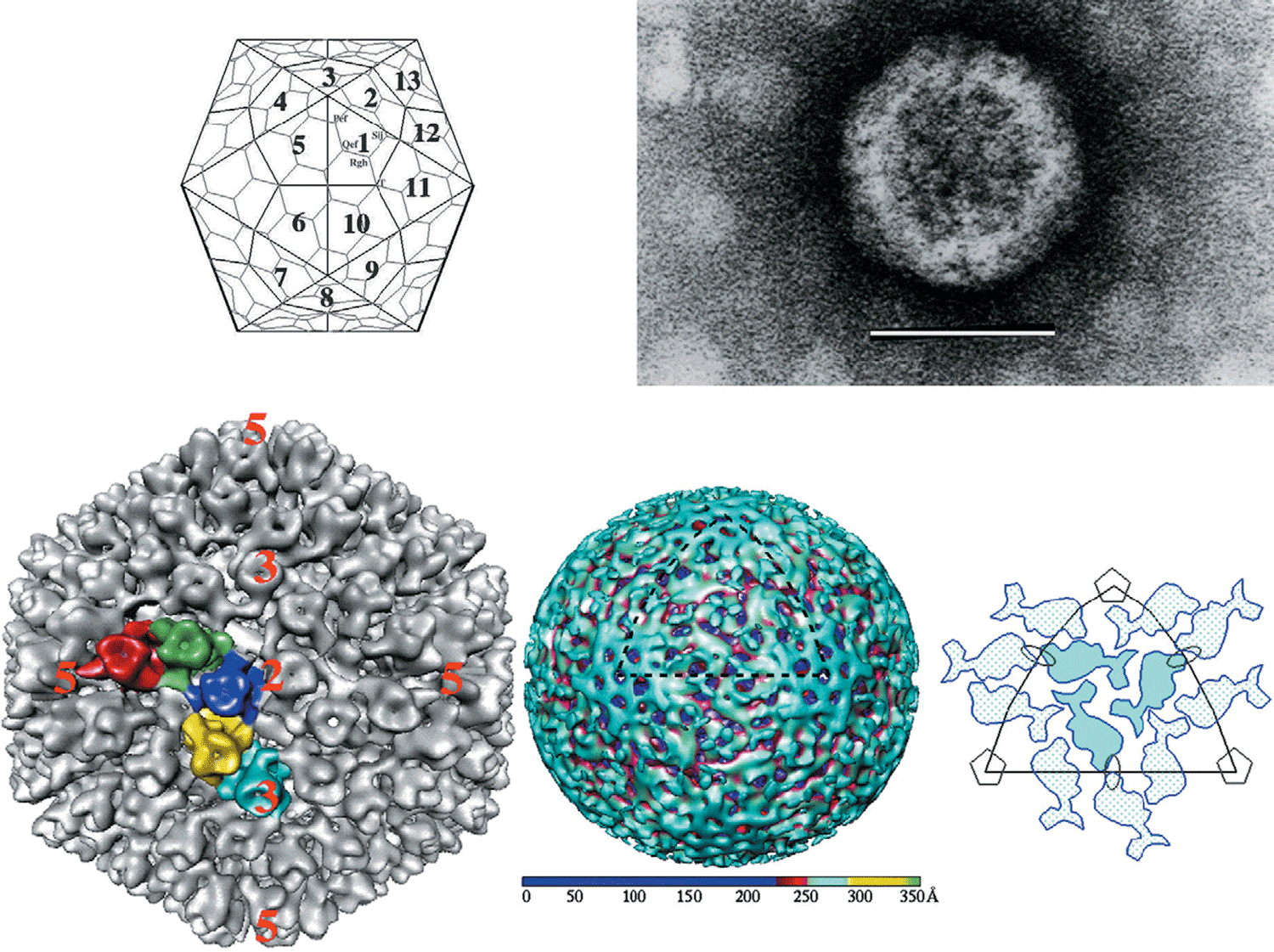
Figure 31 Phylogenetic (neighbor-joining) tree based on phytoreovirus Seg8 sequences from the following accession numbers HoVRV-BA (GU362071), HoVRV-FI (FJ497796), HoVRV-ME (GU369689), HoVRV-NC (GU384990), HoVRV-PA (GU370369), HoVRV-RI (GU350428), HoVRV-RO (GU395195), HoVRV-SC (GU390596), HoVRV-TI (GU437834), RDV-A (D10219), RDV-B (D00536), RDV-CN (U36565), RDV-S (D13773), RGDV-BL (AY999077), RGDV-CH (AY999078), RGDV-DQ (AY999079), RGDV-GD (AY216767), RGDV-GX (DQ364683), RGDV-GZ (AY999080), RGDV-TJ (D13410), RGDV-XY (AY999081), TLEV (AY587759), WTV (J04344). Tree was produced in MEGA4 (maximum composite likelihood distances) with 10,000 booststrap replicates (values shown when >60%).

Figure 32 Negative contrast electron micrograph of CsCl purified particles of Eriocheir sinensis reovirus.
(From Zhang et al. (2004). J. Fish Dis., 27, 687692.)
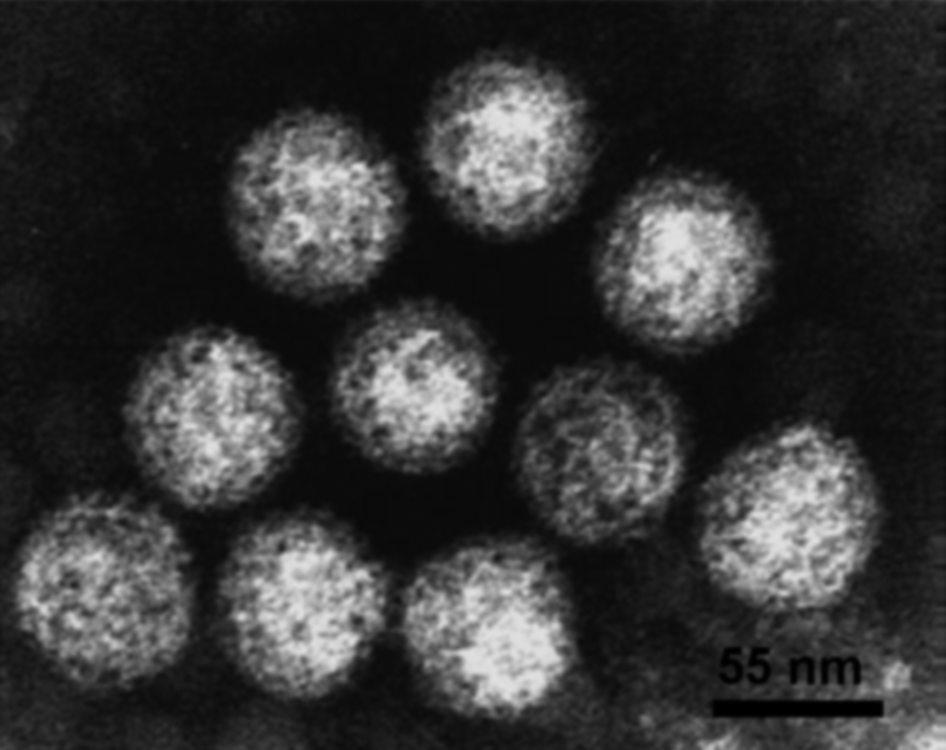
Figure 33 (A) MpRV virus particle purified on Percoll, with a diameter of 95 nm. A damaged particle is shown at the upper left corner, showing an outer layer about 15 nm thick (indicated by a white arrow), surrounding a more compact structure with a diameter of about 75 nm (indicated by a black arrow). (B) A virus particle purified by CsCl gradient centrifugation, with a diameter of about 75 nm. (C) Particles pelleted from the clarified lyaste of infected Micromonas pusilla. Some particles (indicated by arrow) have a larger diameter. (D) Particles generated by treating whole MpRV particles (purified on Percoll) with 1.5 M CaCl2 and subsequent purification on CsCl gradient, generating cores (or sub-cores) with smooth outline.
(Courtesy of H. Attoui and C. Brussaard.)
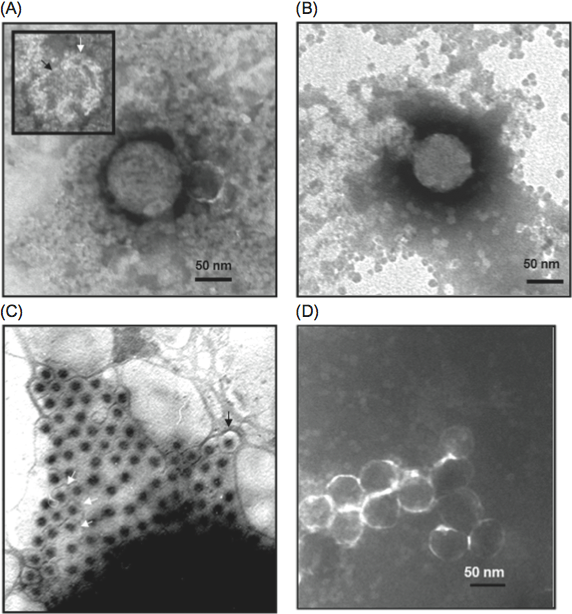
Figure 34 Agarose gel electrophoretic profile (electropherotype) of genome segments of MpRV in 1% agarose gel. These migration patterns (electropherotype) are thought to be characteristic of each virus species.
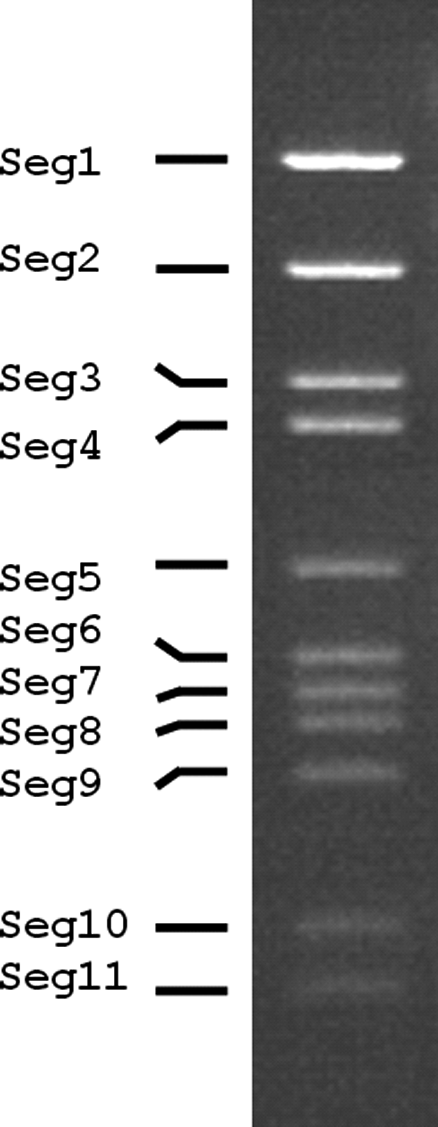
Figure 35 Neighbor joining tree constructed with the AA sequences of putative RdRp of representative viruses from the following genera of the family Reoviridae [accession numbers]: Seadornavirus, Banna virus: isolate BAV-Ch [AF168005], Kadipiro virus: isolate KDV-Ja7075 [AF133429], Liao ning virus: isolate LNV-NE9731 [AY317099]. Coltivirus, Colorado tick fever virus, isolate CTFV-Fl [AF134529], Eyach virus, isolate EYAV-Fr578 [AF282467]. Orthoreovirus, Mammalian orthoreovirus, serotype-1 (MRV-1) [M24734], serotype-2 (MRV-2) [M31057], serotype-3 (MRV-3) [M31058], serotype-4 (MRV-4) also known as Ndelle virus [AF368033]. Aquareovirus, Aquareovirus C, isolate golden shiner virus (GSRV) [AF403399], grass carp reovirus (GCRV) [AF260512], Aquareovirus A, isolate striped bass reovirus (SBRV) [AF450318], isolate chum salmon reovirus (CSRV) [AF418295], Aquareovirus G isolate golden ide reovirus (GIRV) [AF450323]. Orbivirus, African horse sickness virus, serotype-9 (AHSV-9) [U94887], Bluetongue virus, serotype-2 (BTV-2) [L20508], serotype-10 (BTV-10) [X12819], serotype-11 (BTV-11) [L20445], serotype-13 (BTV-13) [L20446], serotype-17 (BTV-17) [L20447], species Palyam virus, isolate CHUV [Baa76549], St Croix River virus, isolate SCRV[AF133431]. Rotavirus, Rotavirus A, strain BoRV-A/UK [X55444], strain SiRV-A/SA11 [AF015955], Rotavirus B, strain Hu/MuRV-B/IDIR [M97203], Rotavirus C, strain PoRV-C/Co [M74216], Fijivirus, species Nilaparvata lugens reovirus, strain NLRV-Iz [D49693]. Phytoreovirus, species Rice dwarf virus, isolate RDV-Ch [U73201], isolate RDV-H [D10222], isolate RDV-A [D90198]. Mycoreovirus, species Mycoreovirus-1, isolate CpMYRV-1 [AY277888], species Mycoreovirus-3, isolate RnMYRV-3 [AB102674]. Oryzavirus, isolate Rice ragged stunt virus, strain RRSV-Th [U66714]. Cypovirus, Bombyx mori cytoplasmic polyhedrosis virus-1 strain BmCPV-1 [AF323781], Dendrlymus punctatus cytoplasmic polyhedrosis virus-1 strain DsCPV-1 [AAN46860], Lymantria dispar cytoplasmic polyhedrosis virus-14 strain LdCPV-14 [AAK73087]. Dinovernavirus, species Aedes pseudoscutellaris reovirus, isolate APRV [DQ087276]. Cardoreovirus, species Eriocheir sinensis reovirus, isolate ESRV [AY542965]. Mimoreovirus, Micromonas pusilla reovirus, isolate MPRV [DQ126101]. Values at the nodes represent bootstrap confidence levels (500 replications).